Abstract
Background
Lung adenocarcinomas represent a morphologically heterogeneous tumor composed of an admixture of different histologic subtypes (lepidic, papillary, acinar, and solid subtype). The presence of a solid subtype component is reported to be associated with a poorer prognosis. The aim of this study was to evaluate the characteristic immunophenotype of the solid subtype component compared with the immunophenotypes of other components.
Methods
We analyzed the clinicopathological characteristics of stage I adenocarcinoma patients with predominant solid subtype disease. Furthermore, we immunostained adenocarcinomas with predominant lepidic, papillary, acinar, and solid subtype components (n = 23 each) for 10 molecular markers of tumor invasiveness and scored the results.
Results
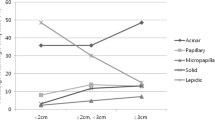
Patients showing predominance of the solid subtype component (solid subtype adenocarcinoma) had a poorer prognosis than those showing predominance of the lepidic, papillary, or acinar component. Lymphovascular invasion was more often detected in solid subtype tumors than in others. The solid subtype component showed a significantly stronger staining intensity of laminin-5 expression than the lepidic, papillary, and acinar components (P < 0.001, P < 0.001, and P = 0.016, respectively). The fibronectin and vimentin expression levels were also significantly higher in the solid subtype component than in other components. This immunostaining character was validated by using mixed-subtype adenocarcinomas containing all four components in the same tumor.
Conclusions
This study concluded that the solid subtype component in lung adenocarcinomas exhibit the invasive immunophenotype, including increased laminin-5 expression, compared with the other components, which may be associated with a poorer prognosis.





Similar content being viewed by others
References
Asamura H, Goya T, Koshiishi Y, et al. A Japanese Lung Cancer Registry study: prognosis of 13,010 resected lung cancers. J Thorac Oncol. 2008;3:46–52.
Travis WD, Brambilla E, Noguchi M, et al. International association for the study of lung cancer/American Thoracic Society/European Respiratory Society: international multidisciplinary classification of lung adenocarcinoma: executive summary. Proc Am Thorac Soc. 2011;8:381–5.
Travis WD, Brambilla E, Noguchi M, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol. 2011;6:244–85.
Travis WD, Brambilla E, Van Schil P, et al. Paradigm shifts in lung cancer as defined in the new IASLC/ATS/ERS lung adenocarcinoma classification. Eur Respir J. 2011;38:239–43.
Travis W, Brambilla E, Muller-Hermelink H, Harris C. World Health Organization classification of tumors. Pathology and genetics of the lung, pleura, thymus and heart. IARC Press; 2004.
Sica G, Yoshizawa A, Sima CS, et al. A grading system of lung adenocarcinomas based on histologic pattern is predictive of disease recurrence in stage I tumors. Am J Surg Pathol. 2010;34:1155–62.
Borczuk AC, Qian F, Kazeros A, et al. Invasive size is an independent predictor of survival in pulmonary adenocarcinoma. Am J Surg Pathol. 2009;33:462–9.
Noguchi M, Morikawa A, Kawasaki M, et al. Small adenocarcinoma of the lung. Histologic characteristics and prognosis. Cancer. 1995;75:2844–52.
Sakurai H, Maeshima A, Watanabe S, et al. Grade of stromal invasion in small adenocarcinoma of the lung: histopathological minimal invasion and prognosis. Am J Surg Pathol. 2004;28:198–206.
Yim J, Zhu LC, Chiriboga L, et al. Histologic features are important prognostic indicators in early stages lung adenocarcinomas. Mod Pathol. 2007;20:233–41.
Barletta JA, Yeap BY, Chirieac LR. Prognostic significance of grading in lung adenocarcinoma. Cancer. 2010;116:659–69.
Ohtaki Y, Yoshida J, Ishii G, et al. Prognostic significance of a solid component in pulmonary adenocarcinoma. Ann Thorac Surg. 2011;91:1051–7.
Petersen I, Kotb WF, Friedrich KH, et al. Core classification of lung cancer: correlating nuclear size and mitoses with ploidy and clinicopathological parameters. Lung Cancer. 2009;65:312–8.
Riquet M, Foucault C, Berna P, et al. Prognostic value of histology in resected lung cancer with emphasis on the relevance of the adenocarcinoma subtyping. Ann Thorac Surg. 2006;81:1988–95.
Al-Saad S, Al-Shibli K, Donnem T, et al. The prognostic impact of NF-kappaB p105, vimentin, E-cadherin and Par6 expression in epithelial and stromal compartment in non-small-cell lung cancer. Br J Cancer. 2008;99:1476–83.
Moriya Y, Niki T, Yamada T, et al. Increased expression of laminin-5 and its prognostic significance in lung adenocarcinomas of small size. An immunohistochemical analysis of 102 cases. Cancer. 2001;91:1129–41.
Sholl LM, Barletta JA, Yeap BY, et al. Sox2 protein expression is an independent poor prognostic indicator in stage I lung adenocarcinoma. Am J Surg Pathol. 2010;34:1193–8.
Motoi N, Szoke J, Riely GJ, et al. Lung adenocarcinoma: modification of the 2004 WHO mixed subtype to include the major histologic subtype suggests correlations between papillary and micropapillary adenocarcinoma subtypes, EGFR mutations and gene expression analysis. Am J Surg Pathol. 2008;32:810–27.
