Abstract
Background
In terms of timing of resection for synchronous liver metastases from colon cancer, some reports recommend leaving an interval (e.g., 3 months) after primary colorectal resection, because of reports of occasional and rapid remnant recurrence of residual liver metastases after simultaneous colorectal and liver resection (LR). For patients with metachronous liver metastasis (MLM), we prospectively examined the appropriateness of a 3-month interval to LR (i.e., delayed resection) following initial detection of hepatic lesions from the viewpoints of (1) detection of new metastases during this interval and (2) postoperative outcome.
Methods
Seventy-nine consecutive patients with modified Japanese criteria H1 class MLM (i.e., ≤4 nodules, 6 cm or less in diameter) and without obvious extrahepatic lesions, presenting between 1990 and 2008, were included in this study. Between 1990 and 2001, 52 patients were treated by LR without an interval (i.e., nondelayed group); from 2002, 27 patients were prospectively scheduled for LR at an interval of 3 months after initial detection of metastases (i.e., delayed group). During the 3-month interval, no adjuvant chemotherapy was given. Just prior to LR, patients were re-evaluated using computed tomography (CT) and deoxy-2-[18F]fluoro-d-glucose positron emission tomography (FDG-PET) to exclude cases unsuitable for surgery.
Results
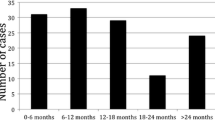
Out of 27 patients in the delayed group, 2 (7.4%) did not undergo LR after the 3-month interval and were excluded from the analysis: one because of multiple nodules in the bilateral lobe of the liver with pulmonary metastases and another because of para-aortic lymph node metastasis. Of the 25 patients for whom LR was indicated, 24 underwent LR as initially planned. In the remaining patient, after the 3-month interval, invasion of the tumor had occurred into the inferior vena cava (IVC) and other surgery in addition to the planned operation was required. When comparing the postoperative outcome data of the delayed group (n = 25) with the nondelayed group (n = 52), overall early recurrence within 1 year after LR was noticed in 30.9% (16/52) of the nondelayed group and 28.0% (7/25) of the delayed group; the incidence of only early extrahepatic recurrence decreased 5.1% in the delayed group (21.1% versus 16.0%, respectively). When comparing disease-free survival after liver resection, however, there was no significant difference between the groups.
Conclusions
Delayed LR for MLM patients after initial detection of hepatic lesions is of no clinical benefit. Only in cases when extrahepatic lesions with MLM are suspected could an interval make such lesions clearer and assist in deciding on a suitable management plan.





Similar content being viewed by others
References
Manfredi S, Lepage C, Hatem C, et al. Epidemiology and management of liver metastases from colorectal cancer. Ann Surg. 2006;244:254–9.
Bramhall SR, Gur U, Coldham C, et al. Liver resection for colorectal metastases. Ann R Coll Surg Engl. 2003;85:334–9.
Garden OJ, Rees M, Poston GJ, et al. Guidelines for resection of colorectal cancer liver metastases. Gut. 2006;55:1–8.
Kaibori M, Iwamoto S, Ishizaki M, et al. Timing of resection for synchronous liver metastases from colorectal cancer. Dig Dis Sci. 2010;55:3262–70.
Shimizu Y, Yasui K, Sano T, et al. Treatment strategy for synchronous metastases of colorectal cancer: is hepatic resection after an observation interval appropriate? Langenbecks Arch Surg. 2007;392:535–58.
Shimizu Y, Yasui K, Sano T, et al. Validity of observation interval for synchronous hepatic metastases of colorectal cancer: changes in hepatic and extrahepatic metastatic foci. Langenbecks Arch Surg. 2008;393:181–4. Epub 2008.
Yoshidome H, Kimura F, Shimizu H, et al. Interval period tumor progression: does delayed hepatectomy detect occult metastases in synchronous colorectal liver metastases? J Gastrointest Surg. 2008;12:1391–8.
Takahashi S, Konishi M, Nakagohri T, et al. Short time to recurrence after hepatic resection correlates with poor prognosis in colorectal hepatic metastasis. Jpn J Clin Oncol. 2006;36:368–75.
