Abstract
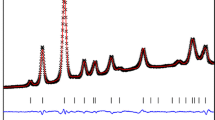
Platy (Te Puke, New Zealand), cylindrical (Spruce Pine, North Carolina), and spherical (North Gardiner mine, Huron, Lawrence County, Indiana) halloysite samples were analyzed by high-pressure differential thermal analysis (HP-DTA) to determine the effect of morphological and chemical differences on their respective thermal stability. In halloysite, these morphological differences imply structural features. The metastable phase relations of each are analogous to those of kaolinite. At 1 bar, the platy, cylindrical, and spherical samples showed peak temperatures (maximum deflection in the dehydroxylation endotherm) of 560°, 578°, and 575°C, respectively, whereas at about 600 bars the peak temperatures were 622°, 655°, and 647°C. At low pressures the observed reaction is related to dehydroxylation: halloysite (H) metahalloysite (MH) + vapor (V), whereas higher pressures produce melting reactions, either H + V = metaliquid (ML) for conditions of P(H2O) = P(total), or H + MH = ML for P(H2O) < P(total). The PT conditions of the invariant point, H + MH + ML + V, for each system are: Te Puke, 612° ± 4°C, 25 ± 7 bars; Spruce Pine, 657 ± 2°C, 30 ± 7 bars; North Gardiner, 652° ± 2°C, 34 ± 7 bars. The lower thermal stability of the Te Puke sample may be related to its higher iron content, although additional data are necessary to confirm that it is not related also to the platy structure. Furthermore, morphological differences between the cylindrical and spherical varieties appear to have had little effect on the energy required to dehydroxylate these halloysite structures. Exceptionally high values obtained for the dehydroxylation enthalpies using the van’t Hoff equation, compared with values derived using other methods, may be explained by a 10–15-bar excess in the intracrystalline H2O fugacity during dehydroxylation. Intracrystalline fugacity is defined here as the H2O fugacity within crystallites and is not related to the partial pressure of H2O around individual particles.
Similar content being viewed by others
References
Alexander, L. T., Faust, G. T., Hendricks, S. B., Insley, H., and McMurdie, H. F. (1943) Relationship of the clay minerals halloysite and endellite: Amer. Mineral. 28, 1–18.
Anderson, G. M. (1977) Fugacity, activity and equilibrium constant: in Application of Thermodynamics to Petrology and Ore Deposits, Mineralogical Association of Canada Short Course Handbook 2, H. J. Greenwood, ed., 17–37.
Askenasy, P. E., Dixon, J. B., and McKee, T. R. (1973) Spheroidal halloysite in a Guatemalan soil: Soil Sci. Amer. Proc. 37, 799–803.
Bates, T. F. (1959) Morphology and crystal chemistry of 1:1 layer lattice silicates: Amer. Mineral. 44, 78–114.
Bates, T. F., Hildebrand, F. A., and Swineford, A. (1950) Morphology and structure of endellite and halloysite: Amer. Mineral. 35, 463–484.
Bramao, L., Cady, J. G., Hendricks, S. B., and Swerdlow, M. (1952) Criteria for the characterization of kaolinite, halloysite, and a related mineral in clays and soils: Soil Science 73, 273–287.
Brindley, G. W. and Robinson, K. (1946) Randomness in the structures of kaolinitic clay minerals: Trans. Faraday Soc. 42, 198–205.
Chukhrov, F. V. and Zvyagin, B. B. (1966) Halloysite, a crystallochemically and mineralogically distinct species: in Proc. Int. Clay Conf., Jerusalem, 1966, Vol. 1, L. Heller and A. Weiss, eds., Israel Prog. Sci. Transi., Jerusalem, 11–25.
Churchman, G. J. and Theng, B. K. G. (1984) Interactions of halloysites with amides: Mineralogical factors affecting complex formation: Clay Miner. 19, 161–175.
Dixon, J. B. and McKee, T. R. (1974) Internal and external morphology of tubular and spheroidal halloysite particles: Clays & Clay Minerals 22, 127–137.
