Abstract
Background
This study aimed to compare the prognostic discrimination power of pretreatment pathologic N stage (prepN), lymph node tumor regression grade (LNTRG), and posttreatment pathologic N (ypN) category for esophageal squamous cell carcinoma (ESCC) patients who received neoadjuvant chemoradiotherapy (nCRT) plus surgery.
Methods
The study reviewed 187 ESCC patients from two medical centers who underwent nCRT plus surgery. Pathologic LNTRG was defined by the proportion of viable tumor area within the tumor bed in lymph nodes (LNs). An average LNTRG then was calculated by averaging the tumor regression grade (TRG) score of all resected LNs. Lymph nodes containing regression changes or vital tumor cells were used for interpretation of the prepN stage, which reflects the estimated number of originally involved LNs.
Results
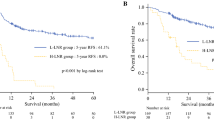
The ypN, prepN, and LNTRG categories had significant prognostic stratification power (p < 0.001, log-rank test). Multivariable cox regression showed that all three categories were independent prognostic factors of disease-free survival (DFS) (p < 0.05). The LNTRG category showed a better prognostic value for DFS prediction than the ypN and prepN categories (Akaike information criterion [AIC]: LNTRG [933.69], ypN [937.56], prepN [937.45]). Additionally, the superior predictive capacity of the LNTRG category was demonstrated by decision curve analysis. Similar results were discovered for patients with remaining diseased LNs.
Conclusions
The three staging categories had prognostic relevance for DFS, with the LNTRG category seeming to have better prognostic indication power. Comprehensive consideration of the ypN status, prepN status, and LN regression may allow for better prognostic stratification of patients.



Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Fiorica F, Di Bona D, Schepis F, et al. Preoperative chemoradiotherapy for oesophageal cancer: a systematic review and meta-analysis. Gut. 2004;53:925–30.
Tepper J, Krasna MJ, Niedzwiecki D, et al. Phase III trial of trimodality therapy with cisplatin, fluorouracil, radiotherapy, and surgery compared with surgery alone for esophageal cancer: CALGB 9781. J Clin Oncol. 2008;26:1086–92.
van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–84.
Allum WH, Stenning SP, Bancewicz J, Clark PI, Langley RE. Long-term results of a randomized trial of surgery with or without preoperative chemotherapy in esophageal cancer. J Clin Oncol. 2009;27:5062–7.
Rice TW, Patil DT, Blackstone EH. 8th-edition AJCC/UICC staging of cancers of the esophagus and esophagogastric junction: application to clinical practice. Ann Cardiothorac Surg. 2017;6:119–30.
Rice TW, Ishwaran H, Kelsen DP, et al. Recommendations for neoadjuvant pathologic staging (ypTNM) of cancer of the esophagus and esophagogastric junction for the 8th-edition AJCC/UICC staging manuals. Dis Esophagus. 2016;29:906–12.
Hagi T, Makino T, Yamasaki M, et al. Pathological regression of lymph nodes better predicts long-term survival in esophageal cancer patients undergoing neoadjuvant chemotherapy followed by surgery. Ann Surg. 2022;275:1121–9.
Davies AR, Myoteri D, Zylstra J, et al. Lymph node regression and survival following neoadjuvant chemotherapy in oesophageal adenocarcinoma. Br J Surg. 2018;105:1639–49.
Shapiro J, Biermann K, van Klaveren D, et al. Prognostic value of pretreatment pathological tumor extent in patients treated with neoadjuvant chemoradiotherapy plus surgery for esophageal or junctional cancer. Ann Surg. 2017;265:356–62.
Dworak O, Keilholz L, Hoffmann A. Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis. 1997;12:19–23.
Wheeler JM, Warren BF, Jones AC, Mortensen NJ. Preoperative radiotherapy for rectal cancer: implications for surgeons, pathologists and radiologists. Br J Surg. 1999;86:1108–20.
Wheeler JM, Warren BF, Mortensen NJ, et al. Quantification of histologic regression of rectal cancer after irradiation: a proposal for a modified staging system. Dis Colon Rectum. 2002;45:1051–6.
Cohen J. Weighted kappa: nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull. 1968;70:213–20.
Cyr L, Francis K. Measures of clinical agreement for nominal and categorical data: the kappa coefficient. Comput Biol Med. 1992;22:239–46.
Sato F, Shimada Y, Li Z, et al. Lymph node micrometastasis and prognosis in patients with oesophageal squamous cell carcinoma. Br J Surg. 2001;88:426–32.
Altorki N, Kent M, Ferrara C, Port J. Three-field lymph node dissection for squamous cell and adenocarcinoma of the esophagus. Ann Surg. 2002;236:177–83.
Leng X, He W, Yang H, et al. Prognostic Impact of Postoperative lymph node metastases after neoadjuvant chemoradiotherapy for locally advanced squamous cell carcinoma of esophagus: from the results of NEOCRTEC5010, a randomized multicenter study. Ann Surg. 2021;274:e1022–9.
Nomura M, Shitara K, Kodaira T, et al. Prognostic impact of the 6th and 7th American Joint Committee on Cancer TNM staging systems on esophageal cancer patients treated with chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2012;82:946–52.
Brinkmann S, Noordman BJ, Hölscher AH, et al. External validation of pretreatment pathological tumor extent in patients with neoadjuvant chemoradiotherapy plus surgery for esophageal cancer. Ann Surg Oncol. 2020;27:1250–8.
Oppedijk V, van der Gaast A, van Lanschot JJ, et al. Patterns of recurrence after surgery alone versus preoperative chemoradiotherapy and surgery in the CROSS trials. J Clin Oncol. 2014;32:385–91.
Shapiro J, van Lanschot J, Hulshof M, et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol. 2015;16:1090–8.
Philippron A, Bollschweiler E, Kunikata A, et al. Prognostic relevance of lymph node regression after neoadjuvant chemoradiation for esophageal cancer. Semin Thorac Cardiovasc Surg. 2016;28:549–58.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 81902396) and Zhongshan Hospital (Grant Nos. 2020ZSLC5 and 2021ZSYQ27).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosure
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Jiang, D., Jian, Z. et al. Identification of Optimal Parameters for Assessing Lymph Node Status of Patients with Esophageal Squamous Cell Carcinoma After Neoadjuvant Chemoradiotherapy. Ann Surg Oncol 31, 883–891 (2024). https://doi.org/10.1245/s10434-023-14135-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-14135-6