Abstract
Background
Among patients with multifocal or multicentric (MF/MC) breast cancer (BC) of similar morphology, concordance in Oncotype DX recurrence scores (RS) between tumors has been reported to be 87%. The effect of age and variation in histologic subtypes on RS concordance according to TAILORx criteria is unknown.
Methods
We identified patients with MF/MC, estrogen receptor-positive, HER2-negative, node-negative BC with two or more RS results treated at our institution from 2009 to 2018. Patients were analyzed by age group (≤ 50 and > 50 years). Low- and high-risk cut-offs were RS ≤ 25 and > 25 for age > 50 years, and RS ≤ 20 and > 20 for age ≤ 50 years. RS concordance was defined as no change in management based on RS variation between lesions.
Results
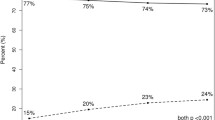
Overall, 120 patients with MF/MC BC were identified—82 (68.3%) aged > 50 years and 38 (31.7%) aged ≤ 50 years. Patients aged ≤ 50 years had higher mean RS for both multifocal (20 vs. 14; p = 0.006) and multicentric (17 vs. 13; p = 0.003) tumors and more frequently had high-risk tumors (p < 0.0001). Among patients aged > 50 years, 95.1% had RS concordance between tumors (same subtype, 98.2%; variable subtype, 88.9%; p = 0.1). Among patients aged ≤ 50 years, RS concordance was 81.6%.
Conclusions
Among patients with MF/MC BC, RS concordance was high, particularly in those aged > 50 years with tumors of the same histologic subtype. RS testing of one focus may be sufficiently prognostic and predictive in patients aged > 50 years, regardless of subtype concordance. Testing of individual foci should be considered in patients aged ≤ 50 years due to a higher likelihood of RS discordance.
Similar content being viewed by others
References
American Cancer Society. Breast Cancer Facts & Figures 2019–2020. Atlanta, GA: American Cancer Society; 2019. Available at: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2019-2020.pdf. Accessed 15 Sep 2020.
Gnerlich JL, Deshpande AD, Jeffe DB, Sweet A, White N, Margenthaler JA. Elevated breast cancer mortality in women younger than age 40 years compared with older women is attributed to poorer survival in early-stage disease. J Am Coll Surg. 2009;208(3):341–7.
Fredholm H, Eaker S, Frisell J, Holmberg L, Fredriksson I, Lindman H. Breast cancer in young women: poor survival despite intensive treatment. PLoS One. 2009;4(11):e7695.
Cancello G, Maisonneuve P, Rotmensz N, et al. Prognosis and adjuvant treatment effects in selected breast cancer subtypes of very young women (< 35 years) with operable breast cancer. Ann Oncol. 2010;21(10):1974–81.
Collins LC, Marotti JD, Gelber S, et al. Pathologic features and molecular phenotype by patient age in a large cohort of young women with breast cancer. Breast Cancer Res Treat. 2012;131(3):1061–6.
Morrison DH, Rahardja D, King E, Peng Y, Sarode VR. Tumour biomarker expression relative to age and molecular subtypes of invasive breast cancer. Br J Cancer. 2012;107(2):382–7.
Rosenberg SM, Partridge AH. Management of breast cancer in very young women. Breast. 2015;24 Suppl 2:S154–8.
Sparano JA, Gray RJ, Makower DF, et al. Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med. 2018;379(2):111–21.
Lynch SP, Lei X, Chavez-MacGregor M, et al. Multifocality and multicentricity in breast cancer and survival outcomes. Ann Oncol. 2012;23(12):3063–9.
Grabenstetter A, Brogi E, Chou JF, et al. Multifocal/multicentric ipsilateral invasive breast carcinomas with similar histology: is multigene testing of all individual foci necessary? Ann Surg Oncol. 2019;26(2):329–35.
Amin MB, Greene FL, Edge SB, et al. The Eighth Edition AJCC Cancer Staging Manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67(2):93–9.
