Abstract
Background
Retrospective studies have shown that primary tumor resection improves the prognosis of patients with colorectal cancer with unresectable metastasis (mCRC). Prognostic significance of lymph node dissection (LND) in mCRC has not been examined previously. The aim of this study was to investigate the prognostic impact of primary tumor resection and LND in mCRC.
Methods
A total of 1,982 patients with mCRC from January 1997 to December 2007 were retrospectively studied. The impact of primary tumor resection and LND on overall survival (OS) was analyzed using Cox proportional hazards model and propensity score analysis to mitigate the selection bias. Covariates in the models for propensity scores included treatment period, institution, age, sex, carcinoembryonic antigen, tumor location, histology, depth, lymph node metastasis, lymphovascular invasion, and number of metastatic organs.
Results
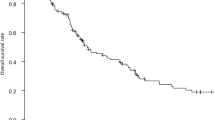
In a multivariate analysis, primary tumor resection and treatment in the latter period were associated with an improved OS, and age over 70 years, female sex, lymph node metastasis, and multiple organ metastasis were associated with a decreased OS. In the propensity-matched cohort, patients treated with primary tumor resection showed a significantly better OS than those without tumor resection (median OS 13.8 vs. 6.3 months; p = 0.0001). Furthermore, among patients treated with primary tumor resection, patients treated with D3 LND showed a significantly better OS than those with less extensive LND (median OS 17.2 vs. 13.7 months; p < 0.0001).
Conclusions
It was suggested that primary tumor resection with D3 LND improves the survival of patients with mCRC.
Similar content being viewed by others
References
Nitzkorski JR, Farma JM, Watson JC, et al. Outcome and natural history of patients with stage IV colorectal cancer receiving chemotherapy without primary tumor resection. Ann Surg Oncol. 2012;19(2):379–383.
Watanabe T, Itabashi M, Shimada Y, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Int J Clin Oncol. 2012;17(1):1–29.
Ruo L, Gougoutas C, Paty PB, Guillem JG, Cohen AM, Wong WD. Elective bowel resection for incurable stage IV colorectal cancer: prognostic variables for asymptomatic patients. J Am Coll Surg. 2003;196(5):722–728.
Verhoef C, de Wilt JH, Burger JW, Verheul HM, Koopman M. Surgery of the primary in stage IV colorectal cancer with unresectable metastases. Eur J Cancer. 2011;47 Suppl 3:S61–66.
Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation–technical notes and outcome. Colorectal Dis. 2009;11(4):354–364; discussion 364–355.
West NP, Hohenberger W, Weber K, Perrakis A, Finan PJ, Quirke P. Complete mesocolic excision with central vascular ligation produces an oncologically superior specimen compared with standard surgery for carcinoma of the colon. J Clin Oncol. 2010;28(2):272–278.
Kanehara S. General rules for clinical and pathological studies on cancer of the colon, rectum and anus. 7th ed. Kanehara & co:Tokyo; 2006.
Stelzner S, Hellmich G, Koch R, Ludwig K. Factors predicting survival in stage IV colorectal carcinoma patients after palliative treatment: a multivariate analysis. J Surg Oncol. 2005;89(4):211–217.
Grothey A, Sugrue MM, Purdie DM, et al. Bevacizumab beyond first progression is associated with prolonged overall survival in metastatic colorectal cancer: results from a large observational cohort study (BRiTE). J Clin Oncol. 2008;26(33):5326–5334.
Tournigand C, Andre T, Achille E, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22(2):229–237.
Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350(23):2335–2342.
Lee WS, Baek JH, Kang JM, Choi S, Kwon KA. The outcome after stent placement or surgery as the initial treatment for obstructive primary tumor in patients with stage IV colon cancer. Am J Surg. 2012;203(6):715–719.
Sabbagh C, Browet F, Diouf M, et al. Is stenting as “a bridge to surgery” an oncologically safe strategy for the management of acute, left-sided, malignant, colonic obstruction? A comparative study with a propensity score analysis. Ann Surg. 2013;258(1):107–115.
Venderbosch S, de Wilt JH, Teerenstra S, et al. Prognostic value of resection of primary tumor in patients with stage IV colorectal cancer: retrospective analysis of two randomized studies and a review of the literature. Ann Surg Oncol. 2011;18(12):3252–3260.
Slanetz CA Jr, Grimson R. Effect of high and intermediate ligation on survival and recurrence rates following curative resection of colorectal cancer. Dis Colon Rectum. 1997;40(10):1205–1218;discussion 1218–1209.
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;230(3):309–318;discussion 318–321.
Battaglia A, Buzzonetti A, Baranello C, et al. Metastatic tumour cells favour the generation of a tolerogenic milieu in tumour draining lymph node in patients with early cervical cancer. Cancer Immunol Immunother. 2009;58(9):1363–1373.
Deng L, Zhang H, Luan Y, et al. Accumulation of foxp3+T regulatory cells in draining lymph nodes correlates with disease progression and immune suppression in colorectal cancer patients. Clin Cancer Res. 2010;16(16):4105–4112.
Boissonnas A, Scholer-Dahirel A, Simon-Blancal V, et al. Foxp3+T cells induce perforin-dependent dendritic cell death in tumor-draining lymph nodes. Immunity. 2010;32(2):266–278.
Stillwell AP, Buettner PG, Ho YH. Meta-analysis of survival of patients with stage IV colorectal cancer managed with surgical resection versus chemotherapy alone. World J Surg. 2010;34(4):797–807.
Little RJ, Rubin DB. Causal effects in clinical and epidemiological studies via potential outcomes: concepts and analytical approaches. Ann Rev Public Health. 2000;21:121–145.
Ahmed S, Howel D, Debrah S. The influence of age on the outcome of treatment of elderly patients with colorectal cancer. J Geriatr Oncol. 2014;5(2):133–140.
van Leersum NJ, Janssen-Heijnen ML, Wouters MW, et al. Increasing prevalence of comorbidity in patients with colorectal cancer in the South of the Netherlands 1995-2010. Int J Cancer. 2013;132(9):2157–2163.
Acknowledgment
The authors completed this study in collaboration with the following: K. Hirata (Sapproro Medical University), A. Murata (Hirosaki University), K. Hatakeyama (Niigata University), K. Hase (National Defense Medical College), K. Kotake (Tochighi Cancer Center), T. Watanabe (University of Tokyo), T. Masaki (Kyorin University), S. Kameoka (Tokyo Women’s Medical University), H. Hasegawa (Keio University), K. Takahashi (Tokyo Metropolitan Cancer and Infectious Disease Center), H. Yano (International Medical Center of Japan), K. Sugihara (Tokyo Medical and Dental University), Y. Hashiguchi (Teikyo University), K. Maeda (Fujita Health University), K. Komori (Aichi Cancer Center), Y. Sakai (Kyoto University), and K. Shirouzu (Kurume University).
Conflicts of Interest and Sources of Funding
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishihara, S., Hayama, T., Yamada, H. et al. Prognostic Impact of Primary Tumor Resection and Lymph Node Dissection in Stage IV Colorectal Cancer with Unresectable Metastasis: A Propensity Score Analysis in a Multicenter Retrospective Study. Ann Surg Oncol 21, 2949–2955 (2014). https://doi.org/10.1245/s10434-014-3719-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3719-1