Abstract
Background
The 7th AJCC T-stage system for adrenocortical carcinoma (ACC), based on size and extra-adrenal invasion, does not adequately stratify patients by survival. Lymphovascular invasion (LVI) is a known poor prognostic factor. We propose a novel T-stage system that incorporates LVI to better risk-stratify patients undergoing resection for ACC.
Method
Patients undergoing curative-intent resections for ACC from 1993 to 2014 at 13 institutions comprising the US ACC Group were included. Primary outcome was disease-specific survival (DSS).
Results
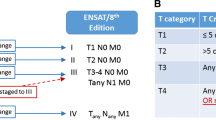
Of the 265 patients with ACC, 149 were included for analysis. The current T-stage system failed to differentiate patients with T2 versus T3 disease (p = 0.10). Presence of LVI was associated with worse DSS versus no LVI (36 mo vs. 168 mo; p = 0.001). After accounting for the individual components of the current T-stage system (size, extra-adrenal invasion), LVI remained a poor prognostic factor on multivariable analysis (hazard ratio 2.14, 95% confidence interval 1.05–4.38, p = 0.04). LVI positivity further stratified patients with T2 and T3 disease (T2: 37 mo vs. median not reached; T3: 36 mo vs. 96 mo; p = 0.03) but did not influence survival in patients with T1 or T4 disease. By incorporating LVI, a new T-stage classification system was created: [T1: ≤ 5 cm, (−)local invasion, (+/−)LVI; T2: > 5 cm, (−)local invasion, (−)LVI OR any size, (+)local invasion, (−)LVI; T3: > 5 cm, (−)local invasion, (+)LVI OR any size, (+)local invasion, (+)LVI; T4: any size, (+)adjacent organ invasion, (+/−)LVI]. Each progressive new T-stage group was associated with worse median DSS (T1: 167 mo; T2: 96 mo; T3: 37 mo; T4: 15 mo; p < 0.001).
Conclusions
Compared with the current T-stage system, the proposed T-stage system, which incorporates LVI, better differentiates T2 and T3 disease and accurately stratifies patients by disease-specific survival. If externally validated, this T-stage classification should be considered for future AJCC staging systems.



Similar content being viewed by others
References
Kebebew E, Reiff E, Duh QY, Clark OH, McMillan A. Extent of disease at presentation and outcome for adrenocortical carcinoma: have we made progress? World J Surg. 2006;30:872–78. https://doi.org/10.1007/s00268-005-0329-x.
Ayala-Ramirez M, et al. Adrenocortical carcinoma: clinical outcomes and prognosis of 330 patients at a tertiary care center. Eur J Endocrinol. 2013;169:891–99. https://doi.org/10.1530/EJE-13-0519.
Schulick RD, Brennan MF. Long-term survival after complete resection and repeat resection in patients with adrenocortical carcinoma. Ann Surg Oncol. 1999;6:719–26.
Postlewait, L. M. et al. Outcomes of adjuvant mitotane after resection of adrenocortical carcinoma: a 13-institution study by the US Adrenocortical Carcinoma Group. J Am Coll Surg. 2015. https://doi.org/10.1016/j.jamcollsurg.2015.12.013.
Bilimoria KY, et al. Adrenocortical carcinoma in the United States: treatment utilization and prognostic factors. Cancer. 2008;113:3130–6. https://doi.org/10.1002/cncr.23886.
Livhits M, Li N, Yeh MW, Harari A. Surgery is associated with improved survival for adrenocortical cancer, even in metastatic disease. Surgery. 2014;156:1531–40; discussion 1540–1. https://doi.org/10.1016/j.surg.2014.08.047.
Lughezzani G, et al. The European network for the study of adrenal tumors staging system is prognostically superior to the international union against cancer-staging system: a North American validation. Eur J Cancer. 2010;46:713–9. https://doi.org/10.1016/j.ejca.2009.12.007.
Fassnacht M, et al. Limited prognostic value of the 2004 International Union Against Cancer staging classification for adrenocortical carcinoma: proposal for a revised TNM classification. Cancer. 2009:115:243–50. https://doi.org/10.1002/cncr.24030.
