Abstract
Background
Data to guide axillary management after neoadjuvant endocrine therapy (NET) remain limited.
Methods
We analyzed type of axillary surgery [sentinel lymph node biopsy (SLNB) vs. axillary lymph node dissection (ALND)] and residual nodal disease burden after NET in two cohorts of patients with cT1-4N0-1M0 hormone receptor-positive/human epidermal growth factor receptor 2-negative (HR+/HER2−) breast cancer: Dana-Farber/Brigham and Women’s Cancer Center (DFBWCC) cohort (2015–2018) and the National Cancer Data Base (NCDB) cohort (2012–2016). Cox proportional hazard regression was used to determine adjusted 5-year overall survival (OS) by type of axillary surgery.
Results
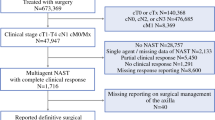
Ninety-four (4.3%) of 2191 HR+/HER2− DFBWCC patients and 4363 (1.5%) of 283,344 NCDB patients were selected for NET. Of those who underwent axillary surgery, 30 (43.5%) in the DFBWCC cohort and 1583 (40.6%) in the NCDB cohort had ALND. Over 90% of cN0 patients in both cohorts had fewer than three positive nodes on final pathology [44 (95.7%) DFBWCC and 2945 (91.3%) NCDB]. In contrast, only 7 (30.4%) DFBWCC patients and 342 (50.7%) NCDB cN1 patients had fewer than three positive nodes. In the DFBWCC patients, there were no locoregional recurrences and four distant recurrences. In the NCDB, 5-year OS did not differ by type of axillary surgery regardless of residual nodal disease burden: 96.6% SLNB versus 97.9% ALND for 0 positive nodes; 84.4% versus 84.4% for one to two positive nodes, and 75.9% versus 77.3% for three or more positive nodes (all p > 0.10).
Conclusions
In cN0 patients selected for NET, > 90% have fewer than three positive nodes at surgery. The lack of a survival difference between SLNB and ALND suggests an opportunity to de-escalate treatment of the axilla in patients with limited residual nodal disease.

Similar content being viewed by others
References
Curigliano G, Burstein HJ, Winer EP, et al. De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann Oncol. 2017;28(8):1700–12.
Spring LM, Gupta A, Reynolds KL, et al. Neoadjuvant endocrine therapy for estrogen receptor-positive breast cancer: a systematic review and meta-analysis. JAMA Oncol. 2016;2(11):1477–86.
Ellis MJ, Suman VJ, Hoog J, et al. Randomized phase II neoadjuvant comparison between letrozole, anastrozole, and exemestane for postmenopausal women with estrogen receptor-rich stage 2 to 3 breast cancer: clinical and biomarker outcomes and predictive value of the baseline PAM50-based intrinsic subtype—ACOSOG Z1031. J Clin Oncol. 2011;29(17):2342–9.
Boughey JC, McCall LM, Ballman KV, et al. Tumor biology correlates with rates of breast-conserving surgery and pathologic complete response after neoadjuvant chemotherapy for breast cancer: findings from the ACOSOG Z1071 (Alliance) Prospective Multicenter Clinical Trial. Ann Surg. 2014;260(4):608–16.
Weiss A, Wong S, Golshan M, et al. Patterns of axillary management in stages 2 and 3 hormone receptor-positive breast cancer by initial treatment approach. Ann Surg Oncol. 2019;26(13):4326–36.
Kantor O, Wong S, Weiss A, et al. Prognostic significance of residual nodal disease after neoadjuvant endocrine therapy for hormone receptor-positive breast cancer. NPJ Breast Cancer. 2020;6:35.
Bilimoria KY, Stewart AK, Winchester DP, Ko CY. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15(3):683–90.
Giuliano AE, Ballman KV, McCall L, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: the ACOSOG Z0011 (Alliance) Randomized Clinical Trial. JAMA. 2017;318(10):918–26.
Donker M, van Tienhoven G, Straver ME, et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): a randomised, multicentre, open-label, phase 3 non-inferiority trial. Lancet Oncol. 2014;15(12):1303–10.
Fisher B, Jeong JH, Anderson S, Bryant J, Fisher ER, Wolmark N. Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N Engl J Med. 2002;347(8):567–75.
Krag DN, Anderson SJ, Julian TB, et al. Technical outcomes of sentinel-lymph-node resection and conventional axillary-lymph-node dissection in patients with clinically node-negative breast cancer: results from the NSABP B-32 randomised phase III trial. Lancet Oncol. 2007;8(10):881–8.
Krag DN, Anderson SJ, Julian TB, et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol. 2010;11(10):927–33.
