Abstract
An experimental program was conducted to investigate the water-vapor sorption characteristics of smectite and kaolinite mixtures. End-member smectite and kaolinite were slurry-mixed together at mass-controlled ratios corresponding to 0%, 20%, 50%, 70%, 80%, 90% and 100% smectite. Vapor desorption isotherms for the mixtures were measured at 24°C for relative humidity (RH) ranging from ∼95% to 0%.
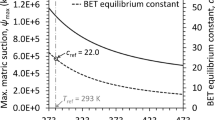
Results show that the amount of water adsorbed by the clay mixtures at a given RH increases systematically with increasing smectite content. Derivative analysis of the sorption isotherms shows evidence of transitions between the two-, one- and zero-layer hydrate-states for the smectite-rich mixtures. The transitions become less apparent as the smectite content decreases. Monolayer coverage, specific surface area, and heat of adsorption were estimated from the isotherms using BET theory. It is shown that monolayer coverage and specific surface for the clay mixtures can be reasonably approximated by weighted averaging of the end-member clay properties. General methodologies are presented for predicting the sorption behavior (i.e. soil-water characteristics) and effective specific surface area from measurements of the end-member sorption isotherms.
Similar content being viewed by others
References
ASTM (2000) Annual Book of ASTM Standards, 4.08, 4.09, D-18 Committee on Soils and Rock, West Conshohocken, PA.
Barshad, I. (1949) The nature of lattice expansion and its relation to hydration in montmorillonite and vermiculite. American Mineralogist, 34, 675–684.
Berend, I., Cases, J., Francois, M., Uriot, J., Michot, L., Maison, A. and Thomas, F. (1995) Mechanism of adsorption and desorption of water vapor by homoionic montmorillonites. Clays and Clay Minerals, 43, 324–336.
Brunauer, S. (1945) The Adsorption of Gases and Vapors: Physical Adsorption. Princeton University Press, New Jersey, 511 pp.
Brunauer, S., Emmett, P.H. and Teller, E. (1938) Adsorption of gases in multimolecular layers. Journal of the American Chemical Society, 60, 309.
Cases, J.M., Berend, I., Besson, G., Francois, M., Uriot, J.P., Thomas, F. and Poirier, J.E. (1992) Mechanism of adsorption and desorption of water vapor by homoionic montmorillonite. I. The sodium exchanged form. Langmuir, 8, 2730–2739.
Chipera, S.J, Carey, J.W. and Bish, D.L. (1997) Controlled-humidity XRD analyses: application to the study of smectite expansion/contraction. Advances in X-ray Analysis, 39, 713–721.
Collis-George, N. (1955) The hydration and dehydration of Na-montmorillonite (Belle Fourche). Journal of Soil Science, 6, 99–110.
Del Pennino, U., Mazzega, E., Valeri, S., Alietti, A., Franca-Brigatti, M. and Poppi, L. (1981) Interlayer water and swelling properties of monoionic montmorillonites. Journal of Colloid and Interface Science, 84, 301.
Gillery, F. H. (1959) Adsorption-desorption characteristics of synthetic montmorillonoids in humid atmospheres. American Mineralogist, 44, 806.
Jo, H., Katsumi, T., Benson, C.H. and Edil, T.B. (2001) Hydraulic conductivity and swelling of non-prehydrated GCLs permeated with single-species salt solutions. Journal of Geotechnical and Geoenvironmental Engineering, 111, 557–567.
Karaborni, S., Smit, B., Heidug, W. and van Oort, E. (1996) The swelling of clays: molecular simulations of the hydration of montmorillonite. Science, 111, 1102.
Keenan, A.G., Mooney, R.W. and Wood, L.A. (1951) The relation between exchangeable ions and water adsorption on kaolinite. Journal of Physics and Colloid Chemistry, 55, 1462–1474.
Keren, R. and Shainberg, I. (1975) Water vapor isotherms and heat of immersion of Na/Ca-montmorillonite systems-I: homoionic clay. Clays and Clay Minerals, 13, 193–200.
Keren, R. and Shainberg, I. (1979) Water vapor isotherms and heat of immersion of Na/Ca-montmorillonite systems-II: Mixed systems. Clays and Clay Minerals, 27, 145–151.
Likos, W.J. (2000) Total suction-moisture content characteristics for expansive soils. Ph.D. dissertation, Colorado School of Mines, Golden, Colorado, 182 pp.
Likos, W.J. and Lu, N. (2001) Automated measurement of total suction characteristics in the high suction range: application to the assessment of swelling potential. Journal of the Transportation Research Board, 1155, 119–128.
Likos, W.J. and Lu, N. (submitted) An automated humidity system for measuring total suction characteristics of clays. Geotechnical Testing Journal (in press).
Lowell, S. (1979) Introduction to Powder Surface Area. J. Wiley and Sons, New York, 199 pp.
Mesri, G. and Olson, R.E. (1971) Mechanisms controlling the permeability of clays. Clays and Clay Minerals, 19, 151–158.
Mitchell, J.K. (1993) Fundamentals of Soil Behavior. J. Wiley, New York, 437 pp.
Mooney, R.W, Keenan, A.G. and Wood, L.A. (1952) Adsorption of water vapor by montmorillonite. II. Effect of exchangeable ions and lattice swelling as measured by X-ray diffraction. Journal of the American Chemical Society, 14, 1371–1374.
Moore, D.M. and Reynolds, R.C., Jr. (1997) X-ray Diffraction and the Identification and Analysis of Clay Minerals. Oxford University Press, New York, 377 pp.
Parker, J.C. (1986) Hydrostatics of water in porous media. Pp. pp. 209–296 in: Soil Physical Chemistry (D.L., Sparks, editor). CRC Press, Inc., Boca Raton, Florida.
Quirk, J.P. (1955) Significance of surface area calculated from water vapor sorption isotherms by use of the BET equation. Soil Science, 80, 423–430.
Rumer, R.R. and Mitchell, J.K., editors (1995) Assessment of Barrier Containment Technologies. International Containment Technology Workshop, Baltimore, Maryland, 437 pp.
Van Olphen, H. (1965) Thermodynamics of interlayer adsorption of water in clays. Journal of Colloid Science, 10, 822–837.
Van Olphen, H. (1991) An Introduction to Clay Colloid Chemistry, Krieger, Germany, 318 pp.
Woods, R.D., editor (1987) Geotechnical Practice for Waste Disposal. American Society of Civil Engineers, Geotechnical Special Publication 13, 864 pp.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Likos, W.J., Lu, N. Water vapor sorption behavior of smectite-kaolinite mixtures. Clays Clay Miner. 50, 553–561 (2002). https://doi.org/10.1346/000986002320679297
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1346/000986002320679297