Abstract
Background
Consideration of sentinel lymph node biopsy (SLNB) is recommended for patients with T1b melanomas and T1a melanomas with high-risk features; however, the proportion of patients with actionable results is low. We aimed to identify factors predicting SLNB positivity in T1 melanomas by examining a multi-institutional international population.
Methods
Data were extracted on patients with T1 cutaneous melanoma who underwent SLNB between 2005 and 2018 at five tertiary centers in Europe and Canada. Univariable and multivariable logistic regression analyses were performed to identify predictors of SLNB positivity.
Results
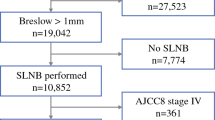
Overall, 676 patients were analyzed. Most patients had one or more high-risk features: Breslow thickness 0.8–1 mm in 78.1% of patients, ulceration in 8.3%, mitotic rate > 1/mm2 in 42.5%, Clark’s level ≥ 4 in 34.3%, lymphovascular invasion in 1.4%, nodular histology in 2.9%, and absence of tumor-infiltrating lymphocytes in 14.4%. Fifty-three patients (7.8%) had a positive SLNB. Breslow thickness and mitotic rate independently predicted SLNB positivity. The odds of positive SLNB increased by 50% for each 0.1 mm increase in thickness past 0.7 mm (95% confidence interval [CI] 1.05–2.13) and by 22% for each mitosis per mm2 (95% CI 1.06–1.41). Patients who had one excised node (vs. two or more) were three times less likely to have a positive SLNB (3.6% vs. 9.6%; odds ratio 2.9 [1.3–7.7]).
Conclusions
Our international multi-institutional data confirm that Breslow thickness and mitotic rate independently predict SLNB positivity in patients with T1 melanoma. Even within this highly selected population, the number needed to diagnose is 13:1 (7.8%), indicating that more work is required to identify additional predictors of sentinel node positivity.
Similar content being viewed by others
References
Global Cancer Observatory. Melanoma of skin. World Health Organization; 2020. Available at: https://gco.iarc.fr/today/data/factsheets/cancers/16-Melanoma-of-skin-fact-sheet.pdf.
Shaikh WR, Dusza SW, Weinstock MA, Oliveria SA, Geller AC, Halpern AC. Melanoma thickness and survival trends in the United States, 1989–2009. J Natl Cancer Inst. 2016;108(1):djv294. https://doi.org/10.1093/jnci/djv294.
Cordeiro E, Gervais MK, Shah PS, Look Hong NJ, Wright FC. Sentinel lymph node biopsy in thin cutaneous melanoma: a systematic review and meta-analysis. Ann Surg Oncol. 2016;23(13):4178–88. https://doi.org/10.1245/s10434-016-5137-z.
Balch CM, Soong SJ, Gershenwald JE, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer Melanoma Staging System. J Clin Oncol. 2001;19(16):3622–34. https://doi.org/10.1200/JCO.2001.19.16.3622.
Statius Muller MG, van Leeuwen PA, de Lange-De Klerk ES, et al. The sentinel lymph node status is an important factor for predicting clinical outcome in patients with Stage I or II cutaneous melanoma. Cancer. 2001;91(12):2401–8.
Cascinelli N, Bombardieri E, Bufalino R, et al. Sentinel and nonsentinel node status in stage IB and II melanoma patients: two-step prognostic indicators of survival. J Clin Oncol. 2006;24(27):4464–71. https://doi.org/10.1200/JCO.2006.06.3198.
Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med. 2014;370(7):599–609. https://doi.org/10.1056/NEJMoa1310460.
Melanoma: Cutaneous. National comprehensive cancer network; 2021. Available at: https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf. Accessed 2 May 2021.
Eggermont AMM, Blank CU, Mandala M, et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma. N Engl J Med. 2018;378(19):1789–801. https://doi.org/10.1056/NEJMoa1802357.
Weber J, Mandala M, Del Vecchio M, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med. 2017;377(19):1824–35. https://doi.org/10.1056/NEJMoa1709030.
