Abstract
Background
With more effective screening and treatment strategies, there is debate over whether surgical axillary staging should be deescalated for patients with small favorable breast cancers, such as tubular carcinoma (TC).
Patients and Methods
We identified patients with TC [defined as > 90% tubular tubules (angulated, not multilayered)] and known surgical axillary staging from our institutional database (2000–2018). Using the National Cancer Database (NCDB) (2004–2015), we identified patients with TC, ductal carcinoma in situ (DCIS), and pT1 estrogen receptor (ER)-positive invasive ductal carcinoma (IDC). We determined the rates of lymph node (LN) metastases, and the 5- and 10-year overall survival (OS) for patients with LN-negative versus LN-positive disease using the Kaplan–Meier method and propensity match analysis.
Results
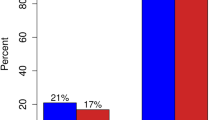
In our institutional cohort, we identified 112 patients with T1 TC; only one (0.9%) patient had nodal involvement. In the NCDB cohort, we identified 6938 patients with T1 TC; 323 (4.7%) patients had axillary LN disease. The rate of axillary LN involvement for TC was comparable to that identified for patients with DCIS (4.2%), and much lower than that found for patients with grade I–III, T1, ER-positive IDC (20.5%), and patients with grade I, T1, ER-positive IDC (14.4%). There was no difference in 5-year (94.6% versus 95.4%, p = 0.67) and 10-year (83.9% versus 85.2%, p = 0.98) OS between TC patients with or without LN involvement. Kaplan–Meier survival curves even after propensity score matching suggest that tubular histology is independently associated with improved survival.
Conclusions
T1 TC is an excellent starting point for deescalation of surgical axillary staging.



Similar content being viewed by others
References
Sentinel Node Vs Observation After Axillary Ultra-souND (SOUND). ClinicalTrials.gov Identifier: NCT 02167490.
Comparison of Axillary Sentinel Lymph Node Biopsy versus No Axillary Surgery (INSEMA). ClinicalTrials.gov Identifier NCT 02466737.
Land SR, Kopec JA, Julian TB, et al. Patient-reported outcomes in sentinel node-negative adjuvant breast cancer patients receiving sentinel-node biopsy or axillary dissection: National Surgical Adjuvant Breast and Bowel Project phase III protocol B-32. J Clin Oncol. 2010;28(25):3929–36.
DiSipio T, Rye S, Newman B, Hayes S. Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. Lancet Oncol. 2013;14(6):500–15.
Gartner R, Jensen MB, Nielsen J, Ewertz M, Kroman N, Kehlet H. Prevalence of and factors associated with persistent pain following breast cancer surgery. JAMA. 2009;302(18):1985–92.
Krag DN, Anderson SJ, Julian TB, et al. Technical outcomes of sentinel-lymph-node resection and conventional axillary-lymph-node dissection in patients with clinically node-negative breast cancer: results from the NSABP B-32 randomised phase III trial. Lancet Oncol. 2007;8(10):881–88.
Giuliano AE, Hunt KK, Ballman KV, et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA. 2011;305(6):569–75.
Rao R, Euhus D, Mayo HG, Balch C. Axillary node interventions in breast cancer: a systematic review. JAMA. 2013;310(13):1385–94.
Mansel RE, Fallowfield L, Kissin M, et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC trial. J Natl Cancer Inst. 2006;98(9):599–609.
Intra M, Rotmensz N, Veronesi P, et al. Sentinel node biopsy is not a standard procedure in ductal carcinoma in situ of the breast: the experience of the European Institute of Oncology on 854 patients in 10 years. Ann Surg. 2008;247(2):315–9.
Francis AM, Haugen CE, Grimes LM, et al. Is sentinel lymph node dissection warranted for patients with a diagnosis of ductal carcinoma in situ? Ann Surg Oncol. 2015;22(13):4270–9.
Cooper HS, Patchefsky AS, Krall RA. Tubular carcinoma of the breast. Cancer. 1978;42(5):2334–42.
Diab SG, Clark GM, Osborne CK, Libby A, Allred DC, Elledge RM. Tumor characteristics and clinical outcome of tubular and mucinous breast carcinomas. J Clin Oncol. 1999;17(5):1442–8.
Weiss MC, Fowble BL, Solin LJ, Yeh IT, Schultz DJ. Outcome of conservative therapy for invasive breast cancer by histologic subtype. Int J Radiat Oncol Biol Phys. 1992;23(5):941–7.
