Abstract
Purpose
To compare the short-term perioperative results and long-term oncologic outcomes between patients who underwent neoadjuvant chemoradiotherapy (NCRT) and patients who underwent postoperative adjuvant chemoradiotherapy (ACRT) for stage III rectal cancer.
Methods
From January 1997 to December 2008, a total of 47 patients who were diagnosed as clinical stage III rectal cancer followed by NCRT were matched according to age, gender, and operation method to 47 patients with pathologic stage III rectal cancer who underwent ACRT. Clinical characteristics, surgical and pathologic outcomes, postoperative complications and recovery, and oncologic outcomes were compared between the two groups.
Results
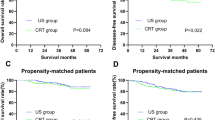
There were no significant differences in demographics or preoperative characteristics between the NCRT and ACRT groups. Though more protective ileostomies were performed in the NCRT group, there was no statistical difference in operation times between the two groups. Patients in the NCRT group had a smaller tumor size (P < 0.001) and a smaller number of lymph nodes retrieved (P < 0.001). No differences were observed with respect to morbidity and recovery outcomes between the two groups. During the median 58-month follow-up periods, the NCRT group showed better disease-free survival and overall survival than the ACRT group (P = 0.002, P = 0.001, respectively).
Conclusions
NCRT in comparison to ACRT did not increase the risk of postoperative morbidity and provided better disease-free and overall survival in stage III rectal cancer patients.

Similar content being viewed by others
References
Kapiteijn E, Marijnen CA, Nagtegaal ID, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345:638–46.
Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351:1731–40.
Weiser MR, Quah HM, Shia J, et al. Sphincter preservation in low rectal cancer is facilitated by preoperative chemoradiation and intersphincteric dissection. Ann Surg. 2009;249:236–42.
Capirci C, Valentini V, Cionini L, et al. Prognostic value of pathologic complete response after neoadjuvant therapy in locally advanced rectal cancer: long-term analysis of 566 ypCR patients. Int J Radiat Oncol Biol Phys. 2008;72:99–107.
Kim NK, Baik SH, Seong JS, et al. Oncologic outcomes after neoadjuvant chemoradiation followed by curative resection with tumor-specific mesorectal excision for fixed locally advanced rectal cancer: impact of postirradiated pathologic downstaging on local recurrence and survival. Ann Surg. 2006;244:1024–30.
Yeo SG, Kim DY, Kim TH, et al. Pathologic complete response of primary tumor following preoperative chemoradiotherapy for locally advanced rectal cancer: long-term outcomes and prognostic significance of pathologic nodal status (KROG 09-01). Ann Surg. 2010;252:998–1004.
Engstrom PF, Arnoletti JP, Benson AB 3rd, et al. NCCN Clinical Practice Guidelines in Oncology: rectal cancer. J Natl Compr Canc Netw. 2009;7:838–81.
Frasson M, Garcia-Granero E, Roda D, et al. Preoperative chemoradiation may not always be needed for patients with T3 and T2 N + rectal cancer. Cancer. 2011;117:3118–25.
Chapuis PH, Dent OF, Fisher R, et al. A multivariate analysis of clinical and pathological variables in prognosis after resection of large bowel cancer. Br J Surg. 1985;72:698–702.
Lindmark G, Gerdin B, Pahlman L, Bergstrom R, Glimelius B. Prognostic predictors in colorectal cancer. Dis Colon Rectum. 1994;37:1219–27.
Chang GJ, Rodriguez-Bigas MA, Eng C, Skibber JM. Lymph node status after neoadjuvant radiotherapy for rectal cancer is a biologic predictor of outcome. Cancer. 2009;115:5432–40.
Lee SH, Lee KC, Choi JH, et al. Chemoradiotherapy followed by surgery in rectal cancer: improved local control using a moderately high pelvic radiation dose. Jpn J Clin Oncol. 2008;38:112–21.
Kerr SF, Norton S, Glynne-Jones R. Delaying surgery after neoadjuvant chemoradiotherapy for rectal cancer may reduce postoperative morbidity without compromising prognosis. Br J Surg. 2008;95:1534–40.
Matthiessen P, Hallbook O, Andersson M, Rutegard J, Sjodahl R. Risk factors for anastomotic leakage after anterior resection of the rectum. Colorectal Dis. 2004;6:462–9.
Buie WD, MacLean AR, Attard JA, Brasher PM, Chan AK. Neoadjuvant chemoradiation increases the risk of pelvic sepsis after radical excision of rectal cancer. Dis Colon Rectum. 2005;48:1868–74.
Tan WS, Tang CL, Shi L, Eu KW. Meta-analysis of defunctioning stomas in low anterior resection for rectal cancer. Br J Surg. 2009;96:462–72.
Boccola MA, Lin J, Rozen WM, Ho YH. Reducing anastomotic leakage in oncologic colorectal surgery: an evidence-based review. Anticancer Res. 2010;30:601–7.
Roh MS, Colangelo LH, O’Connell MJ, et al. Preoperative multimodality therapy improves disease-free survival in patients with carcinoma of the rectum: NSABP R-03. J Clin Oncol. 2009;27:5124–30.
Bipat S, Glas AS, Slors FJ, Zwinderman AH, Bossuyt PM, Stoker J. Rectal cancer: local staging and assessment of lymph node involvement with endoluminal US, CT, and MR imaging—a meta-analysis. Radiology. 2004;232:773–83.
Kim JH, Beets GL, Kim MJ, Kessels AG, Beets-Tan RG. High-resolution MR imaging for nodal staging in rectal cancer: are there any criteria in addition to the size? Eur J Radiol. 2004;52:78–83.
Mizukami Y, Ueda S, Mizumoto A, et al. Diffusion-weighted magnetic resonance imaging for detecting lymph node metastasis of rectal cancer. World J Surg. 2011;35:895–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, J., Jang, S.M., Baek, JH. et al. Short-term Results and Long-term Oncologic Outcomes between Neoadjuvant Chemoradiotherapy and Adjuvant Postoperative Chemoradiotherapy for Stage III Rectal Cancer: A Case-matched Study. Ann Surg Oncol 19, 2494–2499 (2012). https://doi.org/10.1245/s10434-012-2311-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2311-9