Abstract
Background
The impact of fluorodeoxyglucose positron emission tomography–computed tomography (PET-CT) for patients with colorectal liver metastases (CLM) still is debated. Its relevance could be enhanced in the case of recurrent disease. The current study aimed to elucidate the role of PET-CT in restaging and treatment planning for recurrent CLM.
Methods
A series of 352 consecutive patients undergoing their first liver resection for CLM between 2005 and 2014 was reviewed. Of these patients, 224 (63.6 %) had a recurrence. The 107 patients who had received PET-CT at diagnosis of recurrence before chemotherapy were analyzed. CT was available in all cases, and magnetic resonance imaging (MRI) was available in 64 cases.
Results
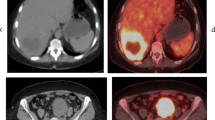
Extrahepatic lesions were found in 59 patients. Liver and lung recurrences were detected with excellent sensitivity by CT/MRI and PET-CT (liver: 100 vs. 96.7 %; lung: 95.8 vs. 95.8 %). In detecting other recurrence sites, PET-CT had higher sensitivity than CT/MRI (91.5 vs. 54.2 %, p < 0.01; lymph nodes: 93.5 vs. 64.5 %, p = 0.011; peritoneum: 80 vs. 20 %, p = 0.023; bones: 87.5 vs. 37.5 %, nonsignificant difference). For 28.8 % (17/59) of the patients, the diagnosis of extrahepatic disease was obtained thanks to PET-CT (39.5 % considering nonpulmonary lesions). PET-CT modified treatment strategy in 16 (14.9 %) patients, excluding from surgery 15 (20.3 %) of 74 patients resectable at CT/MRI. This latter subgroup had a lower survival rate than the patients resectable after PET-CT (2-year survival, 22.7 vs. 77.8 %; p = 0.004), similar to the patients unresectable at CT/MRI (57.6 %).
Conclusions
In the authors’ experience, PET-CT has offered a relevant contribution to restaging of recurrent CLM. It disclosed one fourth of extrahepatic lesions and prevented worthless surgery for about 20 % of patients.

Similar content being viewed by others
References
Adam R, De Gramont A, Figueras J, et al. The oncosurgery approach to managing liver metastases from colorectal cancer: a multidisciplinary international consensus. Oncologist. 2012;17:1225–39.
Vauthey JN, Choti MA, Helton WS. AHPBA/SSO/SSAT Consensus Conference on hepatic colorectal metastases: rationale and overview of the conference. Ann Surg Oncol. 2006;13:1259–60.
Wicherts DA, de Haas RJ, Salloum C, et al. Repeat hepatectomy for recurrent colorectal metastases. Br J Surg. 2013;100:808–18.
Imamura H, Kawasaki S, Miyagawa S, Ikegami T, Kitamura H, Shimada R. Aggressive surgical approach to recurrent tumors after hepatectomy for metastatic spread of colorectal cancer to the liver. Surgery. 2000;127:528–35.
Viganò L, Capussotti L, Lapointe R, et al. Early recurrence after liver resection for colorectal metastases: risk factors, prognosis, and treatment. a LiverMetSurvey-based study of 6025 patients. Ann Surg Oncol. 2014;21:1276–86.
Margonis GA, Spolverato G, Kim Y, Karagkounis G, Choti MA, Pawlik TM. Effect of KRAS mutation on long-term outcomes of patients undergoing hepatic resection for colorectal liver metastases. Ann Surg Oncol. 2015;22:4158–65.
Vauthey JN, Zimmitti G, Kopetz SE, et al. RAS mutation status predicts survival and patterns of recurrence in patients undergoing hepatectomy for colorectal liver metastases. Ann Surg. 2013;258:619–26.
Carpizo DR, D’Angelica M. Liver resection for metastatic colorectal cancer in the presence of extrahepatic disease. Lancet Oncol. 2009;10:801–9.
Elias D, Ouellet JF, Bellon N, Pignon JP, Pocard M, Lasser P. Extrahepatic disease does not contraindicate hepatectomy for colorectal liver metastases. Br J Surg. 2003;90:567–74.
Viganò L, Russolillo N, Ferrero A, Langella S, Sperti E, Capussotti L. Evolution of long-term outcome of liver resection for colorectal metastases: analysis of actual 5-year survival rates over two decades. Ann Surg Oncol. 2012;19:2035–44.
Kekelidze M, D’Errico L, Pansini M, Tyndall A, Hohmann J. Colorectal cancer: current imaging methods and future perspectives for the diagnosis, staging and therapeutic response evaluation. World J Gastroenterol. 2013;19:8502–14.
Moulton CA, Gu CS, Law CH, et al. Effect of PET before liver resection on surgical management for colorectal adenocarcinoma metastases: a randomized clinical trial. JAMA. 2014;311:1863–9.
Ruers TJ, Wiering B, van der Sijp JR, et al. Improved selection of patients for hepatic surgery of colorectal liver metastases with (18)F-FDG PET: a randomized study. J Nucl Med. 2009;50:1036–41.