Weaver DL, Ashikaga T, Krag DN, et al. Effect of occult metastases on survival in node-negative breast cancer. N Engl J Med. 2011;364(5):412–21.
van Nijnatten TJ, Simons JM, Moossdorff M, et al. Prognosis of residual axillary disease after neoadjuvant chemotherapy in clinically node-positive breast cancer patients: isolated tumor cells and micrometastases carry a better prognosis than macrometastases. Breast Cancer Res Treat. 2017;163(1):159–66.
Wong SM, Almana N, Choi J, et al. Prognostic significance of residual axillary nodal micrometastases and isolated tumor cells after neoadjuvant chemotherapy for breast cancer. Ann Surg Oncol. 2019;26(11):3502–9.
Geng C, Chen X, Pan X, Li J. The feasibility and accuracy of sentinel lymph node biopsy in initially clinically node-negative breast cancer after neoadjuvant chemotherapy: a systematic review and meta-analysis. PLoS One. 2016;11(9):e0162605.
Hunt KK, Yi M, Mittendorf EA, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy is accurate and reduces the need for axillary dissection in breast cancer patients. Ann Surg. 2009;250(4):558–66.
Boughey JC, Suman VJ, Mittendorf EA, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial. JAMA. 2013;310(14):1455–61.
Kuehn T, Bauerfeind I, Fehm T, et al. Sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort study. Lancet Oncol. 2013;14(7):609–18.
Boileau JF, Poirier B, Basik M, et al. Sentinel node biopsy after neoadjuvant chemotherapy in biopsy-proven node-positive breast cancer: the SN FNAC study. J Clin Oncol. 2015;33(3):258–64.
Martelli G, Miceli R, Folli S, et al. Sentinel node biopsy after primary chemotherapy in cT2 N0/1 breast cancer patients: long-term results of a retrospective study. Eur J Surg Oncol. 2017;43(11):2012–20.
Galimberti V, Ribeiro Fontana SK, Maisonneuve P, et al. Sentinel node biopsy after neoadjuvant treatment in breast cancer: five-year follow-up of patients with clinically node-negative or node-positive disease before treatment. Eur J Surg Oncol. 2016;42(3):361–8.
Srour MK, Tseng J, Luu M, Alban RF, Giuliano AE, Chung A. Patterns in the use of axillary operations for patients with node-positive breast cancer after neoadjuvant chemotherapy: a national cancer database (NCDB) analysis. Ann Surg Oncol. 2019;26(10):3305–11.
Aihara T, Munakata S, Morino H, Takatsuka Y. Feasibility of sentinel node biopsy for breast cancer after neoadjuvant endocrine therapy: a pilot study. J Surg Oncol. 2004;85(2):77–81.
Sávolt Á, Péley G, Polgár C, et al. Eight-year follow up result of the OTOASOR trial: the optimal treatment of the axilla–surgery or radiotherapy after positive sentinel lymph node biopsy in early-stage breast cancer: a randomized, single centre, phase III, non-inferiority trial. Eur J Surg Oncol. 2017;43(4):672–9.
Phillips JL, Stewart A. Scoping it out: a change in sentinel lymph node surgery coding practice. J Regist Manag. 2012;39(1):29–34.
Funding
Olga Kantor acknowledges the Pamela and Nick Gelsomini Breast Surgical Oncology Fellowship Fund for their support, and Elizabeth A. Mittendorf acknowledges the Rob and Karen Hale Distinguished Chair in Surgical Oncology for their support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Elizabeth A. Mittendorf reports the following personal financial interests: research support for laboratory from GlaxoSmithKline; honoraria from Physician Education Resource; compensated service on Scientific Advisory Boards for Astra-Zeneca, Exact Sciences (formerly Genomic Health), Merck, Peregrine Pharmaceuticals, Roche/Genentech, Sellas Lifesciences, TapImmune Inc; and uncompensated service on steering committees for BMS, Lilly, Roche/Genentech. She also reports the following institutional financial interests: MD Anderson: clinical trial funding from AstraZeneca, EMD Serono, Galena Biopharma, Roche/Genentech; DFCI: clinical trial funding from Roche/Genentech (via an SU2C Grant); and the following non-financial interests and non-remunerated activities: Board of Directors: American Society of Clinical Oncology, Scientific Advisor: Susan G. Komen for the Cure Foundation. Tari A. King reports speaker’s fees and advisory board participation for Exact Sciences (formally Genomic Health).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kantor, O., Wakeman, M., Weiss, A. et al. Axillary Management After Neoadjuvant Endocrine Therapy for Hormone Receptor-Positive Breast Cancer. Ann Surg Oncol 28, 1358–1367 (2021). https://doi.org/10.1245/s10434-020-09073-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09073-6