Abstract
Background
Several recent studies suggest that serum anti-p53 antibodies (s-p53-Abs) may be combined with other markers to detect esophageal and colorectal cancer. In this study, we assessed the sensitivity and specificity of s-p53-Abs detection of a new electrochemiluminescence immunoassay (ECLIA; Elecsys anti-p53).
Methods
Elecsys anti-p53 assay was used to analyze the level of s-p53-Abs in blood sera from patients with esophageal or colorectal cancer taken before treatment. Control blood sera from healthy volunteers, patients with benign diseases, and patients with autoimmune diseases served as a reference. In addition, squamous cell carcinoma antigen (SCC-Ag) and cytokeratin 19 fragments (CYFRA21-1) were assessed in patients with esophageal cancer, and carcinoembryonic antigen (CEA) and carbohydrate antigen (CA) 19-9 were assessed in patients with colorectal cancer.
Results
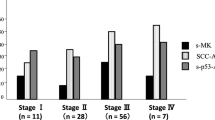
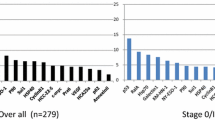
Samples from 281 patients with esophageal cancer, 232 patients with colorectal cancer, and 532 controls were included in the study. The median value of s-p53-Abs in control samples was < 0.02 μg/mL (range < 0.02–29.2 μg/mL). Assuming 98% specificity, the cut-off value was determined as 0.05 μg/mL. s-p53-Abs were detected in 20% (57/281) of patients with esophageal cancer and 18% (42/232) of patients with colorectal cancer. In combination with SCC-Ag and CEA, respectively, s-p53-Abs detected 51% (144/281) of patients with esophageal and 53% (124/232) of patients with colorectal cancer.
Conclusions
The new s-p53-Abs assay Elecsys anti-p53 was useful in detecting esophageal and colorectal cancers with high specificity. Adding s-p53-Abs to conventional markers significantly improved the overall detection rates.





Similar content being viewed by others
Notes
ELECSYS is a trademark of Roche.
References
Soussi T. p53 Antibodies in the sera of patients with various types of cancer: a review. Cancer Res. 2000;60(7):1777–88.
Suppiah A, Greenman J. Clinical utility of anti-p53 auto-antibody: systematic review and focus on colorectal cancer. World J Gastroenterol. 2013;19(29):4651–70.
Shimada H. p53 molecular approach to diagnosis and treatment of esophageal squamous cell carcinoma. Ann Gastroenterol Surg. 2018;2(4):266–73.
Kunizaki M, Hamasaki K, Wakata K, et al. Clinical value of serum p53 antibody in the diagnosis and prognosis of esophageal squamous cell carcinoma. Anticancer Res. 2018;38(3):1807–13.
Tokunaga R, Sakamoto Y, Nakagawa S, Yoshida N, Baba H. The utility of tumor marker combination, including serum P53 antibody, in colorectal cancer treatment. Surg Today. 2017;47(5):636–42.
Liu S, Tan Q, Song Y, Shi Y, Han X. Anti-p53 autoantibody in blood as a diagnostic biomarker for colorectal cancer: a meta-analysis. Scand J Immunol. 2019;91(2):e12829.
Yamashita K, Makino T, Tanaka K, et al. Peritherapeutic serum p53 antibody titers are predictors of survival in patients with esophageal squamous cell carcinoma undergoing neoadjuvant chemotherapy and surgery. World J Surg. 2017;41(6):1566–74.
Kumamoto K, Ishida H, Kuwabara K, et al. Clinical significance of serum anti-p53 antibody expression following curative surgery for colorectal cancer. Mol Clin Oncol. 2017;7(4):595–600.
Daitoku N, Miyamoto Y, Sakamoto Y, et al. Prognostic significance of serum p53 antibody according to KRAS status in metastatic colorectal cancer patients. Int J Clin Oncol. 2020;25(4):651–9.
Suzuki T, Yajima S, Ishioka N, et al. Prognostic significance of high serum p53 antibody titers in patients with esophageal squamous cell carcinoma. Esophagus. 2018;15(4):294–300.
Kawamoto T, Nihei K, Sasai K, Karasawa K. Involved-field chemoradiotherapy for postoperative solitary lymph node recurrence of esophageal cancer. Esophagus. 2018;15(4):256–62.
Muller M, Meyer M, Schilling T, et al. Testing for anti-p53 antibodies increases the diagnostic sensitivity of conventional tumor markers. Int J Oncol. 2006;29(4):973–80.
Xu YW, Peng YH, Chen B, et al. Autoantibodies as potential biomarkers for the early detection of esophageal squamous cell carcinoma. Am J Gastroenterol. 2014;109(1):36–45.
Xu YW, Chen H, Guo HP, et al. Combined detection of serum autoantibodies as diagnostic biomarkers in esophagogastric junction adenocarcinoma. Gastric Cancer. 2019;22(3):546-557.
Wild N, Andres H, Rollinger W, et al. A combination of serum markers for the early detection of colorectal cancer. Clin Cancer Res. 2010;16(24):6111–21.
Shimada H, Ochiai T, Nomura F, Japan p53 Antibody Research G. Titration of serum p53 antibodies in 1,085 patients with various types of malignant tumors: a multiinstitutional analysis by the Japan p53 Antibody Research Group. Cancer. 2003;97(3):682–9.
Himoto T, Yoneyama H, Kurokohchi K, et al. Clinical significance of autoantibodies to p53 protein in patients with autoimmune liver diseases. Can J Gastroenterol. 2012;26(3):125–9.
