Abstract
Background
The American Joint Committee on Cancer tumor-node-metastasis staging system for rectal cancer defines lateral pelvic lymph nodes (LPLNs) only in the internal iliac region as regional. However, the Japanese Society for Cancer of the Colon and Rectum (JSCCR) staging system, also considers obturator lymph nodes (LNs) as regional. This retrospective cohort study evaluated the oncologic status of obturator LNs in low rectal cancer.
Methods
The study identified 3487 patients with pT3-T4 low rectal cancer who had undergone curative resections without preoperative radiotherapy or chemotherapy between 2003 and 2011 in the JSCCR database and divided them into six groups. Overall survival (OS) and recurrence-free survival (RFS) were analyzed by groups.
Results
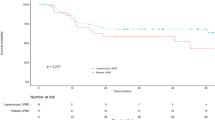
Histologic LPLN metastases were identified in 8% (279/3487) of all the patients and in 18.2% (279/1530) of the patients who underwent lateral pelvic node dissection. The 5-year OS and RFS rates of the obturator-LPLN group (P = 0.095) were worse than those of the internal-LPLN group (P = 0.075), but the difference was not significant. The OS of the obturator-LPLN group was similar to that of the resectable liver metastasis group (P = 0.731), and the RFS of the obturator-LPLN group was significantly better than that of the other-LPLN group (P = 0.016).
Conclusion
The prognosis for obturator LN metastases in low rectal cancer was not significantly worse than for internal iliac LN metastases, defined as regional by the current American Joint Committee on Cancer staging system, and the oncologic status of obturator LNs warrants more studies.




Similar content being viewed by others
References
Akiyoshi T, Watanabe T, Miyata S, Kotake K, Muto T, Sugihara K. Results of a Japanese nationwide multi-institutional study on lateral pelvic lymph node metastasis in low rectal cancer: is it regional or distant disease. Ann Surg. 2012;255:1129–34.
JSCCR. Japanese classification of colorectal, appendiceal, and anal carcinoma: the 3rd English edition secondary publication. J Anus Rectum Colon. 2019;3(10):175–950.
Amin MB, Edge SB, Greene FL, et al. AJCC cancer staging manual. 8th edn. Chicago, IL: American College of Surgeons; 2018.
Roels S, Duthoy W, Haustermans K, et al. Definition and delineation of the clinical target volume for rectal cancer. Int J Radiat Oncol Biol Phys. 2006;65:1129–42.
Hashiguchi Y, Muro K, Saito Y, et al. Japanese society for cancer of the colon and rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 2020;25:1–42.
Ouchi Y, Rakugi H, Arai H, et al. Redefining the elderly as aged 75 years and older: proposal from the Joint Committee of Japan Gerontological Society and the Japan Geriatrics Society. Geriatr Gerontol Int 2017;17:1045–7.
Chen CH, Hsieh MC, Hsiao PK, Lin EK, Lu YJ, Wu SY. A critical reappraisal for the value of tumor size as a prognostic variable in rectal adenocarcinoma. J Cancer. 2017;8:1927–34.
The lymph node committee of JSCCR. Record of proceedings. http://mail.jsccr.jp/committee/pdf/lymph/proj_lymph201606.pdf.
JSCCR. Japanese classification of colorectal, appendiceal, and anal carcinoma. 2nd edn. Tokyo: Kanehara & Co Ltd; 2009.
Ogura A, Konishi T, Beets GL, Cunningham C, Garcia-Aguilar J, Iversen H, et al. Lateral nodal features on restaging magnetic resonance imaging associated with lateral local recurrence in low rectal cancer after neoadjuvant chemoradiotherapy or radiotherapy. JAMA Surg. 2019;154:e192172.
Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 8th edn. Oxford, UK: John Wiley & Sons; 2017.
Edge S, Compton CC, Fritz AG, et al. AJCC cancer staging manual. 7th edn. New York, NY: Springer; 2010.
Benson AB, Venook AP, Al-Hawary MM, et al. Rectal cancer, version 2.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2018;16:874–901.
Ishihara S, Kawai K, Tanaka T, et al. Oncological outcomes of lateral pelvic lymph node metastasis in rectal cancer treated with preoperative chemoradiotherapy. Dis Colon Rectum. 2017;60:469–76.
Wo JY, Anker CJ, Ashman JB, et al. Radiation therapy for rectal cancer: executive summary of an ASTRO clinical practice guideline. Pract Radiat Oncol. 2021;11:13–25.
Kanemitsu Y, Komori K, Shida D, et al. Potential impact of lateral lymph node dissection (LLND) for low rectal cancer on prognoses and local control: a comparison of 2 high-volume centers in Japan that employ different policies concerning LLND. Surgery. 2017;162:303–14.
