Abstract
Purpose
The aims of this study were to assess the risk of early recurrence after liver resection for colorectal metastases (CRLM) and its prognostic value; identify early recurrence predictive factors; clarify the effect of perioperative chemotherapy on its occurrence; and elucidate the best early recurrence management.
Methods
Patients of the LiverMetSurvey registry who underwent complete liver resection (R0/R1) between 1998 and 2009 were reviewed. Early recurrence was defined as any recurrence that occurred within 6 months after resection.
Results
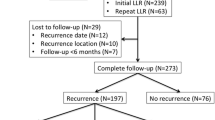
A total of 6,025 patients were included; 2,734 (45.4 %) had recurrence, including 639 (10.6 %) early recurrences. Early recurrence was mainly hepatic (59.5 vs. 54.4 % for late recurrences; p = 0.023). Independent risk factors of early recurrence were: T3–4 primary tumor (p = 0.0002); synchronous CRLM (p = 0.0001); >3 CRLM (p < 0.0001); 0-mm margin liver resection (p = 0.003); and associated intraoperative radiofrequency ablation (p = 0.0005). Response to preoperative chemotherapy (complete/partial) and administration of adjuvant chemotherapy reduced early recurrence risk (p = 0.003 and p < 0.0001, respectively). Intraoperative ultrasonography reduced hepatic early recurrence rate (p = 0.025). Early recurrence negatively affected prognosis: 5-year survival 26.9 versus 49.4 % for the late recurrence group (p < 0.0001, median follow-up 34.4 months). Overall, 234 (36.6 %) patients with early recurrence underwent re-resection. These patients had survival rates higher than non-re-resected patients (5-year survival 47.2 vs. 8.9 %; p < 0.0001) and similar to re-resected patients for late recurrence (48.7 %). Chemotherapy before early recurrence resection improved later survival (5-year survival 61.5 vs. 43.7 %; p = 0.028).
Conclusions
Early recurrence risk is enhanced for extensive disease after poor preoperative disease control and inadequate surgical treatment, but is reduced after adjuvant chemotherapy. Although early recurrence negatively affects prognosis, re-resection may restore better survival. Chemotherapy before early recurrence resection is advocated.



Similar content being viewed by others
References
Adam R, De Gramont A, Figueras J, et al. The oncosurgery approach to managing liver metastases from colorectal cancer: a multidisciplinary international consensus. Oncologist. 2012;17(10):1225–1239.
Kopetz S, Chang GJ, Overman MJ, et al. Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol. 2009;27(22):3677–3683.
Wieser M, Sauerland S, Arnold D, Schmiegel W, Reinacher-Schick A. Peri-operative chemotherapy for the treatment of resectable liver metastases from colorectal cancer: a systematic review and meta-analysis of randomized trials. BMC Cancer. 2010;10:309.
House MG, Ito H, Gönen M, et al. Survival after hepatic resection for metastatic colorectal cancer: trends in outcomes for 1,600 patients during two decades at a single institution. J Am Coll Surg. 2010;210(5):744–752.
Viganò L, Russolillo N, Ferrero A, Langella S, Sperti E, Capussotti L. Evolution of long-term outcome of liver resection for colorectal metastases: analysis of actual 5-year survival rates over two decades. Ann Surg Oncol. 2012;19(6):2035–2044.
de Jong MC, Pulitano C, Ribero D, et al. Rates and patterns of recurrence following curative intent surgery for colorectal liver metastasis: an international multi-institutional analysis of 1669 patients. Ann Surg. 2009;250(3):440–448.
Tanaka K, Shimada H, Ueda M, Matsuo K, Endo I, Togo S. Long-term characteristics of 5-year survivors after liver resection for colorectal metastases. Ann Surg Oncol. 2007;14(4):1336–1346.
Tomlinson JS, Jarnagin WR, DeMatteo RP, et al. Actual 10-year survival after resection of colorectal liver metastases defines cure. J Clin Oncol. 2007;25(29):4575–4580.
Viganò L, Ferrero A, Lo Tesoriere R, Capussotti L. Liver surgery for colorectal metastases: results after 10 years of follow-up. Long-term survivors, late recurrences, and prognostic role of morbidity. Ann Surg Oncol. 2008;15(9):2458–2464.
Adam R, Pascal G, Azoulay D, Tanaka K, Castaing D, Bismuth H. Liver resection for colorectal metastases: the third hepatectomy. Ann Surg. 2003;238(6):871–883.
Petrowsky H, Gonen M, Jarnagin W, et al. Second liver resections are safe and effective treatment for recurrent hepatic metastases from colorectal cancer: a bi-institutional analysis. Ann Surg. 2002;235(6):863–871.
Imamura H, Kawasaki S, Miyagawa S, Ikegami T, Kitamura H, Shimada R. Aggressive surgical approach to recurrent tumors after hepatectomy for metastatic spread of colorectal cancer to the liver. Surgery. 2000;127(5):528–535.
Malik HZ, Gomez D, Wong V, et al. Predictors of early disease recurrence following hepatic resection for colorectal cancer metastasis. Eur J Surg Oncol. 2007;33(8):1003–1009.
Kaibori M, Iwamoto Y, Ishizaki M, et al. Predictors and outcome of early recurrence after resection of hepatic metastases from colorectal cancer. Langenbecks Arch Surg. 2012; 397(3):373–381.
