Abstract
Background
The assessment of muscle mass loss, muscle strength, and physical function has been recommended in diagnosing sarcopenia. However, only muscle mass has been assessed in previous studies. Therefore, this study investigated the effect of comprehensively diagnosed preoperative sarcopenia on the prognosis of patients with esophageal cancer.
Methods
The study analyzed 115 patients with esophageal cancer (age ≥ 65 years) who underwent curative esophagectomy. Preoperative sarcopenia was analyzed using the skeletal mass index (SMI), handgrip strength, and gait speed based on the Asian Working Group for Sarcopenia 2019 criteria. Clinicopathologic factors, incidence of postoperative complications, and overall survival (OS) were compared between the sarcopenia and non-sarcopenia groups. The significance of the three individual parameters also was evaluated.
Results
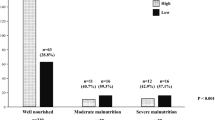
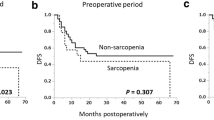
The evaluation identified 47 (40.9%) patients with low SMI, 31 (27.0%) patients with low handgrip strength, and 6 (5.2%) patients with slow gait speed. Sarcopenia was diagnosed in 23 patients (20%) and associated with older age and advanced pT stage. The incidence of postoperative complications did not differ significantly between the two groups. Among the three parameters, only slow gait speed was associated with Clavien-Dindo grade 2 or greater complications. The sarcopenia group showed significantly worse OS than the non-sarcopenia group. Those with low handgrip strength tended to have worse OS, and those with slow gait speed had significantly worse OS than their counterparts.
Conclusions
Preoperative sarcopenia diagnosed using skeletal muscle mass, muscle strength, and physical function may have an impact on the survival of patients with esophageal cancer.



Similar content being viewed by others
References
Chen LK, Liu LK, Woo J, et al. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc. 2014;15:95–101.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on sarcopenia in older people. Age Ageing. 2010;39:412–23.
Chen LK, Woo J, Assantachai P, et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020;21(300–7):e302.
Berardi G, Antonelli G, Colasanti M, et al. Association of sarcopenia and body composition with short-term outcomes after liver resection for malignant tumors. JAMA Surg. 2020;155:e203336.
Deluche E, Lachatre D, Di Palma M, et al. Is sarcopenia a missed factor in the management of patients with metastatic breast cancer? Breast. 2022;61:84–90.
Kawaguchi Y, Hanaoka J, Ohshio Y, et al. Does sarcopenia affect postoperative short- and long-term outcomes in patients with lung cancer? A systematic review and meta-analysis. J Thorac Dis. 2021;13:1358–69.
Takiguchi K, Furuya S, Sudo M, et al. Prognostic effect of sarcopenia in colorectal cancer recurrence. Nutrition. 2021;91–2:111362.
Yamamoto K, Hirao M, Nishikawa K, et al. Sarcopenia is associated with impaired overall survival after gastrectomy for elderly gastric cancer. Anticancer Res. 2019;39:4297–303.
Deng HY, Zha P, Peng L, et al. Preoperative sarcopenia is a predictor of poor prognosis of esophageal cancer after esophagectomy: a comprehensive systematic review and meta-analysis. Dis Esophagus. 2019;32:doy115.
Harada K, Ida S, Baba Y, et al. Prognostic and clinical impact of sarcopenia in esophageal squamous cell carcinoma. Dis Esophagus. 2016;29:627–33.
Nakashima Y, Saeki H, Nakanishi R, et al. Assessment of sarcopenia as a predictor of poor outcomes after esophagectomy in elderly patients with esophageal cancer. Ann Surg. 2018;267:1100–4.
Paireder M, Asari R, Kristo I, et al. Impact of sarcopenia on outcome in patients with esophageal resection following neoadjuvant chemotherapy for esophageal cancer. Eur J Surg Oncol. 2017;43:478–84.
Rice TW, Patil DT, Blackstone EH. 8th-Edition AJCC/UICC staging of cancers of the esophagus and esophagogastric junction: application to clinical practice. Ann Cardiothorac Surg. 2017;6:119–30.
Sugimura K, Miyata H, Tanaka K, et al. Multicenter randomized phase 2 trial comparing chemoradiotherapy and docetaxel plus 5-fluorouracil and cisplatin chemotherapy as initial induction therapy for subsequent conversion surgery in patients with clinical T4b esophageal cancer: short-term results. Ann Surg. 2021;274:e465–72.
Lauretani F, Russo CR, Bandinelli S, et al. Age-associated changes in skeletal muscles and their effect on mobility: an operational diagnosis of sarcopenia. J Appl Physiol (1985). 2003;95:1851–60.
Cesari M, Kritchevsky SB, Newman AB, et al. Added value of physical performance measures in predicting adverse health-related events: results from the Health, Aging And Body Composition Study. J Am Geriatr Soc. 2009;57:251–9.
Hagens ERC, van Berge Henegouwen MI, van Sandick JW, et al. Distribution of lymph node metastases in esophageal carcinoma [TIGER study]: study protocol of a multinational observational study. BMC Cancer. 2019;19:662.
Tamandl D, Paireder M, Asari R, et al. Markers of sarcopenia quantified by computed tomography predict adverse long-term outcome in patients with resected oesophageal or gastro-oesophageal junction cancer. Eur Radiol. 2016;26:1359–67.
Wang PY, Chen XK, Liu Q, et al. Highlighting sarcopenia management for promoting surgical outcomes in esophageal cancers: evidence from a prospective cohort study. Int J Surg. 2020;83:206–15.
