Abstract
Purpose
To explore patterns of failure and postrelapse outcome of patients with retroperitoneal sarcoma primarily treated by extended resection.
Methods
All consecutive patients with primary retroperitoneal sarcoma, treated between January 2002 and December 2011 at two European sarcoma centers were included. Five-year overall survival (OS) and crude cumulative incidence (CCI) of local recurrence (LR) and distant metastases (DM) were calculated. Multivariate analyses for OS and CCI of LR and DM were performed. Postrelapse OS was investigated.
Results
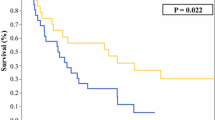
A total of 377 patients were identified. Median follow-up from the time of primary surgery was 44 months [interquartile range (IQR) 27–82]. Five-year OS was 64 % [95 % confidence interval (CI) 0.588, 0710]. CCI of LR and DM were 23.6 % (95 % CI 18.9, 29.4) and 21.9 % (95 % CI 17.6, 27.3), respectively. OS, CCI of LR and DM were 87, 18 % and 0 for well-differentiated liposarcoma; 54, 44 and 9 % for grade II dedifferentiated liposarcoma; 41,33, and 44 % for grade III dedifferentiated liposarcoma; 58, 5, and 55 % for leiomyosarcoma; and 85, 4, and 17 % for solitary fibrous tumor, respectively. Seventy-six patients developed LR. Median postrelapse follow-up was 27 months (IQR 10–58). Twenty-one patients (27 %) underwent a second surgical resection (complete in 18), while 55 (73 %) did not (22 multifocal, 17 inoperable, 16 other causes). Median postrelapse OS was 17 months (IQR 7–31). Well-differentiated liposarcoma histology and disease-free interval predicted postrelapse OS, while surgical resection did not.
Conclusions
When primary extended surgery limits LR, histologic subtype and grade determine the outcome. At recurrence, a second surgery is of limited benefit.


Similar content being viewed by others
References
Singer S, Maki RG, O’Sullivan B. Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA. Cancer: principles and practice of oncology. 9th ed. Philadelphia: Lippincott Williams & Wilkins; 2011. p. 1533–77.
Lewis JJ, Leung D, Woodruff JM. Retroperitoneal soft-tissue sarcoma: analysis of 500 patients treated and followed at a single institution. Ann Surg. 1998;228:355–65.
Stoeckle E, Coindre JM, Bonvalot S, et al. Prognostic factors in retroperitoneal sarcoma: a multivariate analysis of a series of 165 patients of the French Cancer Center Federation Sarcoma Group. Cancer. 2001;92:359–68.
Gronchi A, Casali PG, Fiore M, et al. Retroperitoneal soft tissue sarcomas: patterns of recurrence in 167 patients treated at a single institution. Cancer. 2004;100:2448–55.
Hassan I, Park SZ, Donohue JH, et al. Operative management of primary retroperitoneal sarcomas: a reappraisal of an institutional experience. Ann Surg. 2004;239:244–50.
van Dalen T, Plooij JM, van Coevorden F, et al. Long-term prognosis of primary retroperitoneal soft tissue sarcoma. Eur J Surg Oncol. 2007;33:234–8.
Lehnert T, Cardona S, Hinz U, et al. Primary and locally recurrent retroperitoneal soft-tissue sarcoma: local control and survival. Eur J Surg Oncol. 2009;35:986–93.
Anaya DA, Lahat G, Liu J, et al. Multifocality in retroperitoneal sarcoma: a prognostic factor critical to surgical decision-making. Ann Surg. 2009;249:137–42.
Strauss DC, Hayes AJ, Thway K, et al. Surgical management of primary retroperitoneal sarcoma. Br J Surg. 2010;101:520–3.
Bonvalot S, Rivoire M, Castaing M, et al. Primary retroperitoneal sarcomas: a multivariate analysis of surgical factors associated with local control. J Clin Oncol. 2009;27:31–7.
Gronchi A, Lo Vullo S, Fiore M, et al. Aggressive surgical policies in a retrospectively reviewed single-institution case series of retroperitoneal soft tissue sarcoma patients. J Clin Oncol. 2009;27:24–30.
