Summary
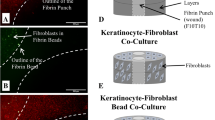
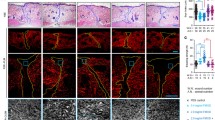
Previously our laboratory, and others, described an in vitro model for the study of fibroblast wound repopulation. The so-called punch-wounded, fibroblast-populated collagen lattice has been used extensively in tissue repair research. We now identify certain shortcomings with this model, which have led to its enhancement by the introduction of a provisional matrix fabricated in situ from fibrinogen and α-thrombin. In the previous model, fibroblasts repopulate the wound defect (WD) as a monolayer of cells and on reaching confluence, a process reminiscent of fibroplasia fills the wound space. The enhanced model, with fibrin acting as a provisional matrix, allowed fibroblasts to repopulate the WD as a three-dimensional network of cells that were morphologically different from cells migrating over the collagen substratum of the previous model. Fibroblast repopulation of the fibrin matrix was typically around double the rate of repopulation of the empty wound space. We propose this model as an enhanced, yet sufficiently reproducible, model for the study of fibroblast responses to tissue damage. It can be further enhanced by the addition of other cell types and matrix components.
Similar content being viewed by others
References
Auger, F. A.; Rouabhia, M.; Goulet, F.; Berthod, F.; Moulin, V.; Germain, L. Tissue-engineered human skin substitutes developed from collagen-populated hydrated gels: clinical and fundamental applications. Med. Biol. Eng. Comput. 36:801–812; 1998.
Baschong W; Sutterlin, R.; Aebi, U. Punch-wounded, fibroblast populated collagen matrices: a novel approach for studying cytoskeletal changes in three dimensions by confocal laser scanning microscopy. Eur. J. Cell Biol. 72:189–201; 1997.
Bell, E.; Ivarsson, B.; Merrill, C. Production of tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc. Natl. Acad. Sci. USA 76:1274–1278; 1979.
Bogatkevich, G. S.; Tourkina, E.; Silver, R. M.; Ludwicka-Bradley, A. Thombin differentiates normal lung fibroblasts to a myofibroblast phenotype via the proteolytically activated receptor-1 and a protein kinase C-dependent pathway. J. Biol. Chem. 276:45184–45192; 2001.
Brown, L. F.; Lanir, N.; McDonagh, J.; Tognazzi, K.; Dvorak, A. M.; Dvorak, H. F. Fibroblast migration in fibrin gel matrices. Am. J. Pathol. 142:273–283, 1993.
Chambers, R. C.; Leoni, P.; Blanc-Brude, O. P.; Wembridge, D. E.; Laurent, G. J. Thrombin is a potent inducer of connective tissue growth factor production via proteolytic activation of protease-activated receptor-1. J. Biol. Chem. 275:35584–35591; 2000.
Clark, R. A. F. Fibrin and wound healing. In: Fibrinogen, Ann. NY Acad. Sci. 936:355–367; 2001.
Dawes, K. E.; Gray, A. J.; Laurent, G. J. Thrombin stimulates fibroblast chemotaxis and replication. Eur. J. Cell Biol. 61:126–130; 1993.
Genever, P. G.; Wood, E. J.; Cunliffe, W. J. The wounded dermal equivalent offers a simplified model for studying wound repair in vitro. Exp. Dermatol. 2:266–273; 1993.
Gorodetsky, R.; Vexler, A.; An J. Q.; Mou, X. D.; Marx, G. Haptotactic and growth stimulatory effects of fibrin(ogen) and thrombin on cultured fibroblasts. J. Lab. Clin. Med. 131:269–280; 1998.
Greiling, D.; Clark, R. A. F. Fibronectin provides a conduit for fibroblast transmigration from collagenous stroma into fibrin clot provisional matrix. J. Cell Sci. 110:861–870; 1997.
Herrick, S.; Blanc-Brude, O.; Gray, A.; Laurent, G. Fibrinogen. Int. J. Biochem. Cell Biol. 31:741–746; 1999.
Rowling, P. J. E.; Raxworthy, M. J.; Wood, E. J.; Kearney, J. N.; Cunliffe, W. J. Fabrication and reorganization of dermal equivalents suitable for skin-grafting after major cutaneous injury. Biomaterials 11:181–185; 1990.
Senior, R. M.; Skogen, W. F.; Griffin, G. L.; Wilner, G. D. Effects of fibrinogen derivatives upon the inflammatory response—studies with human fibrinopeptide-D. J. Clin. Invest. 77:1014–1019; 1986.
Singer, A. J.; Clark, R. A. F. Mechanisms of disease—cutaneous wound healing. N. Engl. J. Med. 341:738–746; 1999.
Stephens, P.; Genever, P. G.; Wood, E. J.; Raxworthy, M. J. Integrin receptor involvement in actin cable formation in an in vitro model of events associated with wound contraction. Int. J. Biochem. Cell Biol. 29:121–128; 1997.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
O'Leary, R., Wood, E.J. A novel in vitro dermal wound-healing model incorporating a response to mechanical wounding and repopulation of a fibrin provisional matrix. In Vitro Cell.Dev.Biol.-Animal 39, 204–207 (2003). https://doi.org/10.1290/1543-706X(2003)039<0204:ANIVDW>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1543-706X(2003)039<0204:ANIVDW>2.0.CO;2