Abstract
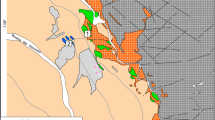
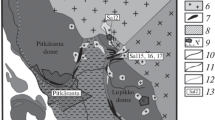
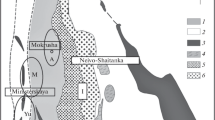
Micas are described from the kaolin type Tertiary weathering crusts formed on granites and gneisses of Lower Silesia, Poland. In the weathering crust, large scale processes of transformation of micas (muscovite, biotite) take place. Weathering of micas is a gradual transformation of their structures by removal of the mobile cations Mg2+ Fe2+ Fe3+ and finally K +. Muscovite transforms into kaolinite through the intergrowths of two phases, mica and kaolinite. A degradation series, muscovite → mica/montmorillonite → montmorillonite → kaolinite also is observed. Biotite passes directly into kaolinite, but at higher concentrations of K and Al one observes the following sequence of biotite degradation: biotite → dioctahedral mica rich in Fe → dioctahedral mica poor in Fe → kaolinite. This process involves the exchange of Mg2+ and then Fe2+ of octahedral layers by Al3+ less mobile under weathering conditions. In the next stage of degradation, K + removal and kaolinite formation takes place. The micas representing different stages of this transformation, dark green, light-green, and silver-white, have been separated and investigated.
Similar content being viewed by others
References
Barany, R. (1964) Heat and free energy of formation of muscovite: U.S. Bur. Mines, Rep. Invest. 6356.
Barshad, I. and Kishk, J. M. (1968) Oxidation of ferrous iron in vermiculite and biotite alters fixation and replaceability of potassium: Science 162, 1401–1402.
Budkiewicz, M. 1964 Zloza kaolinu w Polsce: Przegl. Geol. 12, 207–208.
Farmer, V. C., Russell, J. D., McHardy, W. J., Newman, A. C. D., Ahlichs, J. L. and Rimsaite, J. Y. H. (1971) Evidence for loss of protons and octahedral iron from oxidized biotites and vermiculites: Miner. Mag. 38, 121–137.
Foster, M. D. (1960) Interpretation of the composition of trioctahedral micas: U.S. Geol. Surv. Prof. Papers 354-B, 11–49.
Garrels, R. M. and Christ, Ch. L. (1965) Solutions, Minerals, Equilibria. Harper & Row, New York.
Garrels, R. M. and Howard, P. (1959) Reaction of feldspar and mica with water at low temperature and pressure: Clays & Clay Minerals 6, 68–88.
Ginzburg, I. I. (1963) Tipy drevnich kor vyvetrivanija 6. Izd. AN SSSR 71–101.
Hess, P. C. (1966) Phase equilibria of some minerals in the K2O-Na2O-Al2O3-SiO2-H2O system at 25°C and 1 atmosphere: Am. J. Sci. 264, 289–309.
Konta, J., Borovec, Z., Šramek, J. and Tolar, VI. (1972) Changes of primary biotite and muscovite during kaolinization of granites, Carlsbad Area, Czechoslovakia: 5th Conf. Clay Miner. Petrol. pp. 27–43. Praha 1970.
Kukovsky, E. G. (1969) Alteration processes in clay minerals: Clay Miner. 8, 234–237.
Leonard, R. A. and Weed, S. B. (1970) Effect of potassium removal on the b-dimension of phlogopite: Clays & Clay Minerals 18, 197–202.
Petrov, V. P. (1967) Osnovy učenija o drevnich korach vyvetrivanija. Nedra, Moskva.
Reesman, A. L. and Keller, W. D. (1968) Aqueous solubility studies of high-alumina and clay minerals: Am. Miner. 53, 929–942.
Rossini, F. D., Wagman, D. D., Ewans, W. A., Jaffe, J. and Levine, S. (1952) Selected values of chemical thermodynamic properties: U.S. Nat. Bur. Stand. Circ. 500.
Sikora, W. and Stoch, L. (1972) Mineral forming processes in weathering crusts of acid magmatic and metamorphic rocks of Lower Silesia: Mineralogia Polonica 3, 39–52.
Stoch, L. (1960) Intergrowth of kaolinite and muscovite in clays from Boleslawiec region, Lower Silesia: Bull. Acad. Pol. Sci., sér. sci. géol. géogr. 8, 21–29.
Tardy, Y. (1971) Characterization of the principal weathering types by the geochemistry of waters from some European and African crystalline massifs: Chem. Geol. 7, 253–276.
Taylor, H. F. W. (1960) Aspects of the crystal structures of calcium silicates and aluminates: J. appl. Chem. 10, 317–323.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stoch, L., Sikora, W. Transformations of Micas in the Process of Kaolinitization of Granites and Gneisses. Clays Clay Miner. 24, 156–162 (1976). https://doi.org/10.1346/CCMN.1976.0240402
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1976.0240402