Sobin L, Wittekind C. TNM classification of malignant tumors. New York: Wiley and Sons; 2002.
Tsuta K, Ishii G, Nitadori J, et al. Comparison of the immunophenotypes of signet-ring cell carcinoma, solid adenocarcinoma with mucin production, and mucinous bronchioloalveolar carcinoma of the lung characterized by the presence of cytoplasmic mucin. J Pathol. 2006;209:78–87.
Hsu FD, Nielsen TO, Alkushi A, et al. Tissue microarrays are an effective quality assurance tool for diagnostic immunohistochemistry. Mod Pathol. 2002;15:1374–80.
Bryant CM, Albertus DL, Kim S, et al. Clinically relevant characterization of lung adenocarcinoma subtypes based on cellular pathways: an international validation study. PLoS One. 2010;5:e11712.
Eren B, Sar M, Oz B, Dincbas FH. MMP-2, TIMP-2 and CD44v6 expression in non-small-cell lung carcinomas. Ann Acad Med Singapore. 2008;37:32–9.
Maatta M, Soini Y, Paakko P, et al. Expression of the laminin gamma2 chain in different histological types of lung carcinoma. A study by immunohistochemistry and in situ hybridization. J Pathol. 1999;188:361–8.
Tsutsumida H, Nomoto M, Goto M, et al. A micropapillary pattern is predictive of a poor prognosis in lung adenocarcinoma, and reduced surfactant apoprotein A expression in the micropapillary pattern is an excellent indicator of a poor prognosis. Mod Pathol. 2007;20:638–47.
Yun J, Son CH, Um SJ, et al. A different TRAP220 expression in distinct histologic subtypes of lung adenocarcinoma and the prognostic significance. Lung Cancer. 2011;71:312–8.
Takatsuki H, Komatsu S, Sano R, et al. Adhesion of gastric carcinoma cells to peritoneum mediated by alpha3beta1 integrin (VLA-3). Cancer Res. 2004;64:6065–70.
Koshikawa N, Moriyama K, Takamura H, et al. Overexpression of laminin gamma2 chain monomer in invading gastric carcinoma cells. Cancer Res. 1999;59:5596–601.
Kosmehl H, Berndt A, Strassburger S, et al. Distribution of laminin and fibronectin isoforms in oral mucosa and oral squamous cell carcinoma. Br J Cancer. 1999;81:1071–9.
Pyke C, Romer J, Kallunki P, et al. The gamma 2 chain of kalinin/laminin 5 is preferentially expressed in invading malignant cells in human cancers. Am J Pathol. 1994;145:782–91.
Soini Y, Maatta M, Salo S, et al. Expression of the laminin gamma 2 chain in pancreatic adenocarcinoma. J Pathol. 1996;180:290–4.
Yamaguchi Y, Ishii G, Kojima M, et al. Histopathologic features of the tumor budding in adenocarcinoma of the lung: tumor budding as an index to predict the potential aggressiveness. J Thorac Oncol. 2010;5:1361–8.
Salo S, Boutaud A, Hansen AJ, et al. Antibodies blocking adhesion and matrix binding domains of laminin-332 inhibit tumor growth and metastasis in vivo. Int J Cancer. 2009;125:1814–25.
Sawai H, Okada Y, Funahashi H, et al. Activation of focal adhesion kinase enhances the adhesion and invasion of pancreatic cancer cells via extracellular signal–regulated kinase-1/2 signaling pathway activation. Mol Cancer. 2005;4:37.
Shibata K, Kikkawa F, Nawa A, et al. Both focal adhesion kinase and c-Ras are required for the enhanced matrix metalloproteinase 9 secretion by fibronectin in ovarian cancer cells. Cancer Res. 1998;58:900–3.
Meng XN, Jin Y, Yu Y, et al. Characterisation of fibronectin-mediated FAK signalling pathways in lung cancer cell migration and invasion. Br J Cancer. 2009;101:327–34.
Leader M, Collins M, Patel J, Henry K. Vimentin: an evaluation of its role as a tumour marker. Histopathology. 1987;11:63–72.
Bindels S, Mestdagt M, Vandewalle C, et al. Regulation of vimentin by SIP1 in human epithelial breast tumor cells. Oncogene. 2006;25:4975–85.
Hu L, Lau SH, Tzang CH, et al. Association of Vimentin overexpression and hepatocellular carcinoma metastasis. Oncogene. 2004;23:298–302.
Takeyama Y, Sato M, Horio M, et al. Knockdown of ZEB1, a master epithelial-to-mesenchymal transition (EMT) gene, suppresses anchorage-independent cell growth of lung cancer cells. Cancer Lett. 2010;296:216–24.
Nieto MA. Epithelial–mesenchymal transitions in development and disease: old views and new perspectives. Int J Dev Biol. 2009;53:1541–7.
Voulgari A, Pintzas A. Epithelial–mesenchymal transition in cancer metastasis: mechanisms, markers and strategies to overcome drug resistance in the clinic. Biochim Biophys Acta. 2009;1796:75–90.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Takuwa, T., Ishii, G., Nagai, K. et al. Characteristic Immunophenotype of Solid Subtype Component in Lung Adenocarcinoma. Ann Surg Oncol 19, 3943–3952 (2012). https://doi.org/10.1245/s10434-012-2428-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2428-x