Abstract
Background
Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) is a treatment option in patients with carcinomatosis from high-grade appendiceal (HGA) primaries. It is unknown if there is a Peritoneal Carcinomatosis Index (PCI) upper limit above which a complete CRS/HIPEC does not assure long-term survival.
Methods
Retrospective analysis from three centers was performed. The PCI was used to grade volume of of disease. Survival in relation to PCI was studied on patients with complete cytoreduction.
Results
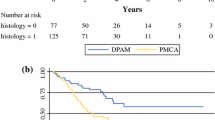
Overall, 521 HGA patients underwent CRS/HIPEC from 1993 to 2015, with complete CRS being achieved in 50% (260/622). Mean PCI was 14.8 (standard deviation 8.7, range 0–36). Median survival for the complete CRS cohort was 6.1 years, while 5- and 10-year survival was 51.7% (standard error [SE] 4.6) and 36.1% (SE 6.3), respectively. Arbitrary cut-off PCI limits with 5-point splits (p = 0.63) were not predictive of a detrimental effect on survival as long as a complete CRS was achieved. A linear effect of the PCI on survival (p = 0.62) was not observed, and single-point PCI cohort splits within a PCI range of < 5 to > 10 were not predictive of survival for complete CRS patients. The PCI correlated with the ability to achieve a complete CRS, with a mean PCI of 14.7 (8.7) for completeness of cytoreduction (CC)0, 22.3 (7.8) for CC1 and 26.1 (9.5) for CC2/3 resections (p = 0.0001, hazard ratio 1.12, 95% confidence interval 1.09), with an HR of 1.15 for each 1-unit increase in the PCI score. Only 21% of the cohort achieved a complete CRS with a PCI ≥ 21.
Conclusions
The PCI correlates with the ability to achieve a complete CRS in carcinomatosis from HGA. PCI is not associated with survival as long as a complete CRS can be achieved.

Similar content being viewed by others
References
Levine EA, Iv JHS, Shen P, Russell GB, Loggie BL, Votanopoulos KI. Intraperitoneal chemotherapy for peritoneal surface malignancy: experience with 1-00 patients. J Am Coll Surg. 2014;218(4):573–85.
Votanopoulos K, Russell G, Randle RW, Shen P, Stewart JH, Levine EA. Peritoneal surface disease (PSD) from apppendiceal cancer treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy: overview of 481 cases. Ann Surg Oncol. 2015;22(4):1274–9.
Glehen O, Kwiatkowski F, Sugarbaker PH, Elias D, Levine EA, De Simone M, et al. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: a multi-institutional study. J Clin Oncol. 2004;22(16):3284–92.
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the eastern cooperative oncology group. Am J Clin Oncol. 1982;5(6):649–55.
Bradley RF, Stewart JH, Russell GB, Levine EA, Geisinger KR. Pseudomyxoma peritonei of appendiceal origin: clinicopathologic analysis of 101 patients uniformly treated at a single institution, with literature review. Am J Surg Pathol. 2006;30(5):551–9.
da Silva RG, Sugarbaker PH. Analysis of prognostic factors in seventy patients having a complete cytoreduction plus perioperative intraperitoneal chemotherapy for carcinomatosis from colorectal cancer. J Am Coll Surg. 2006;203(6):878–86.
Yonemura Y, Canbay E, Ishibashi H. Prognostic factors of peritoneal metastases from colorectal cancer following cytoreductive surgery and perioperative chemotherapy. Sci World J. 2013;2013:978394.
Yan TD, Morris DL. Cytoreductive surgery and perioperative intraperitoneal chemotherapy for isolated colorectal peritoneal carcinomatosis: experimental therapy or standard of care? Ann Surg. 2008;248(5):829–35.
Elias D, Gilly F, Boutitie F, Quenet F, Bereder JM, Mansvelt B et al. Peritoneal colorectal carcinomatosis treated with surgery and perioperative intraperitoneal chemotherapy: retrospective analysis of 523 patients from a multicentric french study. J Clin Oncol. 2010;28(1):63–8.
Verwaal VJ, van Tinteren H, van Ruth S, Zoetmulder FAN. Predicting the survival of patients with peritoneal carcinomatosis of colorectal origin treated by aggressive cytoreduction and hyperthermic intraperitoneal chemotherapy. Br J Surg. 2004;91(6):739–46.
Faron M, Macovei R, Goéré D, Honoré C, Benhaim L, Elias D. Linear relationship of peritoneal cancer index and survival in patients with peritoneal metastases from colorectal cancer. Ann Surg Oncol. 2016;23(1):114–9.
Goéré D, Souadka A, Faron M, Cloutier AS, Viana B, Honoré C, et al. Extent of colorectal peritoneal carcinomatosis: attempt to define a threshold above which HIPEC does not offer survival benefit: a comparative study. Ann Surg Oncol. 2015;22(9):2958–64.
Elias D, Souadka A, Fayard F, Mauguen A, Dumont F, Honore C, et al. Variation in the peritoneal cancer index scores between surgeons and according to when they are determined (before or after cytoreductive surgery). Eur J Surg Oncol. 2012;38(6):503–8.
Kusamura S, Torres Mesa PA, Cabras A, Baratti D, Deraco M. The role of Ki-67 and pre-cytoreduction parameters in selecting diffuse malignant peritoneal mesothelioma (DMPM) patients for cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC). Ann Surg Oncol. 2016;23(5):1468–73.
Levine EA, Iii DGB, Kim MK, Shen P, Iv JHS, Guy C, et al. Gene Expression profiling of peritoneal metastases from appendiceal and colon cancer demonstrates unique biologic signatures and predicts patient outcomes. J Am Coll Surg. 2012;214(4):599–606.
Disclosures
Konstantinos Ioannis Votanopoulos, David Bartlett, Brendan Moran, Choudry M. Haroon, Greg Russell, James F. Pingpank, Lekshmi Ramalingam, Chandrakumaran Kandiah, Konstantinos Chouliaras, Perry Shen, and Edward A. Levine have no disclosures to declare.
Funding
This work was supported by Wake Forest University Biostatistics shared resource NCI CCSG P30CA012197.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Votanopoulos, K.I., Bartlett, D., Moran, B. et al. PCI is Not Predictive of Survival After Complete CRS/HIPEC in Peritoneal Dissemination from High-Grade Appendiceal Primaries. Ann Surg Oncol 25, 674–678 (2018). https://doi.org/10.1245/s10434-017-6315-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6315-3