Abstract
Background
Controversies still persist regarding the terminology and pathologic classification of appendiceal mucinous neoplasms and associated pseudomyxoma peritonei (PMP). We assessed reproducibility and prognostic significance of the classification recently proposed by the Peritoneal Surface Oncology Group International (PSOGI).
Methods
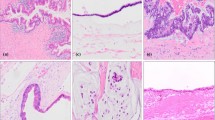
A prospective database of 265 PMP patients uniformly treated by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) from 1995 to 2017 was reviewed. According to the PSOGI, peritoneal disease was retrospectively classified into three categories: low-grade (LG-PMP), high-grade (HG-PMP), and signet-ring cells (SRC-PMP). Acellular mucin (AC) was classified separately. The extent of peritoneal involvement was quantified by the peritoneal cancer index (PCI).
Results
Twenty-six patients were diagnosed with AC (9.8%), 197 with LG-PMP (74.4%), 38 with HG-PMP (14.3%), and 4 with SRC-PMP (1.5%). In the overall series, median follow-up was 65.5 months (95% confidence interval 53.7–78.8) and 10-year overall survival was 62.9% (median 148.7 months). Operative death occurred in 10 patients (3.8%) and major complications occurred in 89 patients (33.6%). Ten-year survival was 89.6% for AC, 63.2% for LG-PMP, 40.1% for HG-PMP, and 0 for SRC-PMP. In a multivariate model, the World Health Organization (WHO) pathological classification independently correlated with survival (p = 0.028). In a separate model, the PSOGI classification did not reach statistical significance (p = 0.149). Completeness of cytoreduction and PCI > 22 correlated with prognosis in both models.
Conclusions
AC and SRC-PMP pathological categories of the PSOGI classification identified two subsets of patients with favorable and exceedingly dismal prognosis, respectively. It remains unclear whether the PSOGI classification might provide better prognostic stratification than the current WHO classification. Further studies in larger prospective series are needed.


Similar content being viewed by others
References
Sugarbaker PH. New standard of care for appendiceal epithelial neoplasms and pseudomyxoma peritonei syndrome? Lancet Oncol. 2006;7:69-76.
Pai RK, Longacre TA. Appendiceal mucinous tumors and pseudomyxoma peritonei: histologic features, diagnostic problems, and proposed classification. Adv Anat Pathol. 2005;12:291-311.
Misdraji J. Yantiss RK, Graeme-Cook FM, et al. Appendiceal mucinous neoplasms: a clinicopathologic analysis of 107 cases. Am J Surg Pathol. 2003; 27:1089–103.
Panarelli NC, Yantiss RK. Mucinous neoplasms of the appendix and peritoneum. Arch Pathol Lab Med. 2011;135:1261–8.
Shetty S, Natarajan B, Thomas P, et al. Proposed classification of pseudomyxoma peritonei: influence of signet ring cells on survival. Am Surg. 2013;79:1171–76.
Davison JM, Choudry HA, Pingpank JF, et al. Clinicopathologic and molecular analysis of disseminated appendiceal mucinous neoplasms: identification of factors predicting survival and proposed criteria for a three-tiered assessment of tumor grade. Mod Pathol. 2014;27:1521–39.
Ronnett BM, Zahn CM, Kurman RJ, et al. Disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis: a clinicopathologic analysis of 109 cases with emphasis on distinguishing pathologic features, site of origin, prognosis and relationship to “pseudomyxoma peritonei”. Am J Surg Pathol. 1995;19:1390–1408.
Szych C, Staebler A, Connolly DC, et al. Molecular genetic evidence supporting the clonality and appendiceal origin of pseudomyxoma peritonei in women. Am J Pathol. 1999;154:1849–55
Carr NJ, Emory TS, Sobin LH. Epithelial neoplasms of the appendix and colorectum: an analysis of cell proliferation, apoptosis and expression of p53, CD44, bcl-2. Arch Pathol Lab Med. 2002;126:837–41.
Sugarbaker PH. Results of treatment of 385 patients with peritoneal surface spread of appendiceal malignancy. Ann Surg Oncol. 1999;6:727–31.
González-Moreno S, Sugarbaker PH. Right hemicolectomy does not confer a survival advantage in patients with mucinous carcinoma of the appendix and peritoneal seeding. Br J Surg. 2004;91:304–11.
Bradley RF, Stewart JH 4th, Russell GB, et al. Pseudomyxoma peritonei of appendiceal origin: a clinicopathologic analysis of 101 patients uniformly treated at a single institution, with literature review. Am J Surg Pathol. 2006;30:551–9.
Smeenk RM, Verwaal VJ, Antonini N, et al. Survival analysis of pseudomyxoma peritonei patients treated by cytoreductive surgery and hyperthermic chemotherapy. Ann Surg. 2007;245:104–9.
Elias D, Honoré C, Ciuchendéa R, et al. Peritoneal pseudomyxoma: results of a systematic policy of complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Br J Surg. 2008;95:1164–71.
