Summary
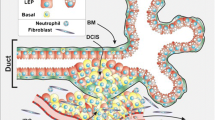
The microenvironment plays a key role in the cellular differentiation of the two main cell lineages of the human breast, luminal epithelial, and myoepithelial. It is not clear, however, how the components of the microenvironment control the development of these cell lineages. To investigate how lineage development is regulated by 3-D culture and microenvironment components, we used the PMC42-LA human breast carcinoma cell line, which possesses stem cell characteristics. When cultured on a two-dimensional glass substrate, PMC42-LA cells formed a monolayer and expressed predominantly luminal epithelial markers, including cytokeratins 8, 18, and 19; E-cadherin; and sialomucin. The key myoepithelial-specific proteins α-smooth muscle actin and cytokeratin 14 were not expressed. When cultured within Engelbreth-Holm-Swarm sarcoma-derived basement membrane matrix (EHS matrix), PMC42-LA cells formed organoids in which the expression of luminal markers was reduced and the expression of other myoepithelial-specific markers (cytokeratin 17 and P-cadherin) was promoted. The presence of primary human mammary gland fibroblasts within the EHS matrix induced expression of the key myoepithelial-specific markers, α-smooth muscle actin and cytokeratin 14. Immortalized human skin fibroblasts were less effective in inducing expression of these key myoepithelial-specific markers. Confocal dual-labeling showed that individual cells expressed luminal or myoepithelial proteins, but not both. Conditioned medium from the mammary fibroblasts was equally effective in inducing myoepithelial marker expression. The results indicate that the myoepithelial lineage is promoted by the extracellular matrix, in conjunction with products secreted by breast-specific fibroblasts. Our results demonstrate a key role for the breast microenvironment in the regulation of breast lineage development.
Similar content being viewed by others
References
Ackland, M. L.; Michalczyk, A.; Whitehead, R. H. PMC42, a novel model for the differentiated human breast. Exp. Cell Res. 263:14–22; 2001.
Ackland, L.; Newgreen, D.; Fridman, M.; Waltham, M.; Arvanitis, A.; Minichiello, J.; Proce, J.; Thompson, E. Epidermal growth factor-induced epithelio-mesenchymal transition in human breast carcinoma cells. Lab. Invest. 83(3):1–14; 2003.
Alvi, A. J.; Clayton, H.; Joshi, C.; Enver, T.; Ashworth, A.; Vivanco, M. D. M.; Dale, T. C.; Smalley, M. Functional and molecular characterisaion of mammary side population cells. Breast Cancer Res. Treat. 5(1):R1-R8; 2002.
Barcellos-Hoff, M. H.; Ravani, S. A. Irradiated mammary gland stroma promotes the expression of tumorigenic potential by unirradiated epithelial cells. Cancer Res. 60(5):1254–1260; 2000.
Barsky, S. H. Myoepithelial mRNA expression profiling reveals a common tumor-suppressor phenotype. Exp. Mol. Pathol. 74(2):113–122; 2003.
Bhowmick, N. A.; Neilson, E. G.; Moses, H. L. Stroinal fibroblasts in cancer initiation and progression. Nature 432(7015):332–337; 2004.
Bissell, M. J.; Radisky, D., Putting tumours in context. Natl. Rev. Cancer 1(1):46–54; 2001.
Bocker, W.; Moll, R.; Poremba, C., et al. Common adult stem cells in the human breast give rise to glandular and myoepithelial cell lineages: a new biological cell concept. Lab. Invest. 82(6):737–745; 2002.
Daniel, C. W.; Strickland, P.; Friedmann Y. Expression and functional role of E- and P-cadherins in mouse mammary ductal morphogenesis and growth. Dev. Biol. 169(2):511–519; 1995.
DEugnier, M. A.; Faraldo, M. M.; Rousselle, P.; Thiery, J. P.; Glukhova, M. A. Cell-extracellular matrix interactions and EGF are important regulators of the basal mammary epithelial cell phenotype. J. Cell Sci. 112(Pt 7):1035–1044; 1999.