Bonvalot S, Miceli R, Berselli M., et al. Aggressive surgery in retroperitoneal soft tissue sarcoma carried out at high-volume centers is safe and is associated with improved local control. Ann Surg Oncol. 2010;17:1507–14.
Gronchi A, Miceli R, Colombo C, et al. Frontline extended surgery is associated with improved survival in retroperitoneal low-intermediate grade soft tissue sarcomas. Ann Oncol. 2012;23:1067–73.
Pisters PW. Resection of some—but not all—clinically uninvolved adjacent viscera as part of surgery for retroperitoneal soft tissue sarcomas. J Clin Oncol. 2009;27:6–8.
Gronchi A, Pollock RE. Surgery in retroperitoneal soft tissue sarcoma: a call for a consensus between Europe and North America. Ann Surg Oncol. 2011;18:2107–10.
Gronchi A, Pollock RE. Quality of local treatment or biology of the tumor: which are the trump cards for loco-regional control of retroperitoneal sarcoma? Ann Surg Oncol. 2013;20:2111–3.
Bonvalot S, Raut CP, Pollock RE, et al. Technical considerations in surgery for retroperitoneal sarcomas: position paper from E-Surge, a master class in sarcoma surgery, and EORTC-STBSG. Ann Surg Oncol. 2012;19:2981–91.
Trojani M, Contesso G, Coindre JM, et al. Soft-tissue sarcomas of adults;study of pathological prognostic variables and definition of a histopathological grading system. Int J Cancer. 1984;33:37–42.
Marubini E, Valsecchi MG. Analysing survival data for clinical trials and observational studies. Chichester, UK: Wiley; 1995.
Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988;16:1141–54.
Durrleman S, Simon R. Flexible regression models with cubic splines. Stat Med. 1989;8:551–61.
Schoenfeld D. Partial residuals for the proportional hazards regression model. Biometrika. 1982;69:239–41.
Institute for Statistics and Mathematics of WU (Wirtschaftsuniversität Wien). The R Project for Statistical Computing. Available at: http://www.r-project.org/.
Gronchi A, Miceli R, Shurell E, et al. Outcome prediction in primary resected retroperitoneal soft tissue sarcoma: histology-specific overall survival and disease-free survival nomograms built on major sarcoma center datasets. J Clin Oncol. 2013;31:1649–55.
Dalal KM, Kattan MW, Antonescu CR, et al. Subtype specific prognostic nomogram for patients with primary liposarcoma of the retroperitoneum, extremity, or trunk. Ann Surg. 2006;244:381–91.
Tseng W, Madewell J, Wei W, et al. Locoregional disease patterns in well differentiated and dedifferentiated retroperitoneal liposarcoma: implications for the extent of resection? Ann Surg Oncol. In press.
Park JO, Qin LX, Prete FP, et al. Predicting outcome by growth rate of locally recurrent retroperitoneal liposarcoma: the one centimeter per month rule. Ann Surg. 2009;250:977–82.
Collini P, Negri T, Barisella M, etal. High-grade sarcomatous overgrowth in solitary fibrous tumors: a clinical-pathological study of 10 cases. Am J Surg Pathol. 2012;36:1202–15.
Baldi G, Stacchiotti S, Mauro V, et al. Solitary fibrous tumor of all sites: outcome of late recurrences in 14 patients. Clin Sarcoma Res. 2013;3:4.
ClinicalTrials.gov. Available at: http://clinicaltrials.gov/ct2/show/NCT01344018.
Mussi C, Colombo P, Bertuzzi A, et al. Retroperitoneal sarcoma: is it time to change the surgical policy? Ann Surg Oncol. 2011;18:2136–42.
Conflict of Interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gronchi, A., Miceli, R., Allard, M.A. et al. Personalizing the Approach to Retroperitoneal Soft Tissue Sarcoma: Histology-specific Patterns of Failure and Postrelapse Outcome after Primary Extended Resection. Ann Surg Oncol 22, 1447–1454 (2015). https://doi.org/10.1245/s10434-014-4130-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-4130-7