Takatori H, Kawashima H, Suzuki K, Nakajima H. Role of p53 in systemic autoimmune diseases. Crit Rev Immunol. 2014;34(6):509–16.
Lubin R, Schlichtholz B, Bengoufa D, et al. Analysis of p53 antibodies in patients with various cancers define B-cell epitopes of human p53: distribution on primary structure and exposure on protein surface. Cancer Res. 1993;53(24):5872–76.
Shiratori F, Ito M, Yajima S, et al. The effectiveness of serum midkine in detecting esophageal squamous cell carcinoma. Esophagus. 2019;16(3):246–51.
Schork MA, Williams GW. Number of observations required for the comparison of two correlated proportions. J Commun Stat Simul C. 1980;9(4):349–57.
Ushigome M, Shimada H, Miura Y, et al. Changing pattern of tumor markers in recurrent colorectal cancer patients before surgery to recurrence: serum p53 antibodies, CA19-9 and CEA. Int J Clin Oncol. 2020;25(4):622–32.
Yang Y, Huang X, Zhou L, et al. Clinical use of tumor biomarkers in prediction for prognosis and chemotherapeutic effect in esophageal squamous cell carcinoma. BMC Cancer. 2019;19(1):526.
Acknowledgments
The authors thank Seiko Otsuka for providing technical assistance. Editorial support was provided by Olga Ucar and Clair Clowes of inScience Communications, Springer Healthcare (Chester, UK) and was funded by Roche Diagnostics.
Funding
This work was supported by JSPS KAKENHI (Grant No. JP16K10520) and a research grant from Roche Diagnostics.
Author information
Authors and Affiliations
Contributions
Hideaki Shimada and Roche Diagnostics K.K. were responsible for the study design. Satoshi Yajima, Takashi Suzuki, Yoko Oshima, Fumiaki Shiratori, Kimihiko Funahashi, Shinichi Kawai, Toshihiro Nanki, Sei Muraoka, Yoshihisa Urita, Yoshihisa Saida, Shinichi Okazumi, Yuko Kitagawa, Yuki Hirata, Hirotoshi Hasegawa, Koji Okabayashi, Masahiko Murakami, Takeshi Yamashita, Rei Kato, Hisahiro Matsubara, Kentaro Murakami, Yasuaki Nakajima, and Hideaki Shimada were responsible for sample and data collection. Martin Klammer performed the statistical data analysis. Hideaki Shimada and Hironobu Sugita drafted the initial version of the manuscript. All authors critically reviewed the manuscript and approved the final version for submission.
Corresponding author
Ethics declarations
Disclosure
All sites received research funding from Roche Diagnostics K.K. Hironobu Sugita is an employee of Roche Diagnostics K.K., and Martin Klammer is an employee of Roche Diagnostics GmbH. Sei Muraoka has received consultant fees from Asahikasei Pharma Corporation, and speakers bureau fees from Ono Pharmaceutical Co., Ltd, Eisai Co., Ltd, Asahikasei Pharma Corporation, and Astellas Pharma Inc. Yuko Kitagawa has relevant financial activities outside the submitted work: Taiho Pharmaceutical Co., Ltd; Chugai Pharmaceutical Co., Ltd; Yakult Honsha Co., Ltd; Daiichi Sankyo Company, Ltd; Merck Serono Co., Ltd; Asahikasei Co., Ltd; EA Pharma Co., Ltd; Otsuka Pharmaceutical Co., Ltd; Takeda Pharmaceutical Co., Ltd; Otsuka Pharmaceutical Factory Inc.; Shionogi & Co., Ltd; Kaken Pharmaceutical Co., Ltd; Kowa Pharmaceutical Co., Ltd; Astellas Pharma Inc.; Medicon Inc.; Dainippon Sumitomo Pharma Co., Ltd; Taisho Toyama Pharmaceutical Co., Ltd; Kyouwa Hakkou Kirin Co., Ltd; Pfizer Japan Inc.; Ono Pharmaceutical Co., Ltd; Nihon Pharmaceutical Co., Ltd; Japan Blood Products Organization; Medtronic Japan Co., Ltd; and Sanofi K.K. Yuko Kitagawa has also received grants from Eisai Co., Ltd; Tsumura & Co.; KCI Licensing, Inc.; Abbott Japan Co., Ltd; Fujifilm Toyama Chemical Co., Ltd; and Medicon Inc. Hideaki Shimada has received lecture fees from Ono Pharmaceuticals Ltd and Taiho Pharmaceuticals Ltd. Satoshi Yajima, Takashi Suzuki, Yoko Oshima, Fumiaki Shiratori, Kimihiko Funahashi, Shinichi Kawai, Toshihiro Nanki, Yoshihisa Urita, Yoshihisa Saida, Shinichi Okazumi, Yuki Hirata, Hirotoshi Hasegawa, Koji Okabayashi, Masahiko Murakami, Takeshi Yamashita, Rei Kato, Hisahiro Matsubara, Kentaro Murakami, and Yasuaki Nakajima have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yajima, S., Suzuki, T., Oshima, Y. et al. New Assay System Elecsys Anti-p53 to Detect Serum Anti-p53 Antibodies in Esophageal Cancer Patients and Colorectal Cancer Patients: Multi-institutional Study. Ann Surg Oncol 28, 4007–4015 (2021). https://doi.org/10.1245/s10434-020-09342-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09342-4