Abstract
Purpose
Analyzing the large patient cohort of the multicenter AGO-CaRE-1 study, we compared isolated sentinel lymph node dissection (SLND) with radical lymph node dissection (LND) of the groin in relation to recurrence rates and survival.
Methods
The AGO-CaRE-1 study retrospectively collected data on treatment patterns and follow-up of vulvar cancer patients [International Federation of Gynecology and Obstetrics (FIGO) stage ≥1B] treated at 29 gynecologic cancer centers between 1998 and 2008. This subgroup analysis evaluated the influence of SLND alone on progression-free survival (PFS) and overall survival (OS).
Results
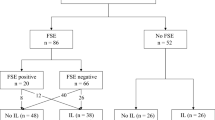
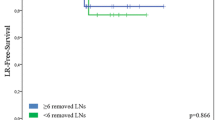
In 487 (63.1%) of 772 included patients with tumors smaller than 4 cm, an LND was performed and no metastatic lymph nodes were detected (LN0). Another 69/772 (8.9%) women underwent SLND alone, showing a negative SLN (SLN0). Tumors in the LN0 group were larger and showed a deeper invasion (LN0 vs. SLN0 tumor diameter: 20.0 vs. 13.0 mm, p < 0.001; depth of invasion: 4.0 vs. 3.0 mm, p = 0.002). After a median follow-up of 33 months (0–156), no significant differences in relation to isolated groin recurrence rates (SLN0 3.0% vs. LN0 3.4%, p = 0.845) were detected. Similarly, univariate 3-year PFS analysis showed no significant differences between both groups (SLN0 82.7% vs. LN0 77.6%, p = 0.230). A multivariate Cox regression analysis, including tumor diameter, depth of invasion, age, grading, and lymphovascular space invasion was performed: PFS [hazard ratio (HR) 0.970, 95% confidence interval (CI) 0.517–1.821] and OS (HR 0.695, 95% CI 0.261–1.849) did not differ significantly between both cohorts.
Conclusion
This subgroup analysis of the large AGO-CaRE-1 study showed similar results for groin LND and SLND alone with regard to recurrence rates and survival in node-negative patients with tumors <4 cm.


Similar content being viewed by others
References
Levenback C, Burke TW, Gershenson DM, Morris M, Malpica A, Ross MI. Intraoperative lymphatic mapping for vulvar cancer. Obstet Gynecol. 1994;84(2):163–7.
Gaarenstroom KN, Kenter GG, Trimbos JB, et al. Postoperative complications after vulvectomy and inguinofemoral lymphadenectomy using separate groin incisions. Int J Gynecol Cancer. 2003;13(4):522–7.
Van der Zee, Ate GJ, Oonk MH, De Hullu, Joanne A, et al. Sentinel node dissection is safe in the treatment of early-stage vulvar cancer. J Clin Oncol. 2008;26(6):884–89.
Wills A, Obermair A. A review of complications associated with the surgical treatment of vulvar cancer. Gynecol Oncol. 2013;131(2):467–79.
Burger MP, Hollema H, Emanuels AG, Krans M, Pras E, Bouma J. The importance of the groin node status for the survival of T1 and T2 vulval carcinoma patients. Gynecol Oncol. 1995;57(3):327–34.
te Grootenhuis NC, van der Zee A, van Doorn HC, et al. Sentinel nodes in vulvar cancer: long-term follow-up of the GROningen INternational Study on Sentinel nodes in Vulvar cancer (GROINSS-V) I. Gynecol Oncol. 2016;140(1):8–14.
Levenback CF, Ali S, Coleman RL, et al. Lymphatic mapping and sentinel lymph node biopsy in women with squamous cell carcinoma of the vulva: a gynecologic oncology group study. J Clin Oncol. 2012;30(31):3786–91.
de Hullu JA, Hollema H, Piers DA, et al. Sentinel lymph node procedure is highly accurate in squamous cell carcinoma of the vulva. J Clin Oncol. 2000;18(15):2811–6.
Hampl M, Hantschmann P, Michels W, Hillemanns P. Validation of the accuracy of the sentinel lymph node procedure in patients with vulvar cancer: results of a multicenter study in Germany. Gynecol Oncol. 2008;111(2):282–8.
de Cicco C, Sideri M, Bartolomei M, et al. Sentinel node biopsy in early vulvar cancer. Br J Cancer. 2000;82(2):295–9.
Hassanzade M, Attaran M, Treglia G, Yousefi Z, Sadeghi R. Lymphatic mapping and sentinel node biopsy in squamous cell carcinoma of the vulva: systematic review and meta-analysis of the literature. Gynecol Oncol. 2013;130(1):237–45.
Mahner S, Jueckstock J, Hilpert F, et al. Adjuvant therapy in lymph node-positive vulvar cancer: the AGO-CaRE-1 Study. J Natl Cancer Inst. 2015;107(3):dju426.
Levenback CF, van der Zee, Ate GJ, Rob L, et al. Sentinel lymph node biopsy in patients with gynecologic cancers. Gynecol Oncol. 2009;114(2):151–6.
