Abstract
Background
The relative contribution of biologic subtype to locoregional recurrence (LRR) in patients treated with neoadjuvant chemotherapy (NAC), mastectomy, and postmastectomy radiotherapy (PMRT) is not clearly defined.
Methods
233 patients with stages 2 and 3 breast cancer who received NAC, mastectomy, and PMRT between 2000 and 2009 were included: 53 % (n = 123) had HR+ (ER or PR+/HER2−), 23 % (n = 53) had HER2+ (HER2+/HR+ or HR−), and 24 % (n = 57) had triple-negative (TN) disease (HR−/HER2−). The 5-year LRR rates were estimated by Kaplan–Meier methods. Cox regression analysis was performed to evaluate covariates associated with LRR.
Results
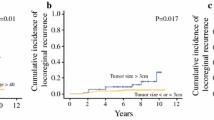
The median follow-up period was 62 months. A pathologic complete response (pCR) was seen in 14 % of the patients. The 5-year LRR rate was 8 % for the entire cohort. The LRR rate was 0 % for the patients with a pCR versus 9 % for the patients without a pCR (p = 0.05). TN disease [Hazard ratio (HR) 4.4; p = 0.003] and pathologic node positivity (HR 9.8; p = 0.03) were associated with LRR. Patients with TN disease had a higher LRR rate than patients with HER2+ or HR+ disease (20 vs. 6 and 4 %; p = 0.005). Among patients without a pCR, TN subtype was associated with increased LRR risk (26 versus 7 % HER+ and 4 % HR+; p < 0.001).
Conclusions
Patients with TN breast cancer had the highest LRR rate after NAC, mastectomy and PMRT. Whereas no LRR was observed among TN patients with a pCR, TN patients with residual disease had a significantly higher LRR risk. Patients with HR+ and HER2+ breast cancer had favorable LRR rates regardless of NAC response, likely due to receipt of adjuvant systemic targeted therapies.

Similar content being viewed by others
References
Overgaard M, Hansen PS, Overgaard J, et al. Postoperative radiotherapy in high-risk premenopausal women with breast cancer who receive adjuvant chemotherapy. N Engl J Med. 1997;337:949–55.
Overgaard M, Jensen MB, Overgaard J, et al. Postoperative radiotherapy in high-risk postmenopausal breast cancer patients given adjuvant tamoxifen: Danish Breast Cancer Cooperative Group DBCG 82c randomised trial. Lancet. 1999;353:1641–8.
Ragaz J, Olivotto IA, Spinelli JJ, et al. Locoregional radiation therapy in patients with high-risk breast cancer receiving adjuvant chemotherapy: 20-year results of the British Columbia randomized trial. J Natl Cancer Inst. 2005;97:116–26.
EBCTCG, McGale P, Taylor C, et al. Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet. 2014;383:2127–35.
Liedtke C, Mazouni C, Hess KR, et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26:1275–81.
von Minckwitz G, Untch M, Blohmer JU, et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 2012;30:1796–804.
Bonnefoi H, Litiere S, Piccart M, et al. Pathological complete response after neoadjuvant chemotherapy is an independent predictive factor irrespective of simplified breast cancer intrinsic subtypes: a landmark and two-step approach analyses from the EORTC 10994/BIG 1-00 phase III trial. Ann Oncol. 2014;25:1128–36.
Symmans WF, Peintinger F, Hatzis C, et al. Measurement of residual breast cancer burden to predict survival after neoadjuvant chemotherapy. J Clin Oncol. 2007;25:4414–22.
Mamounas EP, Anderson SJ, Dignam JJ, et al. Predictors of locoregional recurrence after neoadjuvant chemotherapy: results from combined analysis of National Surgical Adjuvant Breast and Bowel Project B-18 and B-27. J Clin Oncol. 2012;30:3960–6.
Meyers MO, Klauber-DeMore N, Ollila DW, et al. Impact of breast cancer molecular subtypes on locoregional recurrence in patients treated with neoadjuvant chemotherapy for locally advanced breast cancer. Ann Surg Oncol. 2011;18:2851–7.
Nagar H, Mittendorf EA, Strom EA, et al. Local-regional recurrence with and without radiation therapy after neoadjuvant chemotherapy and mastectomy for clinically staged T3N0 breast cancer. Int J Radiat Oncol. 2011;81:782–7.
Sorlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA. 2001;98:10869–74.
Parker JS, Mullins M, Cheang MC, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. 2009;27:1160–7.
Mamounas EP, Cortazar P, Zhang L, Von Minckwitz G, Mehta K. Locoregional recurrence (LRR) after neoadjuvant chemotherapy (NAC): pooled-analysis results from the collaborative trials in neoadjuvant breast cancer (CTNeoBC). J Clin Oncol. 2014;32(Suppl 26). Abstract 61
Huang EH, Tucker SL, Strom EA, et al. Predictors of locoregional recurrence in patients with locally advanced breast cancer treated with neoadjuvant chemotherapy, mastectomy, and radiotherapy. Int J Radiat Oncol Biol Phys. 2005;62:351–7.
Wright JL, Takita C, Reis IM, et al. Predictors of locoregional outcome in patients receiving neoadjuvant therapy and postmastectomy radiation. Cancer. 2013;119:16–25.
Caudle AS, Yu TK, Tucker SL, et al. Local-regional control according to surrogate markers of breast cancer subtypes and response to neoadjuvant chemotherapy in breast cancer patients undergoing breast conserving therapy. Breast Cancer Res. 2012;14:R83.
Comparison of axillary lymph node dissection with axillary radiation for patients with node-positive breast cancer treated with chemotherapy. NCT01901094. https://clinicaltrials.gov/ct2/show/record/NCT01901094. Accessed 6 Nov 2014.
Disclosure
There are no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, T.J., Morrow, M., Modi, S. et al. The Effect of Molecular Subtype and Residual Disease on Locoregional Recurrence in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy and Postmastectomy Radiation. Ann Surg Oncol 22 (Suppl 3), 495–501 (2015). https://doi.org/10.1245/s10434-015-4697-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4697-7