Amin MB, et al. The eighth edition AJCC cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67:93–9. https://doi.org/10.3322/caac.21388.
Nilubol N, Patel D, Kebebew E. Does lymphadenectomy improve survival in patients with adrenocortical carcinoma? A population-based study. World J Surg. 2016;40:697–705. https://doi.org/10.1007/s00268-015-3283-2.
Schoppmann SF, et al. Prognostic value of lymphangiogenesis and lymphovascular invasion in invasive breast cancer. Ann Surg. 2004;240:306–12.
Patel SH, Kooby DA, Staley CA 3rd, Sarmiento JM, Maithel SK. The prognostic importance of lymphovascular invasion in cholangiocarcinoma above the cystic duct: a new selection criterion for adjuvant therapy? HPB (Oxford). 2011;13:605–11. https://doi.org/10.1111/j.1477-2574.2011.00335.x.
Fisher SB, et al. Lymphovascular and perineural invasion as selection criteria for adjuvant therapy in intrahepatic cholangiocarcinoma: a multi-institution analysis. HPB (Oxford). 2012;14:514–22. https://doi.org/10.1111/j.1477-2574.2012.00489.x.
Postlewait LM, et al. Proposal for a new T-stage classification system for distal cholangiocarcinoma: a 10-institution study from the US Extrahepatic Biliary Malignancy Consortium. HPB (Oxford). 2016;18:793–9.
Jin LX, et al. Factors associated with recurrence and survival in lymph node-negative gastric adenocarcinoma: a 7-institution study of the US Gastric Cancer Collaborative. Ann Surg. 2015;262:999–1005. https://doi.org/10.1097/SLA.0000000000001084.
Lim SB, et al. Prognostic significance of lymphovascular invasion in sporadic colorectal cancer. Dis Colon Rectum. 2010;53:377–84. https://doi.org/10.1007/DCR.0b013e3181cf8ae5.
Cardona K, et al. Detailed pathologic characteristics of the primary colorectal tumor independently predict outcome after hepatectomy for metastases. Ann Surg Oncol 2013;20:148–54. https://doi.org/10.1245/s10434-012-2540-y.
Ethun CG, et al. A novel pathology-based preoperative risk score to predict locoregional residual and distant disease and survival for incidental gallbladder cancer: a 10-institution study from the US Extrahepatic Biliary Malignancy Consortium. Ann Surg Oncol. 2016. https://doi.org/10.1245/s10434-016-5637-x.
Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471–74. https://doi.org/10.1245/s10434-010-0985-4.
Asare EA, et al. A novel staging system for adrenocortical carcinoma better predicts survival in patients with stage I/II disease. Surgery. 2014;156:1378-85. https://doi.org/10.1016/j.surg.2014.08.018; discussion 1385–6.
Libe R. Adrenocortical carcinoma (ACC): diagnosis, prognosis, and treatment. Front Cell Dev Biol. 2015;3:45. https://doi.org/10.3389/fcell.2015.00045.
Miller BS, Gauger PG, Hammer GD, Giordano TJ, Doherty GM. Proposal for modification of the ENSAT staging system for adrenocortical carcinoma using tumor grade. Langenbecks Arch Surg. 2010;395:955–61. https://doi.org/10.1007/s00423-010-0698-y.
Eifel PJ, Burke TW, Delclos L, Wharton JT, Oswald MJ. Early-stage I adenocarcinoma of the uterine cervix: treatment results in patients with tumors less than or equal to 4 cm in diameter. Gynecol Oncol. 1991;41:199–205.
Eguchi T, et al. Histopathological criteria for additional treatment after endoscopic mucosal resection for esophageal cancer: analysis of 464 surgically resected cases. Mod Pathol. 2006;19:475–80. https://doi.org/10.1038/modpathol.3800557.
Disclosure
No authors have any disclosures relevant to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Poorman, C.E., Ethun, C.G., Postlewait, L.M. et al. A Novel T-Stage Classification System for Adrenocortical Carcinoma: Proposal from the US Adrenocortical Carcinoma Study Group. Ann Surg Oncol 25, 520–527 (2018). https://doi.org/10.1245/s10434-017-6236-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6236-1