Abstract
Background
The pathologic status of the sentinel lymph node (SLN) is a powerful prognostic factor for patients with intermediate thickness melanoma. We hypothesize that a high number of SLNs identified may be associated with poor outcome.
Methods
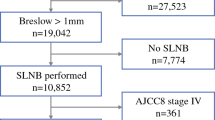
We evaluated the impact of number of SLNs removed in patients undergoing SLN mapping for cutaneous melanoma at our institution between 1996 and 2006. We excluded patients with multiple primary or synchronous primary lesions (n = 144) to eliminate any chance of erroneous association between a SLN and the wrong primary melanoma. We also excluded patients with in-transit disease (n = 37) and one patient in whom no SLN was identified, leaving 970 patients. We evaluated factors associated with the number of SLNs removed by multivariate Poisson regression and determined whether an increased number of SLNs was associated with poorer overall (OS) or recurrence-free survival (RFS).
Results
Clinical factors independently associated with increased number of SLNs were younger age and head and neck primary site. Pathologic factors associated with an increased number of SLNs were lymphovascular invasion and increased Breslow thickness. There was no association between number of SLNs removed and OS or RFS in all patients or in patients with negative SLNs (n = 803).
Conclusions
The number of SLNs identified during staging of the regional nodal basin for primary melanoma is not an independent prognostic factor. Drainage to multiple SLNs is more common in the setting of an increased Breslow depth and lymphovascular invasion suggesting that tumors with these adverse features may enhance peritumoral lymphangiogenesis.


Similar content being viewed by others
References
Balch CM, Soong SJ, Atkins MB, et al. An evidence-based staging system for cutaneous melanoma. CA Cancer J Clin. 2004;54:131–49.
Morton DL, Thompson JF, Cochran AJ, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med. 2006;355:1307–17.
Sabel MS, Griffith K, Sondak VK, et al. Predictors of nonsentinel lymph node positivity in patients with a positive sentinel node for melanoma. J Am Coll Surg. 2005;201:37–47.
Salti GI, Das Gupta TK. Predicting residual lymph node basin disease in melanoma patients with sentinel lymph node metastases. Am J Surg. 2003;186:98–101.
Wagner JD, Gordon MS, Chuang TY, et al. Predicting sentinel and residual lymph node basin disease after sentinel lymph node biopsy for melanoma. Cancer. 2000;89:453–62.
Jimenez RE, Panageas K, Busam KJ, Brady MS. Prognostic implications of multiple lymphatic basin drainage in patients with truncal melanoma. J Clin Oncol. 2005;23:518–24.
Dadras SS, Lange-Asschenfeldt B, Velasco P, et al. Tumor lymphangiogenesis predicts melanoma metastasis to sentinel lymph nodes. Mod Pathol. 2005;18:1232–42.
Dadras SS, Paul T, Bertoncini J, et al. Tumor lymphangiogenesis: a novel prognostic indicator for cutaneous melanoma metastasis and survival. Am J Pathol. 2003;162:1951–60.
Massi D, Puig S, Franchi A, et al. Tumour lymphangiogenesis is a possible predictor of sentinel lymph node status in cutaneous melanoma: a case-control study. J Clin Pathol. 2006;59:166–73.
Callejo Peixoto I, Meneses e Sousa J. Clinical and biological aspects of sentinel node biopsy in malignant melanoma—an update. Clin Transl Oncol. 2005;7:145–9.
Harrell MI, Iritani BM, Ruddell A. Tumor-induced sentinel lymph node lymphangiogenesis and increased lymph flow precede melanoma metastasis. Am J Pathol. 2007;170:774–86.
Stewart LE, Tyler DS, Vollmer RT. The importance of total number of sentinel lymph nodes in patients with stage N0 cutaneous melanoma. Am J Clin Pathol. 2005;124:77–82.
Harlow SP, Krag DN, Ashikaga T, et al. Gamma probe guided biopsy of the sentinel node in malignant melanoma: a multicentre study. Melanoma Res. 2001;11:45–55.
Fincher TR, O’Brien JC, McCarty TM, et al. Patterns of drainage and recurrence following sentinel lymph node biopsy for cutaneous melanoma of the head and neck. Arch Otolaryngol Head Neck Surg. 2004;130:844–8.
Grunhagen DJ, Eggermont AM, van Geel AN, et al. Prognostic factors after cervical lymph node dissection for cutaneous melanoma metastases. Melanoma Res. 2005;15:179–84.
Wong SL, Kattan MW, McMasters KM, Coit DG. A nomogram that predicts the presence of sentinel node metastasis in melanoma with better discrimination than the American Joint Committee on Cancer staging system. Ann Surg Oncol. 2005;12:282–8.
Chagpar RB, Ross MI, Reintgen DS, et al. Factors associated with improved survival among young adult melanoma patients despite a greater incidence of sentinel lymph node metastasis. J Surg Res. 2007;143:164–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schmidt, C.R., Panageas, K.S., Coit, D.G. et al. An Increased Number of Sentinel Lymph Nodes Is Associated with Advanced Breslow Depth and Lymphovascular Invasion in Patients with Primary Melanoