Abstract
Background
This retrospective study was designed to estimate the efficacy and toxicity of definitive radiotherapy with concurrent or sequential docetaxel/S-1 for patients with locally advanced esophageal squamous cell carcinoma (ESCC).
Methods
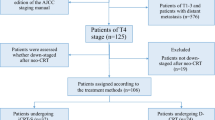
Of the 62 eligible patients enrolled in this study during January 1, 2010 to December 31, 2014 from Qilu Hospital, Shandong University, Shandong Province, 39 patients received 3 cycles of docetaxel/S-1 during and after radiotherapy (concurrent chemoradiotherapy, CCRT), and 23 patients had radiotherapy followed by 3 cycles of docetaxel and S-1 (sequential chemoradiotherapy, SCRT).
Results
The CR of CCRT and SCRT groups were 48.72 and 21.74 %, respectively (p = 0.035). The median progress-free survival (PFS) of CCRT group (23.5 months) was significantly higher than SCRT group (11.7 months; p = 0.004). The median overall survival (OS) of CCRT group (33.5 months) also was significantly higher than SCRT group (24.0 months; p = 0.004). At 2 years, in this patient population, the rate of PFS of CCRT group was (44.2 ± 8.2 %), significantly higher than SCRT group (11.9 ± 9.6 %; p = 0.002). The 2-year OS rate of CCRT (68.6 ± 7.5 %) was significantly higher than SCRT group as well (42.0 ± 14.0 %; p = 0.002). The incidence of adverse events was higher in CCRT than SCRT group. No grade 4 or grade 5 adverse events occurred in our study.
Conclusions
Definitive radiotherapy with concurrent or sequential docetaxel and S-1 for inoperable locally advanced ESCC was very well tolerated and remarkably active. In both CCRT and SCRT groups, acute toxicities were manageable. This regimen holds promises for treatment of esophageal carcinoma and warrants further investigation.

Similar content being viewed by others
References
Chen W, Zheng R, Zeng H, Zhang S, et al. Annual report on status of cancer in China, 2011. Chin J Cancer Res. 2015;27:2–12.
Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics: The impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61:212–36.
Zhang P, Xi M, Zhao L, Li QQ, et al. Efficacy and prognostic analysis of chemoradiotherapy in patients with thoracic esophageal squamous carcinoma with cervical lymph nodal metastasis alone. Radiat Oncol. 2014;9:256.
Mohammad NH, Hulshof MC, Bergman JJ, Geijsen D, et al. Acute toxicity of definitive chemoradiation in patients with inoperable or irresectable esophageal carcinoma. BMC Cancer. 2014;14:56
Tang HR, Ma HF, An SM, Badakhshi H, et al. A phase II study of concurrent chemoradiotherapy with paclitaxel and cisplatin for inoperable esophageal squamous cell carcinoma. Am J Clin Oncol. 2014. doi:10.1097/COC.0000000000000069.
Yvon AM, Wadsworth P, Jordan MA. Taxol suppresses dynamics of individual microtubules in living human tumor cells. Mol Biol Cell. 1999;0:947–59.
Eisenhauer EA, Vermorken JB. The taxoids. Comparative clinical pharmacology and therapeutic potential. Drugs. 1998;55:5–30.
Kelsen DP, Ilson DH. Chemotherapy and combined-modality therapy for esophageal cancer. Chest. 1995;107:224–32.
Eisenhauer EA, Therasseb P, Bogaertsc J, Schwartz LH, et al. New response evaluation criteria in solid tumors: RECIST GUIDELINE VERSION 1.1. EJC Suppl. 2009;45(2):228–47.
Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. 2009.
Kato K, Muro K, Minashi K, Ohtsu A, et al. Phase II study of chemoradiotherapy with 5-fluorouracil and cisplatin for stage II-III esophageal squamous cell carcinoma: JCOG trial (JCOG 9906). Int J Radiat Oncol Biol Phys. 2011;81:684–90.
Gupta A, Roy S, Majumdar A, Hazra A, et al. A randomized study to compare sequential chemoradiotherapy with concurrent chemoradiotherapy for unresectable locally advanced esophageal cancer. Indian J Med Paediatr Oncol. 2014;35:54–9.
Orditura M, Galizia G, Napolitano V, Martinelli E, et al. Weekly chemotherapy with cisplatin and paclitaxel and concurrent radiation therapy as preoperative treatment in locally advanced esophageal cancer: a phase II study. Cancer Invest. 2010;8(8):820–7.
Honing J, Smit JK, Muijs CT, Burgerhof JG, et al. A comparison of carboplatin and paclitaxel with cisplatinum and 5-fluorouracil in definitive chemoradiation in esophageal cancer patients. Ann Oncol. 2014;25:638–43.
Lin CC, Hsu CH, Cheng JC, Wang HP, et al. Concurrent chemoradiotherapy with twice weekly paclitaxel and cisplatin followed by esophagectomy for locally advanced esophageal cancer. Ann Oncol. 2007;18:93–8.
van Meerten E, Muller K, Tilanus HW, Siersema PD, et al. Neoadjuvant concurrent chemoradiation with weekly paclitaxel and carboplatin for patients with oesophageal cancer: a phase II study. Br J Cancer. 2006;94:1389–94.
Pasini F, de Manzoni G, Zanoni A, Grandinetti A. et al. Neoadjuvant therapy with weekly docetaxel and cisplatin, 5-fluorouracil continuous infusion, and concurrent radiotherapy in patients with locally advanced esophageal cancer produced a high percentage of long-lasting pathological complete response: a phase 2 study. Cancer. 2013;119:939–45
van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–84.
Author information
Authors and Affiliations
Corresponding author
Additional information
Bin Yao and Bingxu Tan have contributed equally to this work and should be considered co-first authors.
Rights and permissions
About this article
Cite this article
Yao, B., Tan, B., Wang, C. et al. Comparison of Definitive Chemoradiotherapy in Locally Advanced Esophageal Squamous Cell Carcinoma. Ann Surg Oncol 23, 2367–2372 (2016). https://doi.org/10.1245/s10434-016-5154-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5154-y