Baratti D, Kusamura S, Nonaka D, et al. Pseudomyxoma peritonei: biological features are the dominant prognostic determinants after complete cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann Surg. 2009;249:243–9.
Elias D, Gilly F, Quenet F, et al. Pseudomyxoma peritonei: a French multicentric study of 301 patients treated with cytoreductive surgery and intraperitoneal chemotherapy. Eur J Surg Oncol. 2010;36:456–62.
Chua TC, Moran BJ, Sugarbaker PH, et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Clin Oncol. 2012;30:2449–56.
Moran B, Cecil T, Chandrakumaran K, Arnold S, et al. The results of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in 1200 patients with peritoneal malignancy. Colorectal Dis. 2015;17:772–8.
Esquivel J, Garcia SS, Hicken W, et al. Evaluation of a new staging classification and a Peritoneal Surface Disease Severity Score (PSDSS) in 229 patients with mucinous appendiceal neoplasms with or without peritoneal dissemination. J Surg Oncol. 2014;110:656–60.
Milovanov V, Sardi A, Studeman K, et al. The 7th edition of the AJCC staging classification correlates with biologic behavior of mucinous appendiceal tumor with peritoneal metastases treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC). Ann Surg Oncol. 2016;23:1928–33.
Wagner PL, Austin F, Zenati M, et al. Oncologic Risk Stratification Following Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy for Appendiceal Carcinomatosis. Ann Surg Oncol. 2016;23:1587–93.
Fernandez RN, Daly GM. Pseudomyxoma peritonei. Arch Surg. 1980;115:409–14.
Gough DB, Donohue JH, Schutt AJ, et al. Pseudomyxoma peritonei. Long-term patient survival with an aggressive regional approach. Ann Surg. 1994;219:112–9.
Miner TJ, Shia J, Jaques DP, et al. Long-term survival following treatment of pseudomyxoma peritonei: an analysis of surgical therapy. Ann Surg. 2005;241:300–8.
Järvinen P, Järvinen HJ, Lepistö A. Survival of patients with pseudomyxoma peritonei treated by serial debulking. Colorectal Dis. 2010;12:868–72.
Carr NJ, Sobin LH. Adenocarcinoma of the appendix, In: Bosman FT, Carneiro CF, Hruban RH, Theise ND (eds). WHO Classification of Tumours of the Digestive System. Lyon: IARC Press; 2010. pp. 122–5.
Carr NJ, Cecil TD, Mohamed F, et al.A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am J Surg Pathol. 2016;40:14–26.
Baratti D, Kusamura S, Milione M, et al. Pseudomyxoma Peritonei of Extra-Appendiceal Origin: A Comparative Study. Ann Surg Oncol. 2016;23:4222–30.
Jaquet P, Sugarbaker PH. Current methodologies for clinical assessment of patients with peritoneal carcinomatosis. J Exp Clin Cancer Res. 1996;15:49–58.
Deraco M, Baratti D, Kusamura S, et al. Surgical technique of parietal and visceral peritonectomy for peritoneal surface malignancies. J Surg Oncol. 2009;100:321–8.
Semino-Mora C, Testerman TL, Liu H, et al. Antibiotic treatment decreases microbial burden associated with pseudomyxoma peritonei and affects β-catenin distribution. Clin Cancer Res. 2013;19:3966–76
Choudry HA, Mavanur A, O’Malley ME, et al. Chronic Anti-inflammatory Drug Therapy Inhibits Gel-Forming Mucin Production in a Murine Xenograft Model of Human Pseudomyxoma Peritonei. Ann Surg Oncol. 2012;19:1402–9.
Pietrantonio F, Perrone F, Mennitto A, et al. Toward the molecular dissection of peritoneal pseudomyxoma. Ann Oncol. 2016;27:2097–103.
Asare EA, Compton CC, Hanna NN, et al. The impact of stage, grade, and mucinous histology on the efficacy of systemic chemotherapy in adenocarcinomas of the appendix: Analysis of the National Cancer Data Base. Cancer. 2016;122:213–21.
AJCC Cancer Staging Manual, Eighth Edition. Springer, LLC; 2017.
Huang Y, Alzahrani NA, Chua TC, et al. Histological subtype remains a significant prognostic factor for survival outcomes in patients with appendiceal mucinous neoplasm with peritoneal dissemination. Dis Colon Rectum. 2017;60:360–7.
Acknowledgments
This study was supported, in part, by grants from the Italian Association for Cancer Research (AIRC) and the Italian Health Minister.
Disclosure
The authors have no financial interests to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baratti, D., Kusamura, S., Milione, M. et al. Validation of the Recent PSOGI Pathological Classification of Pseudomyxoma Peritonei in a Single-Center Series of 265 Patients Treated by Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy. Ann Surg Oncol 25, 404–413 (2018). https://doi.org/10.1245/s10434-017-6252-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6252-1