Viganò L, Capussotti L, Barroso E, et al. Progression while receiving preoperative chemotherapy should not be an absolute contraindication to liver resection for colorectal metastases. Ann Surg Oncol. 2012;19(9):2786–2796.
Takahashi S, Konishi M, Nakagohri T, Gotohda N, Saito N, Kinoshita T. Short time to recurrence after hepatic resection correlates with poor prognosis in colorectal hepatic metastasis. Jpn J Clin Oncol. 2006;36(6):368–375.
Greene FL, American Joint Committee on Cancer. American Cancer Society. AJCC cancer staging manual. 6th ed. New York: Springer; 2002.
World Health Organization. Handbook for reporting results of cancer treatment. WHO offset publication no. 48-. Geneva: World Health Organization; 1979.
Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92(3):205–216.
Nordlinger B, Guiguet M, Vaillant JC, et al. Surgical resection of colorectal carcinoma metastases to the liver. A prognostic scoring system to improve case selection, based on 1568 patients. Cancer. 1996;77:1254–1262.
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;230:309–321.
Ribero D, Viganò L, Amisano M, Capussotti L. Prognostic factors after resection of colorectal liver metastases: from morphology to biology. Future Oncol. 2013;9(1):45–57.
Pawlik TM, Scoggins CR, Zorzi D, et al. Effect of surgical margin status on survival and site of recurrence after hepatic resection for colorectal metastases. Ann Surg. 2005;241(5):715–722.
Are C, Gonen M, Zazzali K, et al. The impact of margins on outcome after hepatic resection for colorectal metastases. Ann Surg. 2007;246(2):295–300.
Kokudo N, Miki Y, Sugai S, et al. Genetic and histological assessment of surgical margins in resected liver metastases from colorectal carcinoma: minimum surgical margins for successful resection. Arch Surg. 2002;137(7):833–840.
Nuzzo G, Giuliante F, Ardito F, et al. Influence of surgical margin on type of recurrence after liver resection for colorectal metastases: a single-center experience. Surgery. 2008;143(3):384–393.
de Haas RJ, Wicherts DA, Flores E, Azoulay D, Castaing D, Adam R. R1 resection by necessity for colorectal liver metastases: is it still a contraindication to surgery? Ann Surg. 2008;248(4):626–637.
Mentha G, Terraz S, Morel P, et al. Dangerous halo after neoadjuvant chemotherapy and two-step hepatectomy for colorectal liver metastases. Br J Surg. 2009;96(1):95–103.
Viganò L, Capussotti L, De Rosa G, Oulhaci De Saussure W, Mentha G, Rubbia-Brandt L. Liver resection for colorectal metastases after chemotherapy: impact of chemotherapy-related liver injuries, pathological tumor response and micrometastases on long-term survival. Ann Surg. 2013;258(5):731–740.
Cirocchi R, Trastulli S, Boselli C, et al. Radiofrequency ablation in the treatment of liver metastases from colorectal cancer. Cochrane Database Syst Rev. 2012;6:CD006317.
Wong SL, Mangu PB, Choti MA, et al. American Society of Clinical Oncology 2009 clinical evidence review on radiofrequency ablation of hepatic metastases from colorectal cancer. J Clin Oncol. 2010;28(3):493–508.
Abdalla EK, Vauthey JN, Ellis LM, et al. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg. 2004;239(6):818–825.
van Vledder MG, Pawlik TM, Munireddy S, Hamper U, de Jong MC, Choti MA. Factors determining the sensitivity of intraoperative ultrasonography in detecting colorectal liver metastases in the modern era. Ann Surg Oncol. 2010;17(10):2756–2763.
Torzilli G, Botea F, Procopio F, et al. Use of contrast-enhanced intraoperative ultrasonography during liver surgery for colorectal cancer liver metastases: its impact on operative outcome. Analysis of a prospective cohort study. Eur J Cancer Suppl. 2008;6(11):16–23.
Sietses C, Meijerink MR, Meijer S, van den Tol MP. The impact of intraoperative ultrasonography on the surgical treatment of patients with colorectal liver metastases. Surg Endosc. 2010; 24(8):1917–1922.
Viganò L, Ferrero A, Amisano M, Russolillo N, Capussotti L. Comparison of laparoscopic and open intraoperative ultrasonography for staging liver tumours. Br J Surg. 2013;100(4):535–542.
Nordlinger B, Sorbye H, Glimelius B, et al. Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet. 2008;371(9617):1007–1016.
Adam R, Pascal G, Castaing D, et al. Tumor progression while on chemotherapy: a contraindication to liver resection for multiple colorectal metastases? Ann Surg. 2004;240(6):1052–1061.
Portier G, Elias D, Bouche O, et al. Multicenter randomized trial of adjuvant fluorouracil and folinic acid compared with surgery alone after resection of colorectal liver metastases: FFCD ACHBTH AURC 9002 trial. J Clin Oncol. 2006;24:4976–4982.
Mitry E, Fields A, Bleiberg H, et al. Adjuvant chemotherapy after potentially curative resection of metastases from colorectal cancer: a meta-analysis of two randomized trials. J Clin Oncol. 2008; 26:4906–4911.
Disclosure
The authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Viganò, L., Capussotti, L., Lapointe, R. et al. Early Recurrence After Liver Resection for Colorectal Metastases: Risk Factors, Prognosis, and Treatment. A LiverMetSurvey-Based Study of 6,025 Patients. Ann Surg Oncol 21, 1276–1286 (2014). https://doi.org/10.1245/s10434-013-3421-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-3421-8