House MG, Ito H, Gonen M, et al. Survival after hepatic resection for metastatic colorectal cancer: trends in outcomes for 1,600 patients during two decades at a single institution. J Am Coll Surg. 2010;210:744–52.
Yumoto Y, Umeda M, Ohshima K, et al. Estimation of remnant liver function before hepatectomy by means of technetium-99m-diethylenetriamine-pentaacetic acid galactosyl human albumin. Cancer Chemother Pharmacol. 1994;33:1–6.
Elias D, Liberale G, Vernerey D, et al. Hepatic and extrahepatic colorectal metastases: when resectable, their localization does not matter, but their total number has a prognostic effect. Ann Surg Oncol. 2005;12:900–9.
Byam J, Reuter NP, Woodall CE, et al. Should hepatic metastatic colorectal cancer patients with extrahepatic disease undergo liver resection/ablation? Ann Surg Oncol. 2009;16:3064–9.
Carpizo DR, D’Angelica M. Liver resection for metastatic colorectal cancer in the presence of extrahepatic disease. Lancet Oncol. 2009;10:801–9.
de Haas RJ, Wicherts DA, Adam R. Resection of colorectal liver metastases with extrahepatic disease. Dig Surg. 2008;25:461–6.
Bennett JJ, Schmidt CR, Klimstra DS, et al. Perihepatic lymph node micrometastases impact outcome after partial hepatectomy for colorectal metastases. Ann Surg Oncol. 2008;15:1130–6.
Pawlik TM, Assumpcao L, Vossen JA, et al. Trends in nontherapeutic laparotomy rates in patients undergoing surgical therapy for colorectal metastases. Ann Surg Oncol. 2009;16:371–8.
Marti J, Mondolo MM, Fuster J, et al. Prognostic factors and time-related changes Influence results of colorectal liver metastases surgical treatment: a single-center analysis. World J Gastroenterol. 2009;15:2587–94.
Konopke R, Kersting S, Distler M, et al. Prognostic factors and evaluation of a clinical score for predicting survival after resection of colorectal liver metastases. Liver Int. 2009;29:89–102.
Kokudo N, Imamura H, Sugawara Y, et al. Surgery for multiple hepatic colorectal metastases. J Hepatobiliary Pancreat Surg. 2004;11:84–91.
Tanaka K, Shimada H, Fujii Y, et al. Pre-hepatectomy prognostic staging to determine treatment strategy for colorectal cancer metastases to the liver. Langenbecks Arch Surg. 2004;389:371–9.
Elias D, Goere D, Boige V, et al. Outcome of posthepatectomy-missing colorectal liver metastases after complete response to chemotherapy: impact of adjuvant intra-arterial hepatic oxaliplatin. Ann Surg Oncol. 2007;14:3188–94.
Chiappa A, Bertani E, Makuuchi M, et al. Neoadjuvant chemotherapy followed by hepatectomy for primarily resectable colorectal cancer liver metastases. Hepatogastroenterology. 2009;56:829–34.
Mitry E, Fields AL, Bleiberg H, et al. Adjuvant chemotherapy after potentially curative resection of metastases from colorectal cancer: a pooled analysis of two randomized trials. J Clin Oncol. 2008;26:4906–11.
Tsutsumi S, Yamaguchi S, Tsuboi K, et al. Hepatic arterial infusion combined with oral UFT/UZEL systemic chemotherapy for unresectable liver metastasis of colorectal cancer. Hepatogastroenterology. 2008;55:1419−22.
Elias D. Impact of tumor doubling time on the therapeutic strategy: application to so-called synchronous metastases of colorectal cancers. Ann Chir. 1998;52:413–20.
Cohen SJ, Punt CJ, Iannotti N, et al. Prognostic significance of circulating tumor cells in patients with metastatic colorectal cancer. Ann Oncol. 2009;20:1223–9.
Uchikura K, Ueno S, Takao S, et al. Perioperative detection of circulating cancer cells in patients with colorectal hepatic metastases. Hepatogastroenterology. 2002;49:1611–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ueno, S., Sakoda, M., Kitazono, M. et al. Is Delayed Liver Resection Appropriate for Patients with Metachronous Colorectal Metastases?. Ann Surg Oncol 18, 1104–1109 (2011). https://doi.org/10.1245/s10434-010-1418-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-1418-0