Abstract
Background
Current national guidelines do not include hyperthermic intraperitoneal chemoperfusion (HIPEC) as treatment for gastric cancer, and there are no completed clinical trials of cytoreduction, gastrectomy, and HIPEC from the US.
Methods
Patients with gastric adenocarcinoma and positive peritoneal cytology or carcinomatosis who had completed systemic chemotherapy and laparoscopic HIPEC underwent cytoreduction, gastrectomy, and HIPEC with 30 mg mitomycin C and 200 mg cisplatin. The primary endpoint was overall survival (OS), with a secondary endpoint of postoperative complications (NCT02891447).
Results
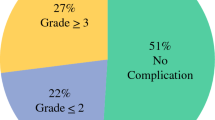
We enrolled 20 patients from September 2016 to March 2019. Six patients had positive cytology only and 14 had carcinomatosis. All patients were treated with systemic chemotherapy with a median of eight cycles (range 5–11 cycles) and at least one laparoscopic HIPEC. The median peritoneal carcinomatosis index at cytoreduction/gastrectomy/HIPEC was 2 (range 0–13). After surgery, the 90-day morbidity and mortality rates were 70% and 0%, respectively. Median length of hospital stay was 13 days (range 7–23 days); median follow-up was 33.5 months; median OS from the date of diagnosis of metastatic disease was 24.2 months; and median OS from the date of cytoreduction, gastrectomy, and HIPEC was 16.1 months. 1-, 2-, and 3-year OS rates from the diagnosis of metastatic disease were 90%, 50%, and 28%, respectively.
Conclusions
Survival rates for patients with gastric adenocarcinoma and peritoneal disease treated with cytoreduction, gastrectomy, and HIPEC are encouraging; our early results are similar to those of recent prospective registry studies. Multi-institutional and cooperative group trials should be supported to confirm survival and safety outcomes.



Similar content being viewed by others
References
Badgwell B, Cormier JN, Krishnan S, et al. Does neoadjuvant treatment for gastric cancer patients with positive peritoneal cytology at staging laparoscopy improve survival? Ann Surg Oncol. 2008;15(10):2684–2691.
National Comprehensive Cancer Network Guidelines Version 4.2019, Gastric Cancer. Available at: www.nccn.org. Accessed 31 Jan 2020.
Thomassen I, Bernards N, van Gestel YR, et al. Chemotherapy as palliative treatment for peritoneal carcinomatosis of gastric origin. Acta Oncol. 2014;53(3):429–432.
Badgwell B, Das P, Ajani J. Treatment of localized gastric and gastroesophageal adenocarcinoma: the role of accurate staging and preoperative therapy. J Hematol Oncol. 2017;10(1):149.
Ikoma N, Blum M, Chiang YJ, et al. Yield of staging laparoscopy and lavage cytology for radiologically occult peritoneal carcinomatosis of gastric cancer. Ann Surg Oncol. 2016;23(13):4332–4337.
Thiels CA, Ikoma N, Fournier K, et al. Repeat staging laparoscopy for gastric cancer after preoperative therapy. J Surg Oncol. 2018;118(1):61–7.
Ikoma N, Chen HC, Wang X, et al. Patterns of initial recurrence in gastric adenocarcinoma in the era of preoperative therapy. Ann Surg Oncol. 2017;24(9):2679–2687.
Yang XJ, Huang CQ, Suo T, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy improves survival of patients with peritoneal carcinomatosis from gastric cancer: final results of a phase III randomized clinical trial. Ann Surg Oncol. 2011;18(6):1575–81.
Rudloff U, Langan RC, Mullinax JE, et al. Impact of maximal cytoreductive surgery plus regional heated intraperitoneal chemotherapy (HIPEC) on outcome of patients with peritoneal carcinomatosis of gastric origin: results of the GYMSSA trial. J Surg Oncol. 2014;110(3):275–284.
Kaplan EMP. Nonparametric estimator from incomplete observations. J Am Stat Assoc. 1958;53:457–481.
Bonnot PE, Piessen G, Kepenekian V, et al. Cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy for gastric cancer with peritoneal metastases (CYTO-CHIP study): a propensity score analysis. J Clin Oncol. 2019;37(23):2028–2040.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–196.
AJCC (2017) Stomach cancer In: Ajani JA, In H, Sano T (Eds.) Cancer staging manual 8. Springer, New York, pp 203–220.
Yan TD, Black D, Sugarbaker PH, et al. A systematic review and meta-analysis of the randomized controlled trials on adjuvant intraperitoneal chemotherapy for resectable gastric cancer. Ann Surg Oncol. 2007;14(10):2702–13.
