Abstract
Introduction
Current recommendations regarding the size of wide local excision (WLE) margins for Merkel cell carcinoma (MCC) are not well established.
Methods
WLE and pathologic margins were respectively reviewed from 79 patients with stage I or II MCC, who underwent WLE at Washington University in St Louis from 2005 to 2019. Outcomes included local recurrence-free survival (LRFS), regional recurrence-free survival (RRFS), distant recurrence-free survival (DRFS), disease-free survival (DFS), and disease-specific survival (DSS).
Results
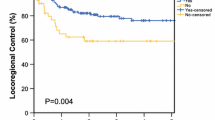
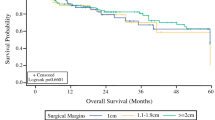
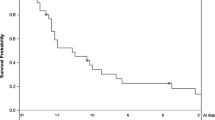
Thirty-two percent of patients received adjuvant radiotherapy (aRT). At 1 year, DFS was 51.3%, 71.4%, and 87.8% for patients with WLE margins < 1 cm, 1–1.9 cm, and ≥ 2 cm, respectively (p = 0.02). At 3 years, the DSS was 57.7%, 82.6%, and 100% for patients with WLE margins < 1 cm, 1–1.9 cm, and ≥ 2 cm, respectively (p = 0.02). Multivariable Cox analysis demonstrated that every 1-cm increase in WLE margins was associated with improved RRFS [hazard ratio (HR) = 0.28, 95% confidence interval (CI): 0.11–0.75], DRFS (HR 0.30, CI 0.08–0.99), DFS (HR 0.42, CI 0.21–0.86), and DSS (HR 0.16, CI 0.04–0.61). WLE and pathologic margin size were moderately-to-strongly correlated (r = 0.66). Close or positive pathologic margins (< 3 mm) were associated with reduced DRFS (HR 6.83, CI 1.80–25.9), DFS (HR 2.98, CI 1.31–6.75), and DSS (HR 3.52, CI 1.14–10.9).
Conclusion
Reduced WLE and pathologic margins were associated with higher risk of relapse and death from MCC. Larger WLE margins are important in populations with lower rates of adjuvant radiation.


Similar content being viewed by others
References
Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78(3):457-463.
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Merkel Cell Carcinoma (Version I.2020). https://www.nccn.org/professionals/physician_gls/pdf/mcc.pdf. Accessed 20 Jan 2020.
Bichakjian CK, Olencki T, Aasi SZ, et al. Merkel cell carcinoma, Version 1.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2018;16(6):742-774.
Bichakjian CK, Olencki T, Alam M, et al. Merkel cell carcinoma, version 1.2014. J Natl Compr Cancer Netw. 2014;12(3):410-424.
Miller SJ, Alam M, Andersen J, et al. Merkel cell carcinoma. J Natl Compr Cancer Netw. 2009;7(3):322-332.
Allen PJ, Bowne WB, Jaques DP, Brennan MF, Busam K, Coit DG. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23(10):2300-2309.
Perez MC, de Pinho FR, Holstein A, et al. Resection margins in Merkel cell carcinoma: is a 1-cm margin wide enough? Ann Surg Oncol. 2018;25(11):3334-3340.
Lewis KG, Weinstock MA, Weaver AL, Otley CC. Adjuvant local irradiation for Merkel cell carcinoma. Arch Dermatol. 2006;142(6):693-700.
Bhatia S, Storer BE, Iyer JG, et al. Adjuvant radiation therapy and chemotherapy in Merkel cell carcinoma: survival analyses of 6908 cases from the National Cancer Data Base. J Natl Cancer Inst. 2016;108(9):dkw042.
Harrington C, Kwan W. Radiotherapy and conservative surgery in the locoregional management of Merkel cell carcinoma: the British Columbia Cancer Agency experience. Ann Surg Oncol. 2016;23(2):573-578.
Mojica P, Smith D, Ellenhorn JD. Adjuvant radiation therapy is associated with improved survival in Merkel cell carcinoma of the skin. J Clin Oncol. 2007;25(9):1043-1047.