Farmer, V. C. and Russell, J. D. (1964) The i.r. spectra of layer silicates: Spectrochim. Acta 20, 1149–1173.
Hofmann, U., Endell, K., and Wilm, D. (1934) Röntgeno-graphische und kolloidchemische Untersuchungen über Ton: Angew. Chem. 47, 539–547.
Holloway, J. R. (1971) Internally heated pressure vessels: in Research for High Pressure and Temperature, G. C. Ulmer, ed., Springer-Verlag, New York, 217–258.
Hope, E. W. and Kittrick, J. A. (1964) Surface tension and the morphology of halloysite: Amer. Mineral. 49, 859–866.
Hughes, I.R. (1966) Mineral changes of halloysite on drying: New Zealand J. Sci. 9, 103–113.
Koster van Groos, A. F. (1979) Differential thermal analysis of the system NaF-Na2COj to 10 kbar: J. Phys. Chem. 83, 2976–2978.
Koster van Groos, A. F. and ter Heege, J. P. (1973). The high-low quartz transition up to 10 kilobar pressure: J. Geol. 81, 281–286.
Kunze, G. W. and Bradley, W. F. (1964) Occurrence of a tabular halloysite in a Texas soil: in Clays and Clay Minerals, Proc. 12th Natl. Conf, Atlanta, Georgia, 1963, W. F. Bradley, ed., Pergamon Press, New York, 523–527.
McKee, T. R., Dixon, J. B., Whitehouse, G., and Harling, D. F. (1973) Study of Te Puke halloysite by a high resolution electron microscope: Abstr. 31st Ann. Electron Microscopy Soc. Amer. Meeting, New Orleans, Louisiana, 1973, 200–201.
Noro, H. (1986) Hexagonal platy halloysite in an altered tufFbed, Komaki City, Aichi Prefecture, central Japan: Clay Miner. 21, 401–415.
Pampuch, R. (1966) Infrared study of thermal transfermation of kaolinite and the structure of metakaolin: Polska Akad. Nauk Prace Mineral. 6, 53–70.
Radoslovich, E. W. (1963) The cell dimensions and symmetry of layer-lattice silicates, VI. Serpentine and kaolin morphology: Amer. Mineral. 48, 368–378.
Ross, C. S. and Kerr, P. F. (1934) Halloysite and allophane: U.S. Geol. Surv. Prof. Pap. 185-G, 135–148.
Roy, R. and Osborn, E. F. (1954) The system Al2O3-SiO2-H2O: Amer. Mineral. 39, 853–885.
Stone, R. L. (1952) Differential thermal analysis of kaolin-group minerals under controlled partial pressures of H2O: J. Amer. Ceram. Soc. 35, 90–99.
Sunderman, J. A. (1963) Mineral deposits at the Mississip-pian-Pennsylvanian unconformity in southwestern Indiana: M.S. thesis, Indiana University, Bloomington, Indiana, 115 pp.
Tazaki, K. (1982) Analytical electron microscopic studies of halloysite formation processes—Morphology and composition of halloysite: in Proc. Int. Clay Conf., Bologna, Pavia, 1981, H. van Olphen and F. Veniale, eds., Elsevier, Amsterdam, 573–584.
Weber, J. N. and Roy, R. (1965a) Dehydroxylation of kaolinite, dickite, and halloysite: J. Amer. Ceram. Soc. 48, 309–311.
Weber, J. N. and Roy, R. (1965b) Dehydroxylation of kaolinite, dickite and halloysite: Heats of reaction and kinetics of dehydration at P(H2O) = 15 psi: Amer. Mineral. 50, 1038–1045.
Yeskis, D., Koster van Groos, A. F., and Guggenheim, S. (1985) The dehydroxylation of kaolinite: Amer. Mineral. 70, 159–164.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Johnson, S.L., Guggenheim, S. & Koster van Groos, A.F. Thermal Stability of Halloysite by High-Pressure Differential Thermal Analysis. Clays Clay Miner. 38, 477–484 (1990). https://doi.org/10.1346/CCMN.1990.0380503
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1990.0380503