Weaver DL, Le UP, Dupuis SL, et al. Metastasis detection in sentinel lymph nodes: comparison of a limited widely spaced (NSABP protocol B-32) and a comprehensive narrowly spaced paraffin block sectioning strategy. Am J Surg Pathol. 2009;33(11):1583–9.
Robison K, Roque D, McCourt C, et al. Long-term follow-up of vulvar cancer patients evaluated with sentinel lymph node biopsy alone. Gynecol Oncol. 2014;133(3):416–20.
Rodier JF, Janser JC, Routiot T, et al. Sentinel node biopsy in vulvar malignancies: a preliminary feasibility study. Oncol Rep. 1999;6(6):1249–1252.
Terada KY, Shimizu DM, Jiang CS, Wong JH. Outcomes for patients with T1 squamous cell cancer of the vulva undergoing sentinel node biopsy. Gynecol Oncol. 2006;102(2):200–203.
Moore RG, Robison K, Brown AK, et al. Isolated sentinel lymph node dissection with conservative management in patients with squamous cell carcinoma of the vulva: a prospective trial. Gynecol Oncol. 2008;109(1):65–70.
Covens A, Vella ET, Kennedy EB, Reade CJ, Jimenez W, Le T. Sentinel lymph node biopsy in vulvar cancer: systematic review, meta-analysis and guideline recommendations. Gynecol Oncol. 2015;137(2):351–61.
Puig-Tintore LM, Ordi J, Vidal-Sicart S, et al. Further data on the usefulness of sentinel lymph node identification and ultrastaging in vulvar squamous cell carcinoma. Gynecol Oncol. 2003;88(1):29–34.
Oonk MH, Hollema H, van der Zee AG. Sentinel node biopsy in vulvar cancer: Implications for staging. Best Pract Res Clin Obstet Gynaecol. 2015;29(6):812–21.
Oonk MH, van Hemel BM, Hollema H, et al. Size of sentinel-node metastasis and chances of non-sentinel-node involvement and survival in early stage vulvar cancer: results from GROINSS-V, a multicentre observational study. Lancet Oncol. 2010;11(7):646–52.
Acknowledgment
The authors thank the statisticians and staff of the Coordinating Center for Clinical Trials of Philipps-University Marburg for their thorough data management, the AGO Study Group staff for their invaluable organizational help, and the Nord-Ostdeutsche Gesellschaft für Gynäkologische Onkologie (NOGGO) for their support. The authors also thank the additional investigators and staff at the 29 clinical sites for their work with clinical data and follow-up: Kreisklinik Altoetting; Charité – University Medicine Berlin Charité Campus Virchow; University Hospital Carl Gustav Carus Dresden; Evangelisches Krankenhaus Duesseldorf; University Hospital Erlangen-Nuernberg; Essen Kliniken Essen-Mitte; Essen University Hospital; Johann Wolfgang Goethe-University Hospital Frankfurt; Alb Fils Kliniken Goeppingen; University Hospital Freiburg; Georg-August University Hospital Goettingen; Ernst-Moritz-Arndt University Hospital University Medicine Greifswald; University Hospital Halle; Hamburg Albertinen Krankenhaus; University Medical Center Hamburg-Eppendorf; Hannover Medical School; University Hospital Kiel; University Hospital Cologne; Otto-Guericke University Hospital Magdeburg; Philipps-University Marburg; University Hospital Munich Grosshadern; University Hospital Munich RdI; Sana Klinikum Offenbach; Oberhavel Kliniken Oranienburg; Elblandkliniken Radebeul; Städtisches Klinikum Solingen; University Hospital Tuebingen; University Hospital Ulm; Klinikum Wolfsburg.
Conflict of interest
Rüdiger Klapdor, Peter Hillemanns, Linn Wölber, Julia Jückstock, Felix Hilpert, Nikolaus de Gregorio, Severine Iborra, Jalid Sehouli, Anika Habermann, Sophie Teresa Fürst, Hans Georg Strauß, Klaus Baumann, Falk Thiel, Alexander Mustea, Werner Meier, Philipp Harter, Pauline Wimberger, Lars Hanker, Barbara Schmalfeldt, Ulrich Canzler, Tanja Fehm, Alexander Luyten, Martin Hellriegel, Jens Kosse, Christoph Heiss, Peer Hantschmann, Peter Mallmann, Berno Tanner, Jacobus Pfisterer, Barbara Richter, Martin Jäger, and Sven Mahner declare that there are no conflicts of interest involved with the presented data.
Funding
The CaRE-1 study was supported by Medac Oncology without restriction in protocol or analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klapdor, R., Hillemanns, P., Wölber, L. et al. Outcome After Sentinel Lymph Node Dissection in Vulvar Cancer: A Subgroup Analysis of the AGO-CaRE-1 Study. Ann Surg Oncol 24, 1314–1321 (2017). https://doi.org/10.1245/s10434-016-5687-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5687-0