Coccolini F, Cotte E, Glehen O, et al. Intraperitoneal chemotherapy in advanced gastric cancer. Meta-analysis of randomized trials. Eur J Surg Oncol. 2014;40(1):12–26.
Sun J, Song Y, Wang Z, et al. Benefits of hyperthermic intraperitoneal chemotherapy for patients with serosal invasion in gastric cancer: a meta-analysis of the randomized controlled trials. BMC Cancer. 2012;12:526.
Brenkman HJF, Paeva M, van Hillegersberg R, Ruurda JP, Haj Mohammad N. Prophylactic hyperthermic intraperitoneal chemotherapy (HIPEC) for gastric cancer: a systematic review. J Clin Med. 2019;8(10):1685.
Ishigami H, Fujiwara Y, Fukushima R, et al. Phase III trial comparing intraperitoneal and intravenous Paclitaxel Plus S-1 Versus Cisplatin Plus S-1 in patients with gastric cancer with peritoneal metastasis: PHOENIX-GC Trial. J Clin Oncol. 2018;36(19):1922–29.
Glehen O, Passot G, Villeneuve L, et al. GASTRICHIP: D2 resection and hyperthermic intraperitoneal chemotherapy in locally advanced gastric carcinoma: a randomized and multicenter phase III study. BMC cancer. 2014;14:183.
Glehen O, Gilly FN, Arvieux C, et al. Peritoneal carcinomatosis from gastric cancer: a multi-institutional study of 159 patients treated by cytoreductive surgery combined with perioperative intraperitoneal chemotherapy. Ann Surg Oncol. 2010;17(9):2370–77.
Brandl A, Pachmayr E, Gul-Klein S, Alberto M, Thuss-Patience P, Rau B. Surgical treatment of peritoneal metastases of gastric cancer. Chirurg. 2018;89(9):669–77.
Koemans WJ, van der Kaaij RT, Boot H, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy versus palliative systemic chemotherapy in stomach cancer patients with peritoneal dissemination, the study protocol of a multicentre randomised controlled trial (PERISCOPE II). BMC Cancer. 2019;19(1):420.
Badgwell B, Blum M, Das P, et al. Phase II trial of laparoscopic hyperthermic intraperitoneal chemoperfusion for peritoneal carcinomatosis or positive peritoneal cytology in patients with gastric adenocarcinoma. Ann Surg Oncol. 2017;24(11):3338–44.
Newhook TE, Agnes A, Blum M, et al. Laparoscopic hyperthermic intraperitoneal chemotherapy is safe for patients with peritoneal metastases from gastric cancer and may lead to gastrectomy. Ann Surg Oncol. 2019;26(5):1394–400.
Badgwell B, Roy-Chowdhuri S, Chiang YJ, et al. Long-term survival in patients with metastatic gastric and gastroesophageal cancer treated with surgery. J Surg Oncol. 2015;111(7):875–81.
Mizrak Kaya D, Wang X, Harada K, et al. 101 Long-term survivors who had metastatic gastroesophageal cancer and received local consolidative therapy. Oncology. 2017;93(4):243–48.
Ishigami H, Yamaguchi H, Yamashita H, Asakage M, Kitayama J. Surgery after intraperitoneal and systemic chemotherapy for gastric cancer with peritoneal metastasis or positive peritoneal cytology findings. Gastric cancer. 2017;20 (Suppl 1):128–34.
Ishigami S, Natsugoe S, Nakajo A, et al. Salvage gastrectomy following a combination of biweekly paclitaxel and S-1 for stage IV gastric cancer. J Gastrointestinal. 2008;12(8):1370–5.
MD Anderson Cancer Center. Paclitaxel for the treatment of gastric or gastroeosphageal cancer. ClinicalTrials.gov identifier NCT04220827. Available at: https://clinicaltrials.gov. Accessed 19 May 2020.
Papenfuss WA, Kukar M, Oxenberg J, et al. Morbidity and mortality associated with gastrectomy for gastric cancer. Ann Surg Oncol. 2014;21(9):3008–14.
Acknowledgment
This trial was supported by the Holy Clegg Gastric Cancer Research Fund, the No Stomach for Cancer Award for Gastric Cancer Research, and the University Cancer Foundation via the Institutional Research Grant program at the University of Texas MD Anderson Cancer Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Prajnan Das has received honorarium from Adlai Nortye USA Inc.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Badgwell, B., Ikoma, N., Murphy, M.B. et al. A Phase II Trial of Cytoreduction, Gastrectomy, and Hyperthermic Intraperitoneal Perfusion with Chemotherapy for Patients with Gastric Cancer and Carcinomatosis or Positive Cytology. Ann Surg Oncol 28, 258–264 (2021). https://doi.org/10.1245/s10434-020-08739-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08739-5