Deugnier, M.-A.; Teuliere, J.; Faraldo, M. M.; Thiery, J. P.; Glukhova, M. A. The importance of being a myoepithelial cell. Breast Cancer Res. Treat. 4(6):224–230; 2002.
Dontu, G.; Abdallah, W. M.; Foley, J. M.; Jackson, K. W.; Clarke, M. F.; Kawamura, M. J.; Wicha, M. S. In vitro propagation and transcriptional profiling of human mammary stem/progenitor cells. Genes Dev. 17(10):1253–1270; 2003.
Easty, G. C.; Easty, D. M.; Monaghan, P.; Ormerod, M. G.; Neville, A. M. Preparation and identification of human breast epithelial cells in culture. Int. J. Cancer 26(5):577–584; 1980.
Guelstein, V. I.; Tchypysheva, T. A.; Ermilova, V. D.; Litvinova, L. V.; Troyanovsky, S. M.; Bannikov, G. A. Monoclonal antibody mapping of ketatins 8 and 17 and of vimentin in normal human mammary gland, benign tumors, dysplasias and breast cancer. Int. J. Cancer 42(2):147–153; 1988.
Hay, E. D. An overview of epithelio-mesenchymal transformation. Acta Anat. 1(154):8–20; 1995.
Hazan, R. B.; Qiao, R.; Keren, R.; Badano, I.; Suyama, K. Cadherin switch in tumor progression. Ann. N.Y. Acad. Sci. 1014:155–163; 2004.
Joshi, K.; Smith, J. A.; Perusinghe, N.; Monoghan, P. Cell proliferation in the human mammary epithelium. Differential contribution of epithelial and myoepithelial cells. Am. J. Pathol. 124:199–206;1986.
Lakhani, S. R.; O'Hare, M. J. The mammary myoepithelial cell—Cinderella or ugly sister. Breast Cancer Res Treat. 3:1–4; 2001.
Lazard, D.; Sastre, X.; Frid, M. G.; Glukhova, M. A.; Thiery, J. P.; Koteliansky, V. E. Expression of smooth muscle-specific proteins in myoepithelium and stromal fibroblasts of normal and malignant human breast fissue. J. Cell Biol. 90:999–1003; 1993.
Nieman, M. T.; Prudoff, R. S.; Johnson, K. R.; Wheelock, M. J. N-cadherin promotes motility in human breast cancer cells regardless of their E-cadherin expression. J. Cell Biol. 147(3):631–644; 1999.
Ozzello, L. Ultrastructure of the human mammary gland. Pathol. Annu. 6:1–59; 1971.
Pachoux, C.; Gudjonsson T.; Ronnov-Jessen, L.; Bissell, M. J.; Petersen, O. W. Human mammary luminal epithelial cells contain progenitors to myoepithelial cells. Dev. Biol. 206:88–99; 1999.
Pollard, J. W. Transforming growth factor-β isoforms and hepatocyte growth factor/scatter factor in mammary gland ductal morphogenesis. Breast Cancer Res. Treat, 3(4):230–237; 2001.
Robinson, G. W.; Karpf, C. A. B.; Kratochwil, K. Regulation of mammary gland development by tissue interaction. J. Mammary Gland Biol. Neoplasia 4(1):9–19; 1999.
Ronnov-Jessen, L. Stromal reaction to invasive cancer: the cellular origin of the myofibroblast and implications for tumor development. Breast J. 2:320–339; 1996.
Rudland, P. S.; Hughes, C. M. Immunocytochemical identification of cell types in human mammary gland: variations in cellular markers are dependant on glandular topography and differentiation. Histochem. Cytochem. 7(37):1087–1100; 1989.
Shekhar, M. P. V.; Pauley, R.; Heppner, G. Extracellular matrix-stromal cell contribution to neoplastic phenotype of epithelial cells in the breast. Breast Cancer Res. Treat. 5(3):130–135; 2003.
Smith, G. H. Experimental mammary epithelial morphogenesis in an in vivo model: evidence for distinct cellular progenitors of the ductal and lobular phenotype. Breast Cancer Res. Treat. 39(1):21–31; 1996.
Steinberg, M. S.; McNutt, P. M. Cadherins and their connections: adhesion junctions have broader functions. Curr. Opin. Cell Biol. 11(5):554–560; 1999.
