Abstract
Background
Eighty percent of patients with resected pancreatic ductal carcinoma (PDC) experience treatment failure within 2 years. We hypothesized that preoperative fixed-dose rate (FDR) gemcitabine (GEM) combined with the angiogenesis inhibitor bevacizumab (BEV) and accelerated 30 Gy radiotherapy (RT) would improve outcomes among patients with potentially resectable PDC.
Methods
This phase II trial tested induction FDR GEM (1,500 mg/m2) plus BEV (10 mg/kg IV) every 2 weeks for three cycles followed by accelerated RT (30 Gy in 10 fractions) plus BEV directed at gross tumor volume plus a 1–2 cm vascular margin. Subjects underwent laparoscopy and resection after day 85. Therapy was considered effective if the complete pathologic response rate exceeded 10 % and the margin-negative resection rate exceeded 80 %.
Results
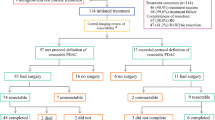
Fifty-nine subjects were enrolled; 29 had potential portal vein involvement. Two grade 4 (3.4 %) and 19 grade 3 toxicities (32.8 %) occurred. Four subjects manifested radiographic progression, and 10 had undetected carcinomatosis. Forty-three pancreatic resections (73 %) were performed, including 19 portal vein resections (44 %). Margin-negative outcomes were observed in 38 (88 %, 95 % confidence interval [CI] 75–96), with one complete pathologic response (2.3 %; 95 % CI 0.1–12). There were seven (6 grade 3; 1 grade 4) wound complications (13 %). Median overall survival for the entire cohort was 16.8 months (95 % CI 14.9–21.3) and 19.7 months (95 % CI 16.5–28.2) after resection.
Conclusions
Induction therapy with FDR GEM and BEV, followed by accelerated BEV/RT to 30 Gy, was well tolerated. Although both effectiveness criteria were achieved, survival outcomes were equivalent to published regimens.


Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300.
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29.
Haeno H, Gonen M, Davis MB, Herman JM, Iacobuzio-Donahue CA, Michor F. Computational modeling of pancreatic cancer reveals kinetics of metastasis suggesting optimum treatment strategies. Cell. 2012;148:362–75.
Stokes JB, Nolan NJ, Stelow EB, et al. Preoperative capecitabine and concurrent radiation for borderline resectable pancreatic cancer. Ann Surg Oncol. 2011;18:619–27.
Quiros RM, Brown KM, Hoffman JP. Neoadjuvant therapy in pancreatic cancer. Cancer Invest. 2007;25:267–73.
Blackstock AW, Bernard SA, Richards F, et al. Phase I trial of twice-weekly gemcitabine and concurrent radiation in patients with advanced pancreatic cancer. J Clin Oncol. 1999;17:2208–12.
Talamonti MS, Catalano PJ, Vaughn DJ, et al. Eastern Cooperative Oncology Group phase I trial of protracted venous infusion fluorouracil plus weekly gemcitabine with concurrent radiation therapy in patients with locally advanced pancreas cancer: a regimen with unexpected early toxicity. J Clin Oncol. 2000;18:3384–9.
Wolff RA, Evans DB, Gravel DM, et al. Phase I trial of gemcitabine combined with radiation for the treatment of locally advanced pancreatic adenocarcinoma. Clin Cancer Res. 2001;7:2246–53.
Evans DB, Rich TA, Byrd DR, et al. Preoperative chemoradiation and pancreaticoduodenectomy for adenocarcinoma of the pancreas. Arch Surg. 1992;127:1335–9.
Crane C, Janjan N, Evans D, et al. Toxicity and efficacy of concurrent gemcitabine and radiotherapy for locally advanced pancreatic cancer. Ing J Gastrointest Cancer. 2001;29:9–18.
Wolff RA. Neoadjuvant chemoradiation for localized adenocarcinoma of the pancreas: great logic, grim reality. Ann Surg Oncol. 2001;8:747–8.
Kindler HL, Friberg G, Singh DA, et al. Phase II trial of bevacizumab plus gemcitabine in patients with advanced pancreatic cancer. J Clin Oncol. 2005;23:8033–40.
Kindler HL, Niedzwiecki D, Hollis D, et al. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J Clin Oncol. 2010;28:3617–22.
Crane CH, Ellis LM, Abbruzzese JL, et al. Phase I trial evaluating the safety of bevacizumab with concurrent radiotherapy and capecitabine in locally advanced pancreatic cancer. J Clin Oncol. 2006;24:1145–51.
Pisters PW, Evans DB, Leung DH, Brennan MF. Re: Surgery for ductal adenocarcinoma of the pancreatic head. World J Surg. 2001;25:533–4.
Callery MP, Chang KJ, Fishman EK, Talamonti MS, William Traverso L, Linehan DC. Pretreatment assessment of resectable and borderline resectable pancreatic cancer: expert consensus statement. Ann Surg Oncol. 2009;16:1727–33.
Pisters PW, Lee JE, Vauthey JN, Charnsangavej C, Evans DB. Laparoscopy in the staging of pancreatic cancer. Br J Surg. 2001;88:325–37.
Zeh HJ, Zureikat AH, Secrest A, Dauoudi M, Bartlett D, Moser AJ. Outcomes after robot-assisted pancreaticoduodenectomy for periampullary lesions. Ann Surg Oncol. 2012;19:864–70.
Zeh HJ 3rd, Bartlett DL, Moser AJ. Robotic-assisted major pancreatic resection. Adv Surg. 2011;45:323–40.
