Abstract
Introduction
Survival in elderly patients undergoing mastectomy or lumpectomy has not been specifically analyzed.
Methods
Patients older than 70 years of age with clinical stage I invasive breast cancer, undergoing mastectomy or lumpectomy with or without radiation, and surveyed within 3 years of their diagnosis, were identified from the Surveillance, Epidemiology, and End Results and medicare health outcomes survey-linked dataset. The primary endpoint was breast cancer-specific survival (CSS).
Results
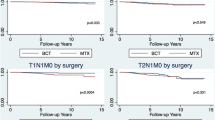
Of 1784 patients, 596 (33.4 %) underwent mastectomy, 918 (51.4 %) underwent lumpectomy with radiation, and 270 (15.1 %) underwent lumpectomy alone. Significant differences were noted in age, tumor size, American Joint Committee on Cancer (AJCC) stage, lymph node status (all p < 0.0001) and number of positive lymph nodes between the three groups (p = 0.003). On univariate analysis, CSS for patients undergoing lumpectomy with radiation [hazard ratio (HR) 0.61, 95 % confidence interval (CI) 0.43–0.85; p = 0.004] was superior to mastectomy. Older age (HR 1.3, 95 % CI 1.09–1.45; p = 0.002), two or more comorbidities (HR 1.57, 95 % CI 1.08–2.26; p = 0.02), inability to perform more than two activities of daily living (HR 1.61, 95 % CI 1.06–2.44; p = 0.03), larger tumor size (HR 2.36, 95 % CI 1.85–3.02; p < 0.0001), and positive lymph nodes (HR 2.83, 95 % CI 1.98–4.04; p < 0.0001) were associated with worse CSS. On multivariate analysis, larger tumor size (HR 1.89, 95 % CI 1.37–2.57; p < 0.0001) and positive lymph node status (HR 1.99, 95 % CI 1.36–2.9; p = 0.0004) independently predicted worse survival.
Conclusions
Elderly patients with early-stage invasive breast cancer undergoing breast conservation have better CSS than those undergoing mastectomy. After adjusting for comorbidities and functional status, survival is dependent on tumor-specific variables. Determination of lymph node status remains important in staging elderly breast cancer patients.



Similar content being viewed by others
References
Howlader N, Noone A, Krapcho M, Garshell J, Miller D, Altekruse S, et al. SEER Cancer Statistics Review, 1975–2012, National Cancer Institute. Bethesda, MD, based on November 2014 SEER data submission, posted to the SEER web site, April 2015. http://seer.cancer.gov/csr/1975_2012/sections.html.
Fisher B, Anderson S, Bryant J, Margolese R, Deutsch M, Fisher ER, et al. Twenty-year follow-up of a randomized trial comparing total for the treatment of invasive breast cancer. N Engl J Med. 2002;347:1233–41.
Lichter AS, Lippman ME, Danforth DN Jr, d’Angelo T, Steinberg SM, deMoss E, et al. Mastectomy versus breast-conserving therapy in the treatment of stage I and II carcinoma of the breast: a randomized trial at the National Cancer Institute. J Clin Oncol. 1992;10:976–83.
Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A, et al. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med. 2002;347:1227–32.
Smith GL, Xu Y, Shih YC, Giordano SH, Smith BD, Hunt KK, et al. Breast-conserving surgery in older patients with invasive breast cancer: current patterns of treatment across the United States. J Am Coll Surg. 2009;209:425–33.e2.
Lee MC, Rogers K, Griffith K, Diehl KA, Breslin TM, Cimmino VM, et al. Determinants of breast conservation rates: reasons for mastectomy at a comprehensive cancer center. Breast J. 2009;15:34–40.
Hwang ES, Lichtensztajn DY, Gomez SL, Fowble B, Clarke CA. Survival after lumpectomy and mastectomy for early stage invasive breast cancer: the effect of age and hormone receptor status. Cancer. 2013;119:1402–11.
Agarwal S, Pappas L, Neumayer L, Kokeny K, Agarwal J. Effect of breast conservation therapy vs mastectomy on disease-specific survival for early-stage breast cancer. JAMA Surg. 2014;149:267–74.
Hofvind S, Holen, Aas T, Roman M, Sebuodegard S, Akslen LA. Women treated with breast conserving surgery do better than those with mastectomy independent of detection mode, prognostic and predictive tumor characteristics. Eur J Surg Oncol. 2015;41:1417–22.
Hutchins LF, Unger JM, Crowley JJ, Coltman CA, Albain KS. Underrepresentation of patients 65 years of age or older in cancer-treatment trials. N Engl J Med. 1999;341:2061–7.
Bouchardy C, Rapiti E, Fioretta G, Laissue P, Neyroud-Caspar I, Schafer P, et al. Undertreatment strongly decreases prognosis of breast cancer in elderly women. J Clin Oncol. 2003;21:3580–7.
Bouchardy C, Rapiti E, Blagojevic S, Vlastos AT, Vlastos G. Older female cancer patients: importance, causes, and consequences of undertreatment. J Clin Oncol. 2007;25:1858–69.
Diab SG, Elledge RM, Clark GM. Tumor characteristics and clinical outcome of elderly women with breast cancer. J Natl Cancer Inst. 2000;92:550–6.
Pierga J-Y, Girre V, Laurence V, Asselain B, Diéras V, Jouve M, et al. Characteristics and outcome of 1755 operable breast cancers in women over 70 years of age. Breast. 2004;13:369–75.
Smith BD, Jiang J, McLaughlin SS, Hurria A, Smith GL, Giordano SH, et al. Improvement in breast cancer outcomes over time: are older women missing out? J Clin Oncol. 2011;29:4647–53.
Schonberg MA, Marcantonio ER, Li D, Silliman RA, Ngo L, McCarthy EP. Breast cancer among the oldest old: tumor characteristics, treatment choices, and survival. J Clin Oncol. 2010;28:2038–45.
Montroni I, Rocchi M, Santini D, Ceccarelli C, Ghignone F, Zattoni D, et al. Has breast cancer in the elderly remained the same over recent decades? A comparison of two groups of patients 70 years or older treated for breast cancer twenty years apart. J Geriatr Oncol. 2014;5:260–5.
Ambs A, Warren JL, Bellizzi KM, Topor M, Haffer SC, Clauser SB. Overview of the SEER–Medicare Health Outcomes Survey linked dataset. Health Care Financ Rev. 2008;29:5–21.
Kent EE, Ambs A, Mitchell SA, Clauser SB, Smith AW, Hays RD. Health-related quality of life in older adult survivors of selected cancers: data from the SEER-MHOS linkage. Cancer. 2015;121:758–65.
Greene FL, American Joint Committee on Cancer, American Cancer Society. AJCC cancer staging manual. New York: Springer; 2002.
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. Geneva: World Health Organization; 2008.
Fritz AG. International classification of diseases for oncology (ICD-O). Geneva: World Health Organization; 2000.
Wallace M, Shelkey M. Katz index of independence in activities of daily living (ADL). Urol Nurs. 2007;27:93–4.
Arias E. United States life tables, 2010. Natl Vital Stat Rep. 2014;63:1–62.
Klepin H, Mohile S, Hurria A. Geriatric assessment in older patients with breast cancer. J Natl Compr Cancer Netw. 2009;7:226–36.
Hughes KS, Schnaper LA, Bellon JR, Cirrincione CT, Berry DA, McCormick B, et al. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013;31:2382–7.
Kunkler IH, Williams LJ, Jack WJL, Cameron DA, Dixon JM. Breast-conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (PRIME II): a randomised controlled trial. Lancet Oncol. 2015;16:266–73.
Fyles AW, Mccready DR, Manchul LA, Trudeau ME, Merante P, Pintilie M, et al. Tamoxifen with or without breast irradiation in women 50 years of age or older with early breast cancer. N Engl J Med. 2004;351:963–70.
Potter R, Gnant M, Kwasny W, Tausch C, Handl-Zeller L, Pakisch B, et al. Lumpectomy plus tamoxifen or anastrozole with or without whole breast irradiation in women with favorable early breast cancer. Int J Radiat Oncol Biol Phys. 2007;68:334–40.
Soulos PR, Yu JB, Roberts KB, Raldow AC, Herrin J, Long JB, et al. Assessing the impact of a cooperative group trial on breast cancer care in the medicare population. J Clin Oncol. 2012;30:1601–7.
McCormick B, Ottesen RA, Hughes ME, Javid SH, Khan SA, Mortimer J, et al. Impact of guideline changes on use or omission of radiation in the elderly with early breast cancer: practice patterns at National Comprehensive Cancer Network institutions. J Am Coll Surg. 2014;219:796–802.
NCCN clinical practice guidelines in oncology (NCCN Guidelines®), Breast Cancer. Version 1.2014. www.NCCN.com (2014).
Rudenstam C-M, Zahrieh D, Forbes JF, Crivellari D, Holmberg SB, Rey P, et al. Randomized trial comparing axillary clearance versus no axillary clearance in older patients with breast cancer: first results of International Breast Cancer Study Group Trial 10-93. J Clin Oncol. 2006;24:337–44.
Early Breast Cancer Trialists’ Collaborative Group. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365:1687–717.
Jagsi R, Abrahamse P, Hawley ST, Graff JJ, Hamilton AS, Katz SJ. Under ascertainment of radiotherapy receipt in surveillance, epidemiology, and end results registry data. Cancer. 2012;118:333–41.
Acknowledgment
This study was supported by Wake Forest University Biostatistics shared resource NCI CCSG P30CA012197.
Conflicts of interest
Harveshp D. Mogal, Clancy Clark, Rebecca Dodson, Nora F. Fino, and Marissa Howard-McNatt have no conflicts of interest to declare.
Ethics statement
Institutional Review Board approval was obtained for this study.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mogal, H.D., Clark, C., Dodson, R. et al. Outcomes After Mastectomy and Lumpectomy in Elderly Patients with Early-Stage Breast Cancer. Ann Surg Oncol 24, 100–107 (2017). https://doi.org/10.1245/s10434-016-5582-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5582-8