Sternlicht, M. D.; Kedeshian, P.; Shao, Z. M.; Safarians, S.; Barsky, S. H. The human myoepithelial cell is a natural tumor suppressor. Clin. Cancer Res. 3(11):1949–1958; 1997.
Stingl, J.; Eaves, C. J.; Kuusk, U.; Emerman, J. T. Phenotypic and functional characterization in vitro of a multipotent epithelial cell present in the normal adult human breast. Differentiation 63:201–213; 1998.
Stingl, J.; Eaves, C. J.; Zandieh, I.; Emerman, J. T. Characterization of bipotent mammary epithelial progenitor cells in normal adult human breast tissue. Breast Cancer Res. Treat. 67:93–109; 2001.
Sympson, C. J.; Talhouk, R. S.; Alexander, C. M.; Chin, J. R.; Clift, S. M.; Bissell, M. J.; Werb, Z. Targeted expression of stromelysin-1 in mammary gland provides evidence for a role of proteinases in branching morphogenesis and the requirement for an intact basement membrane for tissue-specific gene expression. J. Cell Biol, 125(3):681–693; 1994.
Taylor-Papadimitriou, J.; Peterson, J. A.; Arklie, J.; Burchell, J.; Ceriani, R. L.; Bodmer, W. F. Monoclonal antibodies to epithelium-specific components of the human milk fat globule membrane: production and reaction with cells in culture. Int. J. Cancer 28(1):17–21; 1981.
Taylor-Papadimitriou, J.; Stampfer, M.; Bartek, J.; Lewis, A.; Boshell, M.; Lane, E. B.; Leigh, I. M. Keratin expression in human mammary epithelial cells cultured from normal and malignant tissue: relation to in vivo phenotypes and influence of medium. J. Cell Sci. 94:403–413; 1989.
Thiery, J. P. Epithelial-mesenchymal transitions in tumor progression. Nat. Rev. Cancer 2:442–454; 2002.
Troyanovsky, S. M.; Guelstein, V. I.; Tchipysheva, T. A.; Krutovskikh, V. A.; Bannikov, G. A. Patterns of expression of keratin 17 in human epithelia: dependency on cell position. J. Cell Sci. 93(Pt 3):419–426; 1989.
Welm, B. E.; Tepera, S. B.; Venezia, T.; Graubert, T. A.; Rosen, J. M.; Goodell, M. A. Sca-1(pos) cells in the mouse mammary gland represent an enriched progenitor cell population. Dev. Biol. 245(1):42–56; 2002.
Whitehead, R. H.; Bertoncello, I.; Webber, L. M.; Pedersen, J. S. A new human breast carcinoma cell line (PMC42) with stem cell characteristics. I. Morphologic characterization. J. Natl. Cancer Inst. 70(4):649–654; 1983a.
Whitehead, R. H.; Monaghan, P.; Webber, L. M.; Bertoncello, I.; Vitali, A. A. A new human breast carcinoma cell line (PMC42) with stem cell characteristics. II. Characterization of cells growing as organoids. J. Natl. Cancer Inst. 71(6):1193–1196; 1983b.
Whitehead, R. H.; Quirck, S. J.; Vitali, A. A.; Funder, J. W.; Sutherland, R. L.; Murphy, L. C. A new human breast carcinoma cell line (PMC42) with stem cell characteristics. III. Hormone receptor status and responsiveness. J. Natl. Cancer Inst. 73(3):643–647; 1984.
Witty, J. P.; Wright, J. H. Matrix metalloproteinases are expressed during ductal and alveolar mammary morphogenesis and misregulation of stromelysin-1 in transgenic mice induces unscheduled alveolar development. Mol. Biol. Cell 6(10):1287–1303; 1995.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lebret, S.C., Newgreen, D.F., Waltham, M.C. et al. Myoepithelial molecular markers in human breast carcinoma PMC42-LA cells are induced by extracellular matrix and stromal cells. In Vitro Cell.Dev.Biol.-Animal 42, 298–307 (2006). https://doi.org/10.1290/0601004.1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/0601004.1