Magge D, Gooding W, Choudry H, et al. Comparative effectiveness of minimally invasive and open distal pancreatectomy for ductal adenocarcinoma. JAMA Surg. 2013;148:525–31.
Daouadi M, Zureikat AH, Zenati MS, et al. Robot-assisted minimally invasive distal pancreatectomy is superior to the laparoscopic technique. Ann Surg. 2013;257:128–32.
Zureikat AH, Nguyen KT, Bartlett DL, Zeh HJ, Moser AJ. Robotic-assisted major pancreatic resection and reconstruction. Arch Surg. 2011;146:256–61.
Ajani JA, Mansfield PF, Janjan N, et al. Multi-institutional trial of preoperative chemoradiotherapy in patients with potentially resectable gastric carcinoma. J Clin Oncol. 2004;22:2774–80.
Chirieac LR, Swisher SG, Ajani JA, et al. Posttherapy pathologic stage predicts survival in patients with esophageal carcinoma receiving preoperative chemoradiation. Cancer. 2005;103:1347–55.
Pisters PW, Abbruzzese JL, Janjan NA, et al. Rapid-fractionation preoperative chemoradiation, pancreaticoduodenectomy, and intraoperative radiation therapy for resectable pancreatic adenocarcinoma. J Clin Oncol. 1998;16:3843–50.
Evans DB, Pisters PW, Lee JE, et al. Preoperative chemoradiation strategies for localized adenocarcinoma of the pancreas. J Hepatobiliary Pancreat Surg. 1998;5:242–50.
Miller AR, Robinson EK, Lee JE, et al. Neoadjuvant chemoradiation for adenocarcinoma of the pancreas. Surg Oncol Clin North Am. 1998;7:183–97.
Breslin TM, Hess KR, Harbison DB, et al. Neoadjuvant chemoradiotherapy for adenocarcinoma of the pancreas: treatment variables and survival duration. Ann Surg Oncol. 2001;8:123–32.
Loehrer PJ Sr, Feng Y, Cardenes H, et al. Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol. 2011;29:4105–12.
Evans DB, Varadhachary GR, Crane CH, et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496–502.
Talamonti MS, Small W Jr, Mulcahy MF, et al. A multi-institutional phase II trial of preoperative full-dose gemcitabine and concurrent radiation for patients with potentially resectable pancreatic carcinoma. Ann Surg Oncol. 2006;13:150–8.
Small W Jr, Mulcahy MF, Rademaker A, et al. Phase II trial of full-dose gemcitabine and bevacizumab in combination with attenuated three-dimensional conformal radiotherapy in patients with localized pancreatic cancer. Int J Radiat Oncol Biol Phys. 2011;80:476–82.
Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350:1200–10.
Oettle H, Post S, Neuhaus P, et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA. 2007;297:267–77.
Olive KP, Jacobetz MA, Davidson CJ, et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324(5933):1457–61.
Provenzano PP, Cuevas C, Chang AE, Goel VK, Von Hoff DD, Hingorani SR. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell. 2012;21:418–29.
McGee MC, Hamner JB, Williams RF, et al. Improved intratumoral oxygenation through vascular normalization increases glioma sensitivity to ionizing radiation. Int J Radiat Oncol Biol Phys. 2010;76:1537–45.
Myers AL, Williams RF, Ng CY, Hartwich JE, Davidoff AM. Bevacizumab-induced tumor vessel remodeling in rhabdomyosarcoma xenografts increases the effectiveness of adjuvant ionizing radiation. J Pediatr Surg. 2010;45:1080–5.
Shroff RT, Varadhachary GR, Crane CH, et al. Updated survival analysis of preoperative gemcitabine (gem) plus bevacizumab (bev)-based chemoradiation for resectable pancreatic adenocarcinoma. Paper presented at: ASCO Annual Meeting 2012, Chicago.
Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–42.
Roman CD, Choy H, Nanney L, et al. Vascular endothelial growth factor-mediated angiogenesis inhibition and postoperative wound healing in rats. J Surg Res. 2002;105:43–7.
Giantonio BJ, Levy DE, O’Dwyer P J, Meropol NJ, Catalano PJ, Benson AB 3rd. A phase II study of high-dose bevacizumab in combination with irinotecan, 5-fluorouracil, leucovorin, as initial therapy for advanced colorectal cancer: results from the Eastern Cooperative Oncology Group study E2200. Ann Oncol. 2006;17:1399–403.
Giantonio BJ. Gastrointestinal perforation and cancer therapy: managing risk to achieve benefit. Onkologie. 2005;28:177–8.
Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol. 2005;23:1011–27.
Winter JM, Cameron JL, Campbell KA, et al. 1423 pancreaticoduodenectomies for pancreatic cancer: a single-institution experience. J Gastrointest Surg. 2006;10:1199–210.
Acknowledgment
Supported in part by a research grant from Genentech to AJM, AK, and DMP. Genentech had no control over trial design, data acquisition and analysis, or document preparation. Special thanks to Jennifer Steve at the University of Pittsburgh Department of Surgery for data acquisition and analysis.
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Van Buren, G., Ramanathan, R.K., Krasinskas, A.M. et al. Phase II Study of Induction Fixed-Dose Rate Gemcitabine and Bevacizumab Followed by 30 Gy Radiotherapy as Preoperative Treatment for Potentially Resectable Pancreatic Adenocarcinoma. Ann Surg Oncol 20, 3787–3793 (2013). https://doi.org/10.1245/s10434-013-3161-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-3161-9