Morton DL, Cochran AJ, Thompson JF, et al. Sentinel node biopsy for early-stage melanoma: accuracy and morbidity in MSLT-I, an international multicenter trial. Ann Surg. 2005;242(3):302–11. https://doi.org/10.1097/01.sla.0000181092.50141.fa (discussion 311–313).
Faries MB, Thompson JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 2017;376(23):2211–22. https://doi.org/10.1056/NEJMoa1613210.
Wong SL, Faries MB, Kennedy EB, et al. Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American Society of clinical oncology and society of surgical oncology clinical practice guideline update. J Clin Oncol. 2018;36(4):399–413. https://doi.org/10.1200/JCO.2017.75.7724.
Wright FC, Souter LH, Kellett S, et al. Primary excision margins, sentinel lymph node biopsy, and completion lymph node dissection in cutaneous melanoma: a clinical practice guideline. Curr Oncol. 2019;26(4):e541–50. https://doi.org/10.3747/co.26.4885.
Amin MB, Edge S, Greene F, et al editors. AJCC cancer staging manual. 8th edn. Berlin: Springer; 2017.
Egger ME, Stevenson M, Bhutiani N, et al. Should sentinel lymph node biopsy be performed for all T1b melanomas in the new 8th edition American Joint Committee on cancer staging system? J Am Coll Surg. 2019;228(4):466–72. https://doi.org/10.1016/j.jamcollsurg.2018.12.030.
Sinnamon AJ, Neuwirth MG, Yalamanchi P, et al. Association between patient age and lymph node positivity in thin melanoma. JAMA Dermatol. 2017;153(9):866–73. https://doi.org/10.1001/jamadermatol.2017.2497.
Friedman C, Lyon M, Torphy RJ, et al. A nomogram to predict node positivity in patients with thin melanomas helps inform shared patient decision making. J Surg Oncol. 2019;120(7):1276–83. https://doi.org/10.1002/jso.25720.
Cavanaugh-Hussey MW, Mu EW, Kang S, Balch CM, Wang T. Older age is associated with a higher incidence of melanoma death but a lower incidence of sentinel lymph node metastasis in the SEER databases (2003–2011). Ann Surg Oncol. 2015;22(7):2120–6. https://doi.org/10.1245/s10434-015-4538-8.
Andtbacka RHI, Gershenwald JE. Role of sentinel lymph node biopsy in patients with thin melanoma. J Natl Compr Cancer Netw. 2009;7(3):308–17. https://doi.org/10.6004/jnccn.2009.0023.
Maurichi A, Miceli R, Eriksson H, et al. Factors affecting sentinel node metastasis in thin (T1) cutaneous melanomas: development and external validation of a predictive nomogram. J Clin Oncol. 2020;38(14):1591–601. https://doi.org/10.1200/JCO.19.01902.
Cadili A, Dabbs K. Predictors of sentinel lymph node metastasis in melanoma. Can J Surg. 2010;53(1):32–6.
Ribero S, Gualano MR, Osella-Abate S, et al. Association of histologic regression in primary melanoma with sentinel lymph node status: a systematic review and meta-analysis. JAMA Dermatol. 2015;151(12):1301–7. https://doi.org/10.1001/jamadermatol.2015.2235.
Wong SL, Kattan MW, McMasters KM, Coit DG. A nomogram that predicts the presence of sentinel node metastasis in melanoma with better discrimination than the American Joint Committee on cancer staging system. Ann Surg Oncol. 2005;12(4):282–8. https://doi.org/10.1245/ASO.2005.05.016.
Lo SN, Ma J, Scolyer RA, et al. Improved risk prediction calculator for sentinel node positivity in patients with melanoma: The Melanoma Institute Australia Nomogram. J Clin Oncol. 2020;38(24):2719–27. https://doi.org/10.1200/JCO.19.02362.
Bogach J, Wright FC, Austin J, et al. Medical immunosuppression and outcomes in cutaneous melanoma: a population-based cohort study. Ann Surg Oncol. 2021;28(6):3302–11. https://doi.org/10.1245/s10434-020-09224-9.
Austin J, Wright FC, Cheng SY, Sutradhar R, Baxter NN, Look Hong NJ. Outcomes of immunosuppressed patients who develop melanoma: a population-based propensity-matched cohort study. Ann Surg Oncol. 2020;27(8):2927–48. https://doi.org/10.1245/s10434-020-08265-4.
National Cancer Institute Surveillance, Epidemiology, and End Results Program. Cancer stat facts: melanoma of the skin. National Cancer Institute. Available at: https://seer.cancer.gov/statfacts/html/melan.html. Accessed 20 Jul 2021.
Landow SM, Gjelsvik A, Weinstock MA. Mortality burden and prognosis of thin melanomas overall and by subcategory of thickness, SEER registry data, 1992–2013. J Am Acad Dermatol. 2017;76(2):258–63. https://doi.org/10.1016/j.jaad.2016.10.018.
Isaksson K, Mikiver R, Eriksson H, et al. Survival in 31 670 patients with thin melanomas: a Swedish population-based study. Br J Dermatol. 2021;184(1):60–7. https://doi.org/10.1111/bjd.19015.
Gershenwald JE, Scolyer RA, Hess KR, et al. Melanoma staging: evidence-based changes in the American Joint Committee on cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017;67(6):472–92. https://doi.org/10.3322/caac.21409.
Garbe C, Eigentler TK, Bauer J, et al. Mitotic rate in primary melanoma: interobserver and intraobserver reliability, analyzed using H&E sections and immunohistochemistry. J Dtsch Dermatol Ges. 2016;14(9):910–5. https://doi.org/10.1111/ddg.12797.
Schimming TT, Grabellus F, Roner M, et al. pHH3 immunostaining improves interobserver agreement of mitotic index in thin melanomas. Am J Dermatopathol. 2012;34(3):266–9. https://doi.org/10.1097/DAD.0b013e31823135a3.
Smoller BR, Gershenwald JE, Scolyer RA, et al. Protocol for the examination of specimens from patients with melanoma of the skin. Northfield: College of American Pathologists; 2017.
Hale CS, Qian M, Ma MW, et al. Mitotic rate in melanoma: prognostic value of immunostaining and computer-assisted image analysis. Am J Surg Pathol. 2013;37(6):882–9. https://doi.org/10.1097/PAS.0b013e31827e50fa.
Wang M, Aung PP, Prieto VG. Standardized method for defining a 1-mm2 region of interest for calculation of mitotic rate on melanoma whole slide images. Arch Pathol Lab Med. 2021;145(10):1255–63. https://doi.org/10.5858/arpa.2020-0137-OA.
Balkenhol MCA, Tellez D, Vreuls W, et al. Deep learning assisted mitotic counting for breast cancer. Lab Investig. 2019;99(11):1596–606. https://doi.org/10.1038/s41374-019-0275-0.
Gerami P, Cook RW, Wilkinson J, et al. Development of a prognostic genetic signature to predict the metastatic risk associated with cutaneous melanoma. Clin Cancer Res. 2015;21(1):175–83. https://doi.org/10.1158/1078-0432.CCR-13-3316.
Vetto JT, Hsueh EC, Gastman BR, et al. Guidance of sentinel lymph node biopsy decisions in patients with T1–T2 melanoma using gene expression profiling. Future Oncol. 2019;15(11):1207–17. https://doi.org/10.2217/fon-2018-0912.
Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133. https://doi.org/10.1089/thy.2015.0020.
Breast Cancer. National comprehensive cancer network; 2021. Available at: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed 30 Jul 2021.
Funding
No sources of funding were received to assist in the preparation of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Carolyn Nessim has received honoraria for participation in advisory boards from Merck, Novartis, EMD Sorono, and Sanofi. Frances C. Wright has received honoraria from Novartis. Richard J.B. Walker, Nicole J. Look Hong, Marc Moncrieff, Alexander C.J. van Akkooi, Evan Jost, Winan J. van Houdt, Emma H.A. Stahlie, Chanhee Seo, May Lynn Quan, J. Gregory McKinnon, and Michail N. Mavros have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Walker, R.J.B., Look Hong, N.J., Moncrieff, M. et al. Predictors of Sentinel Lymph Node Metastasis in Patients with Thin Melanoma: An International Multi-institutional Collaboration. Ann Surg Oncol 29, 7010–7017 (2022). https://doi.org/10.1245/s10434-022-11936-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11936-z