Abstract
Background
Infective complications particularly in the form of surgical site infections including anastomotic leak represent a serious morbidity after colorectal cancer surgery. Systemic inflammation markers, including C-reactive protein (CRP) and white cell count, have been reported to provide early detection. However, their relative predictive value is unclear. The aim of the present study was to examine the diagnostic accuracy of serial postoperative WCC, albumin and CRP in detecting infective complications.
Methods
White cell count, albumin and CRP were measured postoperatively for 7 days in 454 consecutive patients undergoing surgery for colorectal cancer. All postoperative complications were recorded. The diagnostic accuracy of the white cell count, albumin and CRP values were analyzed by receiver operating characteristics curve analysis with surgical site infective complications as outcome measures.
Results
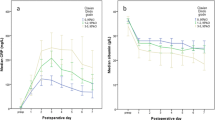
One hundred four patients (23 %) developed infective complications, and 26 of them developed an anastomotic leak. CRP was most sensitive to the development of an infective complication, surgical site or at a remote site. On postoperative day 3 CRP the area under the receiver operating characteristic curve was 0.80 (p < 0.001) and the optimal cutoff value was 170 mg/L. This threshold was also associated with an increase in the length of hospital stay (p < 0.001), 30 day mortality (p < 0.05) and 12 month mortality (p < 0.10).
Conclusions
Postoperative CRP measurement on day 3 postoperatively is clinically useful in predicting surgical site infective complications, including an anastomotic leak, in patients undergoing surgery for colorectal cancer.



Similar content being viewed by others
References
Fujita S, Saito N, Yamada T, et al. Randomized, multicenter trial of antibiotic prophylaxis in elective colorectal surgery: single dose vs. 3 doses of a second-generation cephalosporin without metronidazole and oral antibiotics. Arch Surg. 2007;142:657–61.
Rovera F, Dionigi G, Boni L, et al. Infectious complications in colorectal surgery. Surg Oncol. 2007;16(Suppl. 1):S121–4.
Törnqvist A, Ekelund G, Forsgren A, Leandoer L, Olson S, Ursing J. Single dose doxycycline prophylaxis and peroperative bacteriological culture in elective colorectal surgery. Br J Surg. 1981;68:565–8.
Alves A, Panis Y, Mathieu P, Mantion G, Kwiatkowski F, Slim K. Postoperative mortality and morbidity in French patients undergoingm colorectal surgery: results of a prospective multicenter study. Arch Surg. 2005;140:278–83.
Sjo O, Larsen S, Lunde O, Nesbakken A. Short term outcome after emergency and elective surgery for colon cancer. Colorectal Dis. 2009;11:733–9.
Edwards LD. The epidemiology of 2056 remote site infections and 1966 surgical wound infections occurring in 1865 patients: a four year study of 40,923 operations at Rush–Presbyterian–St. Luke’s Hospital, Chicago. Ann Surg. 1976;184:758–66.
Miki C, Inoue Y, Mohri Y, Kobayashi M, Kusunoki M. Site-specific patterns of surgical site infections and their early indicators after elective colorectal cancer surgery. Dis Colon Rectum. 2006;49:S45–52.
Matsuda A, Matsutani T, Sasajima K, et al. Preoperative plasma adiponectin level is a risk factor for postoperative infection following colorectal cancer surgery. J Surg Res. 2009;157:227–34.
Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR. Guideline for prevention of surgical site infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Am J Infect Control. 1999;27:97–132.
Alves A, Panis Y, Pocard M, Regimbeau JM, Valleur P. Management of anastomotic leakage after nondiverted large bowel resection. J Am Coll Surg. 1999;189:554–9.
Petersen S, Freitag M, Hellmich G, Ludwig K. Anastomotic leakage: impact on local recurrence and survival in surgery of colorectal cancer. Int J Colorectal Dis. 1998;13:160–3.
Buchs NC, Gervaz P, Secic M, Bucher P, Mugnier-Konrad B, Morel P. Incidence, consequences, and risk factors for anastomotic dehiscence after colorectal surgery: a prospective monocentric study. Int J Colorectal Dis. 2008;23:265–70.
Hyman N, Manchester TL, Osler T, Burns B, Cataldo PA. Anastomotic leaks after intestinal anastomosis: it’s later than you think. Ann Surg. 2007;245:254–8.
McArdle CS, McMillan DC, Hole DJ. Impact of anastomotic leakage on long-term survival of patients undergoing curative resection for colorectal cancer. Br J Surg. 2005;92:1150–4.
Jung SH, Yu CS, Choi PW, et al. Risk factors and oncologic impact of anastomotic leakage after rectal cancer surgery. Dis Colon Rectum. 2008;51:902–8.
Marra F, Steffen T, Kalak N, et al. Anastomotic leakage as a risk factor for the long-term outcome after curative resection of colon cancer. Eur J Surg Oncol. 2009;35:1060–4.