Fields RC, Busam KJ, Chou JF, et al. Five hundred patients with Merkel cell carcinoma evaluated at a single institution. Ann Surg. 2011;254(3):465-473; discussion 473-465.
Andea AA, Coit DG, Amin B, Busam KJ. Merkel cell carcinoma: histologic features and prognosis. Cancer. 2008;113(9):2549-2558.
Paulson KG, Iyer JG, Blom A, et al. Systemic immune suppression predicts diminished Merkel cell carcinoma-specific survival independent of stage. J Invest Dermatol. 2013;133(3):642-646.
Boorjian SA, Karnes RJ, Crispen PL, et al. The impact of positive surgical margins on mortality following radical prostatectomy during the prostate specific antigen era. J Urol. 2010;183(3):1003-1009.
Houssami N, Macaskill P, Marinovich ML, Morrow M. The association of surgical margins and local recurrence in women with early-stage invasive breast cancer treated with breast-conserving therapy: a meta-analysis. Ann Surg Oncol. 2014;21(3):717-730.
Van Zee KJ, Subhedar P, Olcese C, Patil S, Morrow M. Relationship between margin width and recurrence of ductal carcinoma in situ: analysis of 2996 women treated with breast-conserving surgery for 30 years. Ann Surg. 2015;262(4):623-631.
Nghiem P, Park SY. Less toxic, more effective treatment—a win-win for patients with Merkel cell carcinoma. JAMA Dermatol. 2019;55(11):1223–1224.
Chen MM, Roman SA, Sosa JA, Judson BL. The role of adjuvant therapy in the management of head and neck merkel cell carcinoma: an analysis of 4815 patients. JAMA Otolaryngol Head Neck Surg. 2015;141(2):137-141.
Eich HT, Eich D, Staar S, et al. Role of postoperative radiotherapy in the management of Merkel cell carcinoma. Am J Clin Oncol. 2002;25(1):50-56.
Servy A, Maubec E, Sugier PE, et al. Merkel cell carcinoma: value of sentinel lymph-node status and adjuvant radiation therapy. Ann Oncol. 2016;27(5):914-919.
Vargo JA, Ghareeb ER, Balasubramani GK, Beriwal S. RE: adjuvant radiation therapy and chemotherapy in Merkel Cell carcinoma: survival analyses of 6908 cases from the National Cancer Data Base. J Natl Cancer Inst. 2017;109(10):djx052.
Tsang G, O’Brien P, Robertson R, Hamilton C, Wratten C, Denham J. All delays before radiotherapy risk progression of Merkel cell carcinoma. Aust. Radiol. 2004;48(3):371-375.
Yusuf M, Gaskins J, Tennant P, Bumpous J, Dunlap N. Survival impact of time to initiation of adjuvant radiation for Merkel cell carcinoma: an analysis of the National Cancer Database. Pract Radiat Oncol. 2019;9(4):e372-e385.
Graboyes EM, Garrett-Mayer E, Ellis MA, et al. Effect of time to initiation of postoperative radiation therapy on survival in surgically managed head and neck cancer. Cancer. 2017;123(24):4841-4850.
Perez CA, Grigsby PW, Castro-Vita H, Lockett MA. Carcinoma of the uterine cervix. I. Impact of prolongation of overall treatment time and timing of brachytherapy on outcome of radiation therapy. Int J Radiat Oncol Biol Phys. 1995;32(5):1275-1288.
Acknowledgments
This work was supported in part by institutional funds from the Departments of Radiation Oncology and Pathology & Immunology, and the Siteman Cancer Center at Washington University in St Louis School of Medicine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors do not have any significant conflicts of interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Andruska, N., Mahapatra, L., Brenneman, R.J. et al. Reduced Wide Local Excision Margins are Associated with Increased Risk of Relapse and Death from Merkel Cell Carcinoma. Ann Surg Oncol 28, 3312–3319 (2021). https://doi.org/10.1245/s10434-020-09145-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09145-7