Abstract
Background
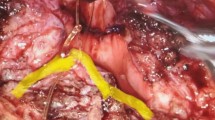
Little data exist about the impact of dissection methods on bladder function during nerve-sparing radical hysterectomy (NSRH). This randomized controlled trial compared the urodynamic and survival outcomes of different methods dissecting the inferior hypogastric plexus (IHP) during laparoscopic NSRH.
Methods
Eligible patients presenting with stage IB cervical cancer from 9 May 2013 to 27 October 2015 were randomized at a ratio of 1:1 and subjected to waterjet (study group) or traditional blunt (control group) dissection of the IHP for laparoscopic type C radical hysterectomy. Participants were subjected to urodynamic evaluations before and after NSRH. The primary measurement was the proportion of patients with residual urine (RU) ≤ 100 ml, while secondary measurements included urodynamic parameters, disease-free survival (DFS), and overall survival (OS).
Results
In total, 191 women met the inclusion criteria, and 160 patients were included in the final analysis, with 80 randomized to each group. At 14 days after NSRH, the study group had more patients with RU ≤ 100 ml than the control group (82.5% vs. 62.5%, p = 0.005). The study group had similar urodynamic outcomes of preoperative and postoperative tests. Comparison with the study group and preoperative tests revealed the control group had significant bladder function impairment at 4 months after NSRH. After a median follow-up of 33 months, the dissection methods had no significant impact on DFS or OS.
Conclusions
Waterjet dissection of the IHP in laparoscopic NSRH resulted in a more rapid return of normal urodynamics without compromising survival outcome.
ClinicalTrials.gov Identifiers NCT03015376 (PUMCH-OBGYN-2013), NCT03291236 (SOCM-1).


Similar content being viewed by others
References
Zullo MA, Manci N, Angioli R, Muzii L, Panici PB. Vesical dysfunctions after radical hysterectomy for cervical cancer: a critical review. Crit. Rev. Oncol. Hematol. 2003;48:287–93.
Xue Z, Zhu X, Teng Y. Comparison of nerve-sparing radical hysterectomy and radical hysterectomy: a systematic review and meta-analysis. Cell Physiol. Biochem. 2016;38:1841–50.
Kim HS, Kim K, Ryoo SB, et al. Conventional versus nerve-sparing radical surgery for cervical cancer: a meta-analysis. J. Gynecol. Oncol. 2015;26:100–10.
Kanao H, Fujiwara K, Ebisawa K, Hada T, Ota Y, Andou M. Various types of total laparoscopic nerve-sparing radical hysterectomies and their effects on bladder function. J. Gynecol. Oncol. 2014;25:198–205.
Todo Y, Kuwabara M, Watari H, et al. Urodynamic study on postsurgical bladder function in cervical cancer treated with systematic nerve-sparing radical hysterectomy. Int. J. Gynecol. Cancer 2006;16:369–75.
Maneschi F. Urodynamic study of bladder function following nerve sparing radical hysterectomy. J. Gynecol. Oncol. 2014;25:159–61.
Maneschi F, Ianiri P, Sarno M, Gagliardi F, Panici PB. Nerve-sparing class III–IV radical hysterectomy: urodynamic study and surgical technique. Int. J. Gynecol. Cancer 2012;22:675–80.
Kruppa J, Kavvadias T, Amann S, Baessler K, Schuessler B. Short and long-term urodynamic and quality of life assessment after nerve sparing radical hysterectomy: a prospective pilot study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016;201:131–4.
Roh JW, Lee DO, Suh DH, et al. Efficacy and oncologic safety of nerve-sparing radical hysterectomy for cervical cancer: a randomized controlled trial. J. Gynecol. Oncol. 2015;26:90–9.
Rob L, Halaska M, Robova H. Nerve-sparing and individually tailored surgery for cervical cancer. Lancet Oncol. 2010;11:292–301.
Cibula D, Abu-Rustum NR, Benedetti-Panici P, et al. New classification system of radical hysterectomy: emphasis on a three-dimensional anatomic template for parametrial resection. Gynecol. Oncol. 2011;122:264–8.
Querleu D, Morrow CP. Classification of radical hysterectomy. Lancet Oncol. 2008;9:297–303.
Common Terminology Criteria for Adverse Events (CTCAE) v4.03. http://evs.nci.nih.gov/ftp1/CTCAE/. Accessed 10 Dec 2012.
Du R, Li L, Ma S, Tan X, Zhong S, Wu M. Lymph nodes metastasis in cervical cancer: Incidences, risk factors, consequences and imaging evaluations. Asia Pac. J. Clin. Oncol. 2018;14:e380–e385.
Haylen BT, de Ridder D, Freeman RM, et al. An international urogynecological association (IUGA)/international continence society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol. Urodyn. 2010;29:4–20.
Possover M, Stober S, Plaul K, Schneider A. Identification and preservation of the motoric innervation of the bladder in radical hysterectomy type III. Gynecol. Oncol. 2000;79:154–7.
Trimbos JB, Maas CP, Deruiter MC, Peters AA, Kenter GG. A nerve-sparing radical hysterectomy: guidelines and feasibility in Western patients. Int. J. Gynecol. Cancer 2001;11:180–6.
