Abstract
Objective
The aim of this study was to explore the impact of thyroid antibody status on central lymph node metastases (CLNM) in papillary thyroid carcinoma (PTC) patients with Hashimoto’s thyroiditis (HT).
Methods
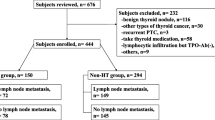
A retrospective analysis was performed on 346 PTC patients with HT who underwent thyroidectomy and ipsilateral central lymph node dissection (CLND). Histopathological characteristics of the tumor and serum levels of thyroid hormone, as well as antibodies, were collected and analyzed.
Results
The multivariate logistic regression analysis showed that being male [odds ratio (OR) 3.269, 95% confidence interval (CI) 1.240–8.619], tumor size > 1 cm [1 cm < diameter (D) ≤ 2 cm: OR 6.947, 95% CI 2.886–16.722; 2 cm < D: OR 5.880, 1.937–17.846], and antibody status [thyroid peroxidase antibody (TPOAb) and thyroglobulin antibody (TgAb) double negative: OR 3.791, 95% CI 1.391–10.331; TPOAb and TgAb double positive: OR 4.047, 95% CI 1.509–10.856; TgAb single positive: OR 6.024, 95% CI 2.019–17.970] were independent risk factors for CLNM. Additionally, a risk-score scale, including sex, antibody status, and tumor size, was established to predict CLNM. The sensitivity, specificity, positive predictive value, and negative predictive value were 55.7%, 84.4%, 74.4%, and 70%, respectively, when the cut-off point was chosen as 3.
Conclusions
Antibody status is a critical independent risk factor for CLNM in PTC patients with HT. For the CLND strategy, a more conservative option could be considered in a low-risk cohort with the following characteristics: female sex, smaller tumor size, and TPOAb single positive.


Similar content being viewed by others
References
Kim SK, Woo JW, Lee JH, Park I, Choe JH, Kim JH, et al. Chronic lymphocytic thyroiditis and BRAF V600E in papillary thyroid carcinoma. Endocr Relat Cancer. 2016;23(1): 27–34.
Ehlers M, Schott M. Hashimoto’s thyroiditis and papillary thyroid cancer: are they immunologically linked?. Trends Endocrinol Metab. 2014;25(12): 656–664.
Ahmed R, Al-shaikh S, Akhtar M. Hashimoto thyroiditis: a century later. Adv Anat Pathol. 2012;19(3): 181–186.
Paulson LM, Shindo ML, Schuff KG. Role of chronic lymphocytic thyroiditis in central node metastasis of papillary thyroid carcinoma. Otolaryngol Head Neck Surg. 2012;147(3): 444–449.
Zeng RC, Jin L, Chen ED, Yang DS, Cai YF, Huang GL, et al. Potential relationship between Hashimoto’s thyroiditis and BRAF(V600E) mutation status in papillary thyroid cancer. Head Neck. 2016;38 Suppl 1:1019–1025.
Zhu YZ, Zheng K, Zhang HH, Chen L, Xue JJ, Ding MJ, et al. The clinicopathologic differences of central lymph node metastasis in predicting lateral lymph node metastasis and prognosis in papillary thyroid cancer associated with or without Hashimoto’s thyroidi. Tumour Biol. 2016;37(5):8037–8045.
Villagelin D, Santos R, Romaldini J. Is diffuse and peritumoural lymphocyte infiltration in papillary thyroid cancer a marker of good prognosis?. J Endocrinol Invest. 2011;34(11):403–408.
Lee J, Kim Y, Choi J, Kim Y. The association between papillary thyroid carcinoma and histologically proven Hashimoto’s thyroiditis: a meta-analysis. Eur J Endocrinol. 2013;168(3):343–349.
Vasileiadis I, Boutzios G, Charitoudis G, Koukoulioti E, Karatzas T. Thyroglobulin antibodies could be a potential predictive marker for papillary thyroid carcinoma. Ann Surg Oncol. 2014;21(8):2725–2732.
Shen C, Zhang X, Qiu Z, Sun Z, Wei W, Hj S, et al. Thyroid autoimmune antibodies in patients with papillary thyroid carcinoma: a double-edged sword. Endocrine. 2017;58(1): 176–183.
Iliadou PK, Effraimidis G, Konstantinos M, Grigorios P, Mitsakis P, Patakiouta F, et al. Chronic lymphocytic thyroiditis is associated with invasive characteristics of differentiated thyroid carcinoma in children and adolescents. Eur J Endocrinol. 2015;173(6):827–833.
[12] Zhang Y, Ma X, Deng F, Liu Z, Wei H, Lwang X, et al. The effect of chronic lymphocytic thyroiditis on patients with thyroid cancer. World J Surg Oncol. 2014;12:277.