Grotenhuis BA, Shapiro J, van Adrichem S, et al. Sarcopenia/muscle mass is not a prognostic factor for short- and long-term outcome after esophagectomy for cancer. World J Surg. 2016;40:2698–704.
Yip C, Goh V, Davies A, et al. Assessment of sarcopenia and changes in body composition after neoadjuvant chemotherapy and associations with clinical outcomes in oesophageal cancer. Eur Radiol. 2014;24:998–1005.
Makiura D, Ono R, Inoue J, et al. Impact of sarcopenia on unplanned readmission and survival after esophagectomy in patients with esophageal cancer. Ann Surg Oncol. 2018;25:456–64.
Song M, Zhang Q, Tang M, et al. Associations of low hand grip strength with 1 year mortality of cancer cachexia: a multicentre observational study. J Cachexia Sarcopenia Muscle. 2021;12:1489–500.
Zhuang CL, Zhang FM, Li W, et al. Associations of low handgrip strength with cancer mortality: a multicentre observational study. J Cachexia Sarcopenia Muscle. 2020;11:1476–86.
Kurita D, Oguma J, Ishiyama K, et al. Handgrip strength predicts postoperative pneumonia after thoracoscopic-laparoscopic esophagectomy for patients with esophageal cancer. Ann Surg Oncol. 2020;27:3173–81.
Sato S, Nagai E, Taki Y, et al. Hand grip strength as a predictor of postoperative complications in esophageal cancer patients undergoing esophagectomy. Esophagus. 2018;15:10–8.
Jiang X, Xu X, Ding L, et al. Predictive value of preoperative handgrip strength on postoperative outcomes in patients with gastrointestinal tumors: a systematic review and meta-analysis. Support Care Cancer. 2022;30:6451–62.
Dociak-Salazar E, Barrueto-Deza JL, Urrunaga-Pastor D, et al. Gait speed as a predictor of mortality in older men with cancer: a longitudinal study in Peru. Heliyon. 2022;8:e08862.
Dong QT, Cai HY, Zhang Z, et al. Influence of body composition, muscle strength, and physical performance on the postoperative complications and survival after radical gastrectomy for gastric cancer: a comprehensive analysis from a large-scale prospective study. Clin Nutr. 2021;40:3360–9.
Hantel A, DuMontier C, Odejide OO, et al. Gait speed, survival, and recommended treatment intensity in older adults with blood cancer requiring treatment. Cancer. 2021;127:875–83.
Liu MA, DuMontier C, Murillo A, et al. Gait speed, grip strength, and clinical outcomes in older patients with hematologic malignancies. Blood. 2019;134:374–82.
Zhang FM, Zhang XZ, Shi HP, et al. Comparisons and impacts of the basic components of sarcopenia definition and their pairwise combinations in gastric cancer: a large-scale study in a Chinese population. Front Nutr. 2021;8:709211.
Kato K, Ito Y, Daiko H, et al. A randomized controlled phase III trial comparing two chemotherapy regimen and chemoradiotherapy regimen as neoadjuvant treatment for locally advanced esophageal cancer, JCOG1109 NexT study. J Clin Oncol. 2022;40:238.
Kitagawa Y, Ishihara R, Ishikawa H, et al. Esophageal cancer practice guidelines 2022 edited by the Japan esophageal society: part 1. Esophagus. 2023;20:343–72.
Nakamura K, Kato K, Igaki H, et al. Three-arm phase III trial comparing cisplatin plus 5-FU (CF) versus docetaxel, cisplatin plus 5-FU (DCF) versus radiotherapy with CF (CF-RT) as preoperative therapy for locally advanced esophageal cancer (JCOG1109, NexT study). Jpn J Clin Oncol. 2013;43:752–5.
Kita R, Miyata H, Sugimura K, et al. Clinical effect of enteral nutrition support during neoadjuvant chemotherapy on the preservation of skeletal muscle mass in patients with esophageal cancer. Clin Nutr. 2021;40:4380–5.
Reisinger KW, Bosmans JW, Uittenbogaart M, et al. Loss of skeletal muscle mass during neoadjuvant chemoradiotherapy predicts postoperative mortality in esophageal cancer surgery. Ann Surg Oncol. 2015;22:4445–52.
Motoori M, Fujitani K, Sugimura K, et al. Skeletal muscle loss during neoadjuvant chemotherapy is an independent risk factor for postoperative infectious complications in patients with advanced esophageal cancer. Oncology. 2018;95:281–7.
Halliday LJ, Boshier PR, Doganay E, et al. The effects of prehabilitation on body composition in patients undergoing multimodal therapy for esophageal cancer. Dis Esophagus. 2023;36:doac046.
Minnella EM, Awasthi R, Loiselle SE, et al. Effect of exercise and nutrition prehabilitation on functional capacity in esophagogastric cancer surgery: a randomized clinical trial. JAMA Surg. 2018;153:1081–9.
Nambara M, Miki Y, Tamura T, et al. The optimal definition of sarcopenia for predicting postoperative pneumonia after esophagectomy in patients with esophageal cancer. World J Surg. 2021;45:3108–18.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Fig. S1
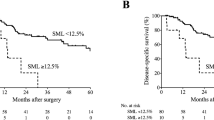
Comparison of disease-specific survival (DSS) between the sarcopenia and non-sarcopenia groups (TIF 3069 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kanemura, T., Takeoka, T., Sugase, T. et al. Significance of Comprehensive Analysis of Preoperative Sarcopenia Based on Muscle Mass, Muscle Strength, and Physical Function for the Prognosis of Patients with Esophageal Cancer. Ann Surg Oncol 31, 818–826 (2024). https://doi.org/10.1245/s10434-023-14306-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-14306-5