Ogura A, Konishi T, Cunningham C, et al. Neoadjuvant (chemo)radiotherapy with total mesorectal excision only is not sufficient to prevent lateral local recurrence in enlarged nodes: results of the multicenter lateral node study of patients with low cT3/4 rectal cancer. J Clin Oncol. 2019;37:33–43.
Watanabe T, Ishihara S. Management of lateral pelvic lymph nodes: rectal cancer. 1st edn. Cham, Switzerland: Springer; 2018.
Inoue H, Sasaki K, Nozawa H, et al. Therapeutic significance of D3 dissection for low rectal cancer: a comparison of dissections between the lateral pelvic lymph nodes and the lymph nodes along the root of the inferior mesenteric artery in a multicenter retrospective cohort study. Int J Colorectal Dis. 2021;36:1263–70.
Ueno M, Oya M, Azekura K, et al. Incidence and prognostic significance of lateral lymph node metastasis in patients with advanced low rectal cancer. Br J Surg. 2005;92:756–63.
Schaap DP, Boogerd L, Konishi T, Cunningham C, Ogura A, Garcia-Aguilar J, et al. Rectal cancer lateral lymph nodes: multicentre study of the impact of obturator and internal iliac nodes on oncological outcomes. Br J Surg. 2021;108:205–13.
Viganò L, Langella S, Ferrero A, Russolillo N, Sperti E, Capussotti L. Colorectal cancer with synchronous resectable liver metastases: monocentric management in a hepatobiliary referral center improves survival outcomes. Ann Surg Oncol. 2013;20:938–45.
Tsukamoto S, Fujita S, Ota M, et al. Long-term follow-up of the randomized trial of mesorectal excision with or without lateral lymph node dissection in rectal cancer (JCOG0212). Br J Surg. 2020;107:586–94.
Georgiou P, Tan E, Gouvas N, Antoniou A, Brown G, Nicholls RJ, et al. Extended lymphadenectomy versus conventional surgery for rectal cancer: a meta-analysis. Lancet Oncol. 2009;10:1053–62.
Wilson MK, Collyar D, Chingos DT, et al. Outcomes and end points in cancer trials: bridging the divide. Lancet Oncol. 2015;16:e43-52.
Kim HJ, Choi GS, Park JS, et al. Selective lateral pelvic lymph node dissection: a comparative study of the robotic versus laparoscopic approach. Surg Endosc. 2018;32:2466–73.
Kawai K, Hata K, Tanaka T, Nishikawa T, Otani K, Murono K, et al. Learning curve of robotic rectal surgery with lateral lymph node dissection: cumulative sum and multiple regression analyses. J Surg Educ. 2018;75:1598–605.
Aiba T, Uehara K, Mukai T, et al. Transanal extended rectal surgery with lateral pelvic lymph node dissection. Tech Coloproctol. 2018;22:893–4.
Masubuchi S, Okuda J, Hamamoto H, et al. Totally extraperitoneal approach to laparoscopic lateral lymph node dissection for patients with recurrent lateral pelvic lymph nodes after rectal cancer surgery: a novel technique-M TEP LLND. Surg Today. 2019;49:981–4.
Sun Y, Zhang Z, Zhou Y, et al. Fascial space priority approach in laparoscopy: lateral pelvic lymph node dissection for advanced low rectal cancer. Tech Coloproctol. 2020;24:335–6.
Yano H, Moran BJ. The incidence of lateral pelvic side-wall nodal involvement in low rectal cancer may be similar in Japan and the West. Br J Surg. 2008;95:33–49.
Bokey L, Chapuis PH, Dent OF. Impact of obesity on complications after resection for rectal cancer. Colorectal Dis. 2014;16:896–906.
Sugihara K, Kobayashi H, Kato T, et al. Indication and benefit of pelvic sidewall dissection for rectal cancer. Dis Colon Rectum. 2006;49:1663–72.
Acknowledgment
Dr. Zhifen Chen was financially supported by the National Clinical Key Specialty Construction Project (General Surgery) of China (Grant No. 2012-649) and sponsored by the Fujian Medical University Union Hospital to study at the University of Tokyo Hospital. The authors thank all members and staff of the JSCCR member institutions for collecting data for the Japanese Colorectal Cancer Registry. The authors also thank Editage (www.editage.cn) for English language editing.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article was partially submitted to the 14th Chinese Academic Congress of Colorectal Surgery in conjunction with the International Summit of Colorectal Surgery as poster which was held in Shanghai, China, on October 15 to 17, 2021.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, Z., Sasaki, K., Murono, K. et al. Oncologic Status of Obturator Lymph Node Metastases in Locally Advanced Low Rectal Cancer: A Japanese Multi-Institutional Study of 3487 Patients. Ann Surg Oncol 29, 4210–4219 (2022). https://doi.org/10.1245/s10434-022-11372-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11372-z