Abstract
Background
The aim of this study was to evaluate the prognostic value of the Jagged-1/Notch-1 expression in patients with head and neck carcinoma and to examine the possible role of the Jagged-1/Notch-1 signaling in tumorigenesis.
Methods
Immunohistochemical staining was performed on 59 formalin-fixed, paraffin-embedded head and neck carcinoma surgical specimens for Jagged-1 and Notch-1 expression. The head and neck cancer cell line, Fadu, with or without ectopic expression of the intracellular domain of Notch-1 (NICD) was also used for examining the tumorigenic capacity in vitro and in vivo.
Results
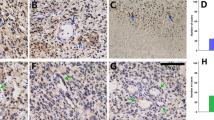
The study included 59 patients with a median age of 54 years (range, 35–73 years). Patients harboring tumors with both high-level Jagged-1 (J1Hi) and high-level Notch-1 (N1Hi) had a worse overall survival than the patients only with J1Hi or N1Hi as well as the patients with low-level Jagged-1 and Notch-1 (J1Lo/N1Lo) (P < 0.001). The 5-year survival rate and the median survival time were 5% and 10.9 months for J1Hi/N1Hi patients, while they were 35.04% and 47.7 months for non-J1Hi/NiHi patients. Ectopic expression of the active Notch-1 (NICD) in Fadu cells greatly enhanced cell migration and invasiveness in vitro and tumorigenic capacity in vivo.
Conclusions
High-level coexpression of Jagged-1 and Notch-1 is associated with poor overall survival in patients with head and neck cancer. Constitutive activation of the Notch signaling, which is possibly initiated by the direct interaction between Jagged-1 and Notch-1 in head and neck cancer, confers tumor cells with a more aggressive phenotype.


Similar content being viewed by others
References
Leong KG, Karsan A. Recent insights into the role of Notch signaling in tumorigenesis. Blood. 2006;107:2223–33.
Koch U, Radtke F. Notch and cancer: a double-edged sword. Cell Mol Life Sci. 2007;64:2746–62.
Radtke F, Raj K. The role of Notch in tumorigenesis: oncogene or tumour suppressor? Nat Rev Cancer. 2003;3:756–67.
Egan SE, St-Pierre B, Leow CC. Notch receptors, partners and regulators: from conserve domains to powerful functions. Curr Top Microbiol Immunol. 1998;228:273–324.
Dontu G, Al-Hajj M, Abdallah WM, et al. Stem cells in normal breast development and breast cancer. Cell Prolif. 2003;36(Suppl 1):59–72.
Dontu G, Jackson KW, McNicholas E, et al. Role of Notch signaling in cell-fate determination of human mammary stem/progenitor cells. Breast Cancer Res. 2004;6:R605–15.
Miele L. Notch signaling. Clin Cancer Res. 2006;12:1074–9.
Miele L, Miao H, Nickoloff BH. Notch signaling as a novel cancer therapeutic target. Curr Cancer Drug Targets. 2006;6:313–23.
Wang Z, Banerjee S, Li Y, et al. Down-regulation of Notch-1 inhibits invasion by inactivation of nuclear factor-κB, vascular endothelial growth factor, and matrix metalloproteinase-9 in pancreatic cancer cells. Cancer Res. 2006;66:2778–84.
Wang Z, Zhang Y, Li Y, et al. Down-regulation of Notch-1 contributes to cell growth inhibition and apoptosis in pancreatic cancer cells. Mol Cancer Ther. 2006;5:483–93.
Dickson BC, Mulligan AM, Zhang H, et al. High-level JAG1 mRNA and protein predict poor outcome in breast cancer. Mod Pathol. 2007;20:685–93.
Santagata S, Demichelis F, Riva A, et al. JAGGED1 expression is associated with prostate cancer metastasis and recurrence. Cancer Res. 2004;64:6854–7.
Mungamuri K, Yang X, Thor AD, et al. Survival signaling by Notch1: mammalian target of rapamycin (mTOR)-dependent inhibition of p53. Cancer Res. 2006;66:4715–24.
Nakamura T, Tsuchiya K, Watanabe M. Crosstalk between Wnt and Notch signaling in intestinal epithelial cell fate decision. J Gastroenterol. 2007;27:705–10.
Osipo C, Golde TE, Osborne BA, et al. Off the beaten pathway: the complex cross talk between Notch and NF-kappaB. Lab Invest. 2008;88:11–7.
Timmerman LA, Grego-Bessa J, Raya A, et al. Notch promotes epithelial-mesenchymal transition during cardiac development and oncogenic transformation. Genes Dev. 2004;18:99–115.
Zavadil J, Cermak L, Soto-Nieves N, et al. Integration of TGF-β/Smad and Jagged1/Notch signaling in epithelial-to-mesenchymal transition. EMBO J. 2004;23:1155–65.
Sahlgren C, Gustafsson MV, Jin S, et al. Notch signaling mediates hypoxia-induced tumor cell migration and invasion. Proc Natl Acad Sci USA. 2008;105:6392–7.
Artavanis-Tsakonas S, Matsuno K, Fortini ME. Notch signaling. Science. 1995;268:225–32.
Dievart A, Beaulieu N, Jolicoeur P. Involvement of Notch1 in the development of mouse mammary tumors. Oncogene. 1999;18:5973–81.
Kiaris H, Politi K, Grimm LM, et al. Modulation of notch signaling elicits signature tumors and inhibits hras1-induced oncogenesis in the mouse mammary epithelium. Am J Pathol. 2004;165:695–705.
Tohda S, Nara N. Expression of Notch1 and Jagged1 proteins in acute myeloid leukemia cells. Leuk Lymphoma. 2001;42:467–72.
Ascano JM, Beverly LJ, Capobianco AJ. The C terminal PDZ-ligand of JAGGED1 is essential for cellular transformation. J Biol Chem. 2003;278:8771–9.
Mani SA, Guo WJ, Liao MJ, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133:704–15.
Zeng QH, Li SG, Chepeha DB, et al. Crosstalk between tumor and endothelial cells promotes tumor angiogenesis by MAPK activation of Notch signaling. Cancer Cell. 2005;8:13–23.
Acknowledgment
We thank Ming-Chu Lu for technical assistance. This work and another related study, “Prognostic value of CD44 mRNA of peripheral blood in patients with locally advanced head and neck cancers,” were supported by the National Science Council (grant 96-2314-B-182A-026-My3) and the grant from Chang Gung Memorial Hospital (NMRPG 666012). The authors report no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, JT., Chen, MK., Yeh, KT. et al. Association of High Levels of Jagged-1 and Notch-1 Expression with Poor Prognosis in Head and Neck Cancer. Ann Surg Oncol 17, 2976–2983 (2010). https://doi.org/10.1245/s10434-010-1118-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-1118-9