Kato K, Suzuka K, Osaki T, Tanaka N. Unilateral or bilateral nerve-sparing radical hysterectomy: a surgical technique to preserve the pelvic autonomic nerves while increasing radicality. Int. J. Gynecol. Cancer 2007;17:1172–8.
Shi R, Wei W, Jiang P. Laparoscopic nerve-sparing radical hysterectomy for cervical carcinoma: emphasis on nerve content in removed cardinal ligaments. Int. J. Gynecol. Cancer 2016;26:192–198.
Liu Z, Li X, Tao Y, et al. Clinical efficacy and safety of laparoscopic nerve-sparing radical hysterectomy for locally advanced cervical cancer. Int. J. Surg. 2016;25:54–58.
Oda Y, Todo Y, Hanley S, et al. Risk factors for persistent low bladder compliance after radical hysterectomy. Int. J. Gynecol. Cancer 2011;21:167–72.
Pieterse QD, Kenter GG, Maas CP, et al. Self-reported sexual, bowel and bladder function in cervical cancer patients following different treatment modalities: longitudinal prospective cohort study. Int. J. Gynecol. Cancer 2013;23:1717–25.
Kim HS, Kim TH, Suh DH, et al. Success factors of laparoscopic nerve-sparing radical hysterectomy for preserving bladder function in patients with cervical cancer: a protocol-based prospective cohort study. Ann. Surg. Oncol. 2015;22:1987–95.
Hao M, Wang Z, Wei F, Wang J, Wang W, Ping Y. Cavitron ultrasonic surgical aspirator in laparoscopic nerve-sparing radical hysterectomy: a pilot study. Int. J. Gynecol. Cancer 2016;26(3):594–9.
Katahira A, Niikura H, Kaiho Y, et al. Intraoperative electrical stimulation of the pelvic splanchnic nerves during nerve-sparing radical hysterectomy. Gynecol. Oncol. 2005;98:462–6.
Chen CL, Guo HX, Yu YH, et al. The measurement of vesical detrusor electromyographic activity during nerve-sparing radical hysterectomy. Reprod. Sci. 2010;17:1144–52.
Tschan CA, Tschan K, Krauss JK, Oertel J. New applicator improves waterjet dissection quality. Br. J. Neurosurg. 2010;24:641–7.
Tschan CA, Keiner D, Muller HD, et al. Waterjet dissection of peripheral nerves: an experimental study of the sciatic nerve of rats. Neurosurgery 2010;67:368–76.
Moinzadeh A, Hasan W, Spaliviero M, et al. Water jet assisted laparoscopic partial nephrectomy without hilar clamping in the calf model. J. Urol. 2005;174:317–21.
Glybochko PV, Alyaev YG, Rapoport LM, et al. The role of waterjet dissection in improving erectile function and continence after nerve-sparing prostatectomy [in Russian]. Urologiia 2017;(1):43–49.
Izquierdo-Luna JS, Campos-Salcedo JG, Estrada-Carrasco CE, Torres-Gomez JJ, Lopez-Silvestre JC, Zapata-Villalba MA. Robot-assisted laparoscopic partial nephrectomy with hydrodissection. Actas Urol. Esp. 2016;40:333–6.
Corvin S, Sturm W, Schlatter E, Anastasiadis A, Kuczyk M, Stenzl A. Laparoscopic retroperitoneal lymph-node dissection with the waterjet is technically feasible and safe in testis-cancer patient. J. Endourol. 2005;19:823–6.
Shekarriz B, Upadhyay J, Jewett MA. Nerve-sparing retroperitoneal lymphadenectomy using hydro-jet dissection: initial experience. J. Endourol. 2004;18:273–6.
Magistro G, Chapple CR, Elhilali M, et al. Emerging minimally invasive treatment options for male lower urinary tract symptoms. Eur. Urol. 2017;72:986–997.
Granick M, Rubinsky L, Parthiban C, Shanmugam M, Ramasubbu N. Dispersion risk associated with surgical debridement devices. Wounds 2017;29:E88–E91.
Contin P, Kulu Y, Bruckner T, et al. Comparative analysis of late functional outcome following preoperative radiation therapy or chemoradiotherapy and surgery or surgery alone in rectal cancer. Int. J. Colorectal Dis. 2014;29:165–75.
Acknowledgments
The authors acknowledge their thanks to Prof. Lan Zhu from the Department of Obstetrics and Gynecology, PUMCH, for the directions regarding urodynamic tests and their implications. They also acknowledge their thanks to Prof. Quancai Cui from the Department of Pathology, PUMCH, for assistance with the pathological evaluations.
Funding
This study was supported by the Chinese Academy of Medical Sciences Initiative for Innovative Medicine (CAMS-2017-I2M-1-002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
DISCLOSURE
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2019_7228_MOESM3_ESM.docx
Different dissection methods for the inferior hypogastric plexus (IHP) did not have an impact on the disease-free survival (A, log rank test P = 0.897) and overall survival (B, log rank test P = 0295) (DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Li, L., Ma, S., Tan, X. et al. The Urodynamics and Survival Outcomes of Different Methods of Dissecting the Inferior Hypogastric Plexus in Laparoscopic Nerve-Sparing Radical Hysterectomy of Type C: A Randomized Controlled Study. Ann Surg Oncol 26, 1560–1568 (2019). https://doi.org/10.1245/s10434-019-07228-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-07228-8