Paparodis R, Imam S, Todorova-koteva K, Staii A, Jaume J. Hashimoto’s thyroiditis pathology and risk for thyroid cancer. Thyroid. 2014;24(7):1107–1114.
Kim EY, Kim WG, Kim WB, Kim TY, Kim JM, Ryu J, et al. Coexistence of chronic lymphocytic thyroiditis is associated with lower recurrence rates in patients with papillary thyroid carcinoma. Clin Endocrinol (Oxf). 2009;71(4): 581–586.
Jeong JS, Kim HK, Lee C, Park S, Park JH, Kang S, et al. Coexistence of chronic lymphocytic thyroiditis with papillary thyroid carcinoma: clinical manifestation and prognostic outcome. J Korean Med Sci. 2012;27(8): 883–889.
Wang P, Wang Y, Miao CD, Yu X, Yan H, Xie QP, et al. Defining a new tumour dimension in staging of papillary thyroid carcinoma. Ann Surg Oncol. 2017;24(6):1551–1556.
Zhao QZ, Ming J, Liu CP, Shi L, Xu X, Nie X, et al. Multifocalityand total tumour diameter predict central neck lymph nodemetastases in papillary thyroid microcarcinoma. Ann Surg Oncol. 2013;20(3):746–752.
Zhang Y, Dai J, Wu T, Yang N, Yin Z. The study of the coexistence of Hashimoto’s thyroiditis with papillary thyroid carcinoma. J Cancer Res Clin Oncol. 2014;140(6):1021–1026.
Liu X, Zhu L, Cui D, Wang Z, Chen H, Duan Y, et al. Coexistence of histologically confirmed hashimoto’s thyroiditis with different stages of papillary thyroid carcinoma in a consecutive Chinese cohort. Int J Endocrinol. 2014;2014:769294.
Ahn D, Heo SJ, Park JH, Kim JH, Sohn JH, Park JY, et al. Clinical relationship between Hashimoto s thyroiditis and papillary thyroid cancer. Acta Oncologica. 2011;50(8):1228–1234.
Karatzas T, Vasileiadis I, Zapanti E, Charitoudis G, Karakostas E, Boutzios G. Thyroglobulin antibodies as a potential predictive marker of papillary thyroid carcinoma in patients with indeterminate cytology. Am J Surg. 2016;212(5):946–952.
Dong SA, Xia Q, Wu YJ. High TPOAb Levels (> 1300 IU/mL) indicate multifocal PTC in Hashimoto’s thyroiditis patients and support total thyroidectomy. Otolaryngol Head Neck Surg. 2015;153(1):20–26.
Liang J, Zeng W, Fang F, Yu T, Zhao Y, Fan X, et al. Clinical analysis of Hashimoto thyroiditis coexistent with papillary thyroid cancer in 1392 patients. Acta Otorhinolaryngol Ital. 2017;37(5):393–400.
Weetman A. Autoimmune thyroid disease. Autoimmunity. 2004;37(4):337–340.
Rebuffat SA, Nguyen B, Robert B, Castex F, Peraldi-roux S. Antithyroperoxidase antibody-dependent cytotoxicity in autoimmune thyroid disease. J Clin Endocrinol Metab. 2007;93(3):929–934.
Xiao G, Gao L. Central lymph node metastasis: is it a reliable indicator of lateral node involvement in papillary thyroid carcinoma? World J Surg. 2010;34(2):237–241.
Lang BH, Ng S, Lau LL, Cowling BJ, Wong KP, Wan KY. A systematic review and meta-analysis of prophylactic central neck dissection on short-term locoregional recurrence in papillary thyroid carcinoma after total thyroidectomy. Thyroid. 2013;23(9):1087–1098.
Lan X, Sun W, Zhang H, Dong W, Wang Z, Zhang T. A meta-analysis of central lymph node metastasis for predicting lateral involvement in papillary thyroid carcinoma. Otolaryngol Head Neck Surg. 2015;153(5):731–738.
Jones MR, Mohamed H, Catlin J, April D, Al-qurayshi Z, Kandil E. The presentation of lymph nodes in Hashimoto’s thyroiditis on ultrasound. Gland Surg. 2015;4(4):301–306.
Paksoy N, Yazal K. Cervical lymphadenopathy associated with Hashimoto’s thyroiditis: an analysis of 22 cases by fine needle aspiration cytology. Acta Cytol. 2009;53(5):491–496.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81703854).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
Xingzhu Wen, Bin Wang, Qianmei Jin, Wei Zhang, and Ming Qiu declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wen, X., Wang, B., Jin, Q. et al. Thyroid Antibody Status is Associated with Central Lymph Node Metastases in Papillary Thyroid Carcinoma Patients with Hashimoto’s Thyroiditis. Ann Surg Oncol 26, 1751–1758 (2019). https://doi.org/10.1245/s10434-019-07256-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-07256-4