Kader HA, Jackson J, Mates D, Andersen S, Hayes M, Olivotto IA. Tubular carcinoma of the breast: a population-based study of nodal metastases at presentation and of patterns of relapse. Breast J. 2001;7(1):8–13.
Thurman SA, Schnitt SJ, Connolly JL, et al. Outcome after breast-conserving therapy for patients with stage I or II mucinous, medullary, or tubular breast carcinoma. Int J Radiat Oncol Biol Phys. 2004;59(1):152–9.
Leonard CE, Philpott P, Shapiro H, et al. Clinical observations of axillary involvement for tubular, lobular, and ductal carcinomas of the breast. J Surg Oncol. 1999;70(1):13–20.
Sullivan T, Raad RA, Goldberg S, et al. Tubular carcinoma of the breast: a retrospective analysis and review of the literature. Breast Cancer Res Treat. 2005;93(3):199–205.
Fernandez-Aguilar S, Simon P, Buxant F, Fayt I, Noel JC. Is complete axillary lymph node dissection necessary in T1 stage invasive pure tubular carcinomas of the breast? Breast. 2005;14(4):325–8.
Leikola J, Heikkila P, von Smitten K, Leidenius M. The prevalence of axillary lymph-node metastases in patients with pure tubular carcinoma of the breast and sentinel node biopsy. Eur J Surg Oncol. 2006;32(5):488–91.
Cserni G, Bianchi S, Vezzosi V, et al. Sentinel lymph node biopsy and non-sentinel node involvement in special type breast carcinomas with a good prognosis. Eur J Cancer. 2007;43(9):1407–14.
Javid SH, Smith BL, Mayer E, et al. Tubular carcinoma of the breast: results of a large contemporary series. Am J Surg. 2009;197(5):674–7.
Dejode M, Sagan C, Campion L, et al. Pure tubular carcinoma of the breast and sentinel lymph node biopsy: a retrospective multi-institutional study of 234 cases. Eur J Surg Oncol. 2013;39(3):248–54.
Lea V, Gluch L, Kennedy CW, Carmalt H, Gillett D. Tubular carcinoma of the breast: axillary involvement and prognostic factors. ANZ J Surg. 2015;85(6):448–51.
Ramzi S, Hyett EL, Wheal AS, Cant PJ. The case for the omission of axillary staging in invasive breast carcinoma that exhibits a predominant tubular growth pattern on preoperative biopsy. Breast J. 2018;24(4):493–500.
Kitchen PR, Smith TH, Henderson MA, et al. Tubular carcinoma of the breast: prognosis and response to adjuvant systemic therapy. ANZ J Surg. 2001;71(1):27–31.
Winchester DJ, Sahin AA, Tucker SL, Singletary SE. Tubular carcinoma of the breast. Predicting axillary nodal metastases and recurrence. Ann Surg. 1996;223(3):342–347.
Downs-Canner SM, Gaber CE, Louie RJ, et al. Nodal positivity decreases with age in women with early-stage, hormone receptor-positive breast cancer. Cancer. 2020;126(6):1193–201.
Chagpar AB, McMasters KM, Edwards MJ, North American Fareston Tamoxifen Adjuvant T. Can sentinel node biopsy be avoided in some elderly breast cancer patients? Ann Surg. 2009;249(3):455–60.
Boughey JC, Haffty BG, Habermann EB, Hoskin TL, Goetz MP. Has the time come to stop surgical staging of the axilla for all women age 70 years or older with hormone receptor-positive breast cancer? Ann Surg Oncol. 2017;24(3):614–7.
Acknowledgements
We acknowledge and thank all the patients who participated in making this research possible.
Funding
None.
Author information
Authors and Affiliations
Contributions
TUB conceived, developed, and supervised the findings of this work. JB performed the pathology review. JB, TUB, and EO analyzed the institutional and NCDB data. Statistical analysis was performed by EO and SW. ER, SW, and MG provided critical feedback and helped shape the research, analysis, and manuscript. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Disclosure
None pertinent to this work. Outside of submitted work: T.U.B. is a consultant for N-of-One/Qiagen. Spouse is consultant for N-of-One/Qiagen, Tango Therapeutics, and MADALON consulting. Spouse is cofounder of Xsphera Biosciences. Spouse has received research funding from Novartis and Bristol Myers Squibb.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Co-senior authors: Jane Brock and Thanh U. Barbie.
Rights and permissions
About this article
Cite this article
Özkurt, E., Wong, S., Rhei, E. et al. Omission of Surgical Axillary Lymph Node Staging in Patients with Tubular Breast Cancer. Ann Surg Oncol 28, 2589–2598 (2021). https://doi.org/10.1245/s10434-020-09223-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09223-w