Maffione AM, Lopci E, Bluemel C, Giammarile F, Herrmann K, Rubello D. Diagnostic accuracy and impact on management of (18)F-FDG PET and PET/CT in colorectal liver metastasis: a meta-analysis and systematic review. Eur J Nucl Med Mol Imaging. 2015;42:152–63.
Fletcher JW, Djulbegovic B, Soares HP, et al. Recommendations on the use of 18F-FDG PET in oncology. J Nucl Med. 2008;49:480–508.
Lopci E, Torzilli G, Poretti D, et al. Diagnostic accuracy of 11C-choline PET/CT in comparison with CT and/or MRI in patients with hepatocellular carcinoma. Eur J Nucl Med Mol Imaging. 2015;42:1399–407.
Yip VS, Poston GJ, Fenwick SW, Wieshmann H, Athwal T, Malik HZ. FDG-PET-CT is effective in selecting patients with poor long-term survivals for colorectal liver metastases. Eur J Surg Oncol. 2014;40:995–9.
Van Cutsem E, Cervantes A, Adam R, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27:1386–422.
Poston GJ, Tait D, O’Connell S, et al. Diagnosis and management of colorectal cancer: summary of NICE guidance. BMJ. 2011;343:d6751.
Serrano PE, Gafni A, Gu CS et al. Positron emission tomography-computed tomography (PET-CT) versus no PET-CT in the management of potentially resectable colorectal cancer liver metastases: cost implications of a randomized controlled trial. J Oncol Pract. 2016;12:e765–74.
de Jong MC, Pulitano C, Ribero D, et al. Rates and patterns of recurrence following curative intent surgery for colorectal liver metastasis: an international multi-institutional analysis of 1669 patients. Ann Surg. 2009;250:440–8.
Torzilli G, Procopio F, Botea F, et al. One-stage ultrasonographically guided hepatectomy for multiple bilobar colorectal metastases: a feasible and effective alternative to the 2-stage approach. Surgery. 2009;146:60–71.
Viganò L, Procopio F, Cimino M, et al. Is tumor detachment from vascular structures equivalent to R0 resection in surgery for colorectal liver metastases? An observational cohort. Ann Surg Oncol. 2016;23(4):1352–60.
Torzilli G. Ultrasound-Guided Liver Surgery: An Atlas. 1st ed. Springer-Verlag, Milan, 2014.
Adam R, Laurent A, Azoulay D, Castaing D, Bismuth H. Two-stage hepatectomy: a planned strategy to treat irresectable liver tumors. Ann Surg. 2000;232:777–85.
Wiering B, Krabbe PF, Jager GJ, Oyen WJ, Ruers TJ. The impact of fluor-18-deoxyglucose-positron emission tomography in the management of colorectal liver metastases. Cancer. 2005;104:2658–70.
Metser U, You J, McSweeney S, Freeman M, Hendler A. Assessment of tumor recurrence in patients with colorectal cancer and elevated carcinoembryonic antigen level: FDG PET/CT versus contrast-enhanced 64-MDCT of the chest and abdomen. AJR Am J Roentgenol. 2010;194:766–71.
Zhang C, Chen Y, Xue H, et al. Diagnostic value of FDG-PET in recurrent colorectal carcinoma: a meta-analysis. Int J Cancer. 2009;124:167–73.
Adam R, de Haas RJ, Wicherts DA, et al. Is hepatic resection justified after chemotherapy in patients with colorectal liver metastases and lymph node involvement? J Clin Oncol. 2008;26:3672–80.
Viganò L, Russolillo N, Ferrero A, et al. Resection of liver metastases from colorectal mucinous adenocarcinoma: is this a different disease? Results of a case-control study. Ann Surg. 2014;260:878–84.
Koh JL, Yan TD, Glenn D, Morris DL. Evaluation of preoperative computed tomography in estimating peritoneal cancer index in colorectal peritoneal carcinomatosis. Ann Surg Oncol. 2009;16:327–33.
Esquivel J, Chua TC, Stojadinovic A, et al. Accuracy and clinical relevance of computed tomography scan interpretation of peritoneal cancer index in colorectal cancer peritoneal carcinomatosis: a multi-institutional study. J Surg Oncol. 2010;102:565–70.
Briggs RH, Chowdhury FU, Lodge JPA, Scarbrook AF. Clinical impact of FDG PET-CT in patients with potentially operable metastatic colorectal cancer. Clin Radiol. 2011;66:1167–74.
van Kessel CS, Buckens CF, van den Bosch MA, et al. Preoperative imaging of colorectal liver metastases after neoadjuvant chemotherapy: a meta-analysis. Ann Surg Oncol. 2012;19:2805–13.
Wiering B, Adang EM, van der Sijp JR, et al. Added value of positron emission tomography imaging in the surgical treatment of colorectal liver metastases. Nucl Med Comm. 2010;31:938–44.
Disclosure
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Viganò, L., Lopci, E., Costa, G. et al. Positron Emission Tomography-Computed Tomography for Patients with Recurrent Colorectal Liver Metastases: Impact on Restaging and Treatment Planning. Ann Surg Oncol 24, 1029–1036 (2017). https://doi.org/10.1245/s10434-016-5644-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5644-y