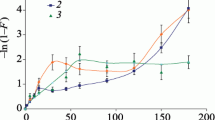
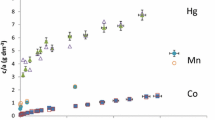
Abstract
The exchange of [Co(NH3)6]3+ from its vermiculite complex has been carried out with a number of inorganic and alkyl quaternary ammonium ions of varying ionic sizes. The distribution and selectivity coefficients of the desorbing ions increase in the order: Li>Na>NH4>K>Rb>H>Cs for the monovalent, Ca>Mg for the bivalent and (C2H5)4N>(CH3)4N≪CTA (cetyl trimethyl ammonium) >CP (cetyl pyridinium) for the organic ions. The AG° values of some of the cation exchange reactions have been calculated by an equation of Kielland (1935).
Similar content being viewed by others
References
Barrer, R. M., Buser, W. and Grütter, W. F. (1956) Synthetic “faujasit”—I: Properties and base exchange character: Helv. Chim. Acta. 39, 518–530.
Barrer, R. M. and Falconer, J. D. (1956) Ion exchange in felspathoids as a solid state reaction: Proc. R. Soc. London A236, 227–249.
Barshad, I. (1954) Cation exchange in micaceous minerals —II. Replaceability of ammonium and potassium from vermiculite, biotite, and montmorillonite: Soil Sci. 78, 57–76.
Beckett, P. H. T. (1964) Soil potassium—I: Confirmation of the ratio law: measurement of potassium potential: J. Soil Sci. 15, 1–8.
Conway, B. E. (1952) Electro-chemical Data pp. 62; 76–78. Elsevier, Amsterdam.
Das Kanungo, J. L. and Chakravarti, S. K. (1971) Evaluation of thermodynamic quantities etc. in the cation exchange of Mg2+ from magnesium saturated bentonite and vermiculite: Kolloid Z. 248, 953–958.
Das Kanungo, J. L. and Chakravarti, S. K. (1971) Behavior of quaternary ammonium ions in the desorption of [Coen3]3+ from H-Coen3-bentonite: J. Colloid Interface Sci. 35, 295–299.
De, D. K. (1968) Studies on sorption and desorption of dyes on aluminosilicates: Ph.D. Thesis, University of Kalyani, India.
Garrett, W. G. and Walker, G. F. (1962) Swelling of some vermiculite organic complexes in water: Proc. Nat. Conf. Clays and Clay Minerals 9, 557–567.
Ghosal, D. N. and Mukherjee, S. K. (1972) Studies on the sorption and desorption of crystal violet on and from bentonite and kaolinite: J. Ind. Chem. Soc. 49, 569–572.
Gruner, J. W. (1934) The structures of vermiculites and their collapse by dehydration: Am. Miner. 19, 557–575.
Helfferich, F. (1962) Ion Exchange p. 186. McGraw-Hill, New York.
Hendricks, S. B. and Alexander, L. T. (1940) Semi quantitative estimation of montmorillonite in clays: Soil Sci. Soc. Am. Proc. 5, 95–99.
Jenkins, I. L. and Monk, C. B. (1951) The conductivities of some complex chlorides and sulphates: J. Chem. Soc. 68–73.
Kielland, J. (1935) Thermodynamics of base exchange equilibria of some different clays: J. Soc. Chem. Ind. London, 54, 232–247.
Kielland, J. (1937) Individual coefficient of ion in aqueous solution: J. Am. Chem. Soc. 59, 1675–1678.
Peterson, F. F., Rhoades, J., Arca, M. and Coleman, N. T. (1965) Selective adsorption of Magnesium ions by vermiculite: Soil Sci. Soc. Am. Proc. 29, 327–328.
Ram Gopal and Rastogi, A. K. (1966) Abnormal partial ionic volumes of some tetra alkyl ammoniun ions in water: J. Ind. Chem. Soc. 43, 269–271.
Sutherland, H. H. and MacEwan, D. M. C. (1961) Organic complexes of vermiculite: Clay Min. Bull. 4, 229–233.
Weiss, A., Mehler, A. and Hofmann, U. (1956) Zur Kenntnis von organophilem vermikulit: Z. Naturf. 11b, 431–434.
Wild, A. and Keay, J. (1964) Cation exchange equilibria with vermiculite: J. Soil Sci. 15, 135–144.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Das Kanungo, J.L. A Study on the Cation Exchange of [Co(NH3)6]3+ from H-Co (NH3)6-Vermiculite by Inorganic and Quaternary Ammonium Ions. Clays Clay Miner. 23, 361–364 (1975). https://doi.org/10.1346/CCMN.1975.0230506
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1975.0230506