Abstract
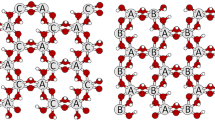
The recent determination of the three-dimensional crystal structure of a dickite:formamide intercalate allows insight into the clay:organic bonding schemes of amides and other small organic molecule intercalates of the kaolin minerals. It is demonstrated that the observed basal spacings of intercalates with these molecules are consistent with hydrogen bonding schemes in which, if possible, triple hydrogen bonds from the clay hydroxyls to 0=C> are formed. Variations in basal spacing within a series of amides can be explained by considering the maximization of hydrogen bonding while avoiding close van der Waals contacts.
Резюме
Недавнее определение трехразмерной кристаллической структуры включений диккита: формамида позволяет детальнее изучить глину: органические связывающие схемы амидов и других малых органических молекулярных включений каолиновых минералов. Показывается, что наблюдаемые основные промежутки между включениями и этими молекулами согласуются с водородными связывающими схемами, в которых, если возможно, из глинистых гидроксилов формируются тройные водородные связи 0=с. Различия в основных промежутках в пределах серий амидов могут быть объяснены максимизацией водородных связей при отсутствии тесных контактов Ван дер Ваалса.
Kurzreferat
Die neue Bestimmung der dreidimensionalen Kristallstruktur einer Dickit-Formamideinbettung erlaubt Einblicke in die Bindungsweise der Einbettungen von organischen Amiden und anderen kleinen organischen Molekülen mit Kaolinmineralien. Es wurde gezeigt, daß die erhaltenen Basisabstände von Einbettungen mit diesen Molekülen im Einklang mit Wasserstoffbrückenschemen sind, in welchen, wenn möglich, dreifache Wasserstoffbrücken von den Tonhydroxylgruppen zu den Karbonylgruppen geformt werden. Variationen in den Basisabständen innerhalb einer Serie von Amiden können erklärt werden, indem die Maximisation von Wasserstoffbrücken und zur selben Zeit das Vermeiden von zu nahen van der Waalschen Kontakten berücksichtigt werden.
Similar content being viewed by others
References
Adams, J. M. and Jefferson, D. A. (1976) The crystal structure of a dickite:formamide intercalate (Al2Si2O5(OH)4HCONH2): Acro Crystallogr. B32, 1180–1183.
Adams, J. M., Pritchard, R. G. and Thomas, J. M. (1976) Preparation and X-ray crystal structure of guamdinium oxalate dihydrate mono-perhydrate: A novel example of crystal engineering: Chem. Commun. 358–359.
Fennol Hach-Ali, P. and Weiss, A. (1969) Estudio de la reaction de caolinita y N-metilformamida: An. Quim. 65, 769–790.
Haase, D. J., Weiss, E. J. and Steinfink, H. (1963) The crystal structure of a hexarnethylenediamme-vermiculite complex: Am. Mineral. 48, 261–270.
Iglesias, J. E. and Steinfink, H. (1974) A structural investigation of a vermiculite-piperidine complex: Clays & Clay Minerals 22, 91–95.
Kanamaru, F. and Vand, V. (1970) The crystal structure of a clay-organic complex of 6-aminohexanoic acid and vermiculite: Am. Mineral. 55, 1550–1561.
Ladell, J. and Post, B. (1954) The crystal structure of formamide: Acta Crystallogr. 7, 559–564.
Ledoux, R. L. and White, J. L. (1966a) Infrared studies of hydrogen bonding interaction between kaolinite surfaces and intercalated potassium acetate, hydrazine, formamide and urea: J. Colloid Interface Sci. 21, 127–152.
Ledoux, R. L. and White, J. L. (1966b) Infrared studies of hydrogen bonding of organic compounds on oxygen and hydroxyl surfaces of layer lattice silicates: Proc. Int. Clay Conf. Jerusalem 1, 361–374.
Olejnik, S., Aylmore, L. A. G., Posner, A. M. and Quirk, J. P. (1968) Infrared spectra of kaolin mineral dimethyl sulphoxide complexes: J. Phys. Chem. 72, 241–249.
Olejnik, S., Posner, A. M. and Quirk, J. P. (1970) The intercalation of polar organic compounds into kaolinite: Clay Miner. 8, 421–434.
Olejnik, S., Posner, A. M. and Quirk, J. P. (1971) Infrared spectrum of the kaolinite-pyridine N-oxide complex: Spectrochim. Acta 27A, 2005–2009.
Olejnik, S., Posner, A. M. and Quirk, J. P. (1971) The I.R. spectra of interlamellar kaolinite-amide complexes—I. The complexes of formamide, N-methyl formamide and dimethylformamide: Clays & Clay Minerals 19, 83–94.
Pimentel, G. S and McClellan, A. L. (1960) The Hydrogen Bond: Freeman, London.
Sanchez Camazano, M. and Gonzalez Garcia, S. (1966) Complejos interlaminares de caolinita u haloisita con liquidos polares: An. Eda-fol. Agrobiol. 25, 9–25.
Susa, K., Steinfink, H. and Bradley, W. F. (1967) The crystal structure of a pyridine:vermiculite complex: Clay Miner. 7, 145–153.
Weiss, A. and Orth, H. (1973) Layer intercalation compounds of kaolinite, nacrite, dickite and halloysite with pyridine N-oxide and pic-oline N-oxide: Z. Naturforsch. B28, 252–254.
Weiss, A., Thielepape, W., Goring, G., Ritter, W. and Shafer, H. (1963) Kaolinit-Einlagerungs-Verbindungen: Proc. Int. Clay Conf. Stockholm I, 287–305.
Weiss, A., Thielepape, W. and Orth, H. (1966) Neue Kaolinit-Einla-gerungouerbindungen: Proc. Int. Clay Conf. Jerusalem I, 277–293.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Adams, J.M. Unifying Features Relating to the 3D Structures of some Intercalates of Kaolinite. Clays Clay Miner. 26, 291–295 (1978). https://doi.org/10.1346/CCMN.1978.0260406
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1978.0260406