Lester SC, Bose S, Chen YY, et al. Protocol for the examination of specimens from patients with invasive carcinoma of the breast. Arch Pathol Lab Med. 2009;133(10):1515–38.
Wolters R, Wockel A, Janni W, et al. Comparing the outcome between multicentric and multifocal breast cancer: what is the impact on survival, and is there a role for guideline-adherent adjuvant therapy? A retrospective multicenter cohort study of 8,935 patients. Breast Cancer Res Treat. 2013;142(3):579–90.
Vlastos G, Rubio IT, Mirza NQ, et al. Impact of multicentricity on clinical outcome in patients with T1-2, N0-1, M0 breast cancer. Ann Surg Oncol. 2000;7(8):581–7.
Vera-Badillo FE, Napoleone M, Ocana A, et al. Effect of multifocality and multicentricity on outcome in early stage breast cancer: a systematic review and meta-analysis. Breast Cancer Res Treat. 2014;146(2):235–44.
Singh K, Wang Y, Marketkar S, Kalife ET, Steinhoff MM. Comparison of estrogen receptor, progesterone receptor and HER2 results in concurrent ipsilateral samples with invasive breast carcinoma: a retrospective study of 246 biopsies from 119 patients. Hum Pathol. 2017;65:123–32.
Yates LR, Gerstung M, Knappskog S, et al. Subclonal diversification of primary breast cancer revealed by multiregion sequencing. Nat Med. 2015;21(7):751–9.
Desmedt C, Fumagalli D, Pietri E, et al. Uncovering the genomic heterogeneity of multifocal breast cancer. J Pathol. 2015;236(4):457–66.
Buggi F, Folli S, Curcio A, et al. Multicentric/multifocal breast cancer with a single histotype: is the biological characterization of all individual foci justified? Ann Oncol. 2012;23(8):2042–6.
Allison KH, Kandalaft PL, Sitlani CM, Dintzis SM, Gown AM. Routine pathologic parameters can predict Oncotype DX recurrence scores in subsets of ER positive patients: who does not always need testing? Breast Cancer Res Treat. 2012;131(2):413–24.
Hanna MG, Bleiweiss IJ, Nayak A, Jaffer S. Correlation of Oncotype DX recurrence score with histomorphology and immunohistochemistry in over 500 patients. Int J Breast Cancer. 2017;2017:1257078.
Anastasiadi Z, Lianos GD, Ignatiadou E, Harissis HV, Mitsis M. Breast cancer in young women: an overview. Updates Surg. 2017;69(3):313–7.
Kheirelseid EH, Boggs JM, Curran C, et al. Younger age as a prognostic indicator in breast cancer: a cohort study. BMC Cancer. 2011;11:383.
Liu Z, Sahli Z, Wang Y, Wolff AC, Cope LM, Umbricht CB. Young age at diagnosis is associated with worse prognosis in the Luminal A breast cancer subtype: a retrospective institutional cohort study. Breast Cancer Res Treat. 2018;172(3):689–702.
Sheridan W, Scott T, Caroline S, et al. Breast cancer in young women: have the prognostic implications of breast cancer subtypes changed over time? Breast Cancer Res Treat. 2014;147(3):617–29.
Liu KH, Zhang L, Chen JX, et al. Should women with early breast cancer under 40 years of age have a routine 21-gene recurrence score testing: a SEER database study. Breast. 2020;49:233–41.
Paik S, Shak S, Tang G, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351(27):2817–26.
Funding
This work was supported in part by NIH/NCI Cancer Center Support Grant P30 CA008748.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Kate R. Pawloski, Hannah Y. Wen, Audree B. Tadros, Kelly Abbate, and Mahmoud El-Tamer declare they have no conflicts of interest. Monica Morrow declares the receipt of speaking honoraria from Genomic Health.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pawloski, K.R., Wen, H.Y., Tadros, A.B. et al. Concordance Between 21-Gene Recurrence Scores in Multifocal or Multicentric Breast Carcinomas Differs by Age and Histologic Subtype. Ann Surg Oncol 28, 4256–4262 (2021). https://doi.org/10.1245/s10434-020-09429-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09429-y