Welsch T, Müller SA, Ulrich A, et al. C-reactive protein as early predictor for infectious postoperative complications in rectal surgery. Int J Colorectal Dis. 2007;22:1499–507.
Matthiessen P, Henriksson M, Hallbook O, Grunditz E, Noren B, Arbman G. Increase of serum C-reactive protein is an early indicator of subsequent symptomatic anastomotic leakage after anterior resection. Colorectal Dis. 2008;10:75–80.
Kørner H, Nielsen HJ, Søreide JA, Nedrebø BS, Søreide K, Knapp JC. Diagnostic accuracy of C-reactive protein for intraabdominal infections after colorectal resections. J Gastrointest Surg. 2009;13:1599–606.
Woeste G, Müller C, Bechstein WO, Wullstein C. Increased serum levels of C-reactive protein precede anastomotic leakage in colorectal surgery. World J Surg. 2010;34:140–6.
Ortega-Deballon P, Radais F, Facy O, et al. C-reactive protein is an early predictor of septic complications after elective colorectal surgery. World J Surg. 2010;34:808–14.
Mackay GJ, Molloy RG, O’Dwyer PJ. C-reactive protein as a predictor of postoperative infective complications following elective colorectal resection. Colorectal Dis. 2011;13:583–7.
Warschkow R, Tarantion I, Torzewski M, Naf F, Lange J, Steffen T. Diagnostic accuracy of C-reactive protein and white blood cell counts in the early detection of inflammatory complication after open resection of colorectal cancer: a retrospective study of 1,187 patients. Int J Colorectal Dis. 2011;26:1405–13.
Ytting H, Christensen IJ, Jensenius JC, Thiel S, Nielsen HJ. Preoperative mannan-binding lectin pathway and prognosis in colorectal cancer. Cancer Immunol Immunother. 2005;54:265–72.
Carstairs V, Morris R. Deprivation and health in Scotland. Health Bull (Edinb). 1990;48:162–75.
Hole DJ, McArdle CS. Impact of socioeconomic deprivation on outcome after surgery for colorectal cancer. Br J Surg. 2002;89:586–90.
Robertson EA, Zweig MH. Use of receiver operating characteristic curves to evaluate the clinical performance of analytical systems. Clin Chem. 1981;27:1569–74.
Zweig MH, Campbell G. Receiver–operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem. 1993;39:561–77.
Soreide K. Receiver–operating characteristic curve analysis in diagnostic, prognostic and predictive biomarker research. J Clin Pathol. 2009;62:1–5.
Chromik AM, Endter F, Uhl W, Thiede A, Reith HB, Mittelkötter U. Pre-emptive antibiotic treatment vs. “standard” treatment in patients with elevated serum procalcitonin levels after elective colorectal surgery: a prospective randomised pilot study. Langenbecks Arch Surg. 2006;391:187–94.
Peisajovich A, Marnell L, Mold C, et al. C-reactive protein at the interface between innate immunity and inflammation. Expert Rev Clin Immunol. 2008;4:379–90.
Coventry BJ, Ashdown ML, Quinn MA, et al. CRP identifies homeostatic immune oscillations in cancer patients: a potential treatment targeting tool? J Transl Med. 2009;7:102.
Du Clos TW, Mold C. C-reactive protein: an activator of innate immunity and a modulator of adaptive immunity. Immunol Res. 2004;30:261–77.
Sander LE, Sackett SD, Dierssen U, et al. Hepatic acutephase proteins control innate immune responses during infection by promoting myeloid-derived suppressor cell function. J Exp Med. 2010;207:1453–64.
Fietta AM, Morosini M, Passadore I, et al. Systemic inflammatory response and downmodulation of peripheral CD25 + Foxp3 + T-regulatory cells in patients undergoing radiofrequency thermal ablation for lung cancer. Hum Immunol. 2009;70:477–86.
Wichmann MW, Huttl TP, Winter H, et al. Immunological effects of laparoscopic vs. open colorectal surgery: a prospective clinical study. Arch Surg. 2005;140:692–7.
Motoyama S, Miura M, Hinai Y, et al. C-reactive protein -717C>T genetic polymorphism associates with esophagectomy-induced stress hyperglycemia. World J Surg. 2010;35:1001–7.
Ramos M, Khalpey Z, Lipsitz S, et al. Relationship of perioperative hyperglycemia and postoperative infections in patients who undergo general and vascular surgery. Ann Surg. 2008;248:585–591.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jonathan J. Platt and Michelle L. Ramanathan contributed equally to this study.
Rights and permissions
About this article
Cite this article
Platt, J.J., Ramanathan, M.L., Crosbie, R.A. et al. C-reactive Protein as a Predictor of Postoperative Infective Complications after Curative Resection in Patients with Colorectal Cancer. Ann Surg Oncol 19, 4168–4177 (2012). https://doi.org/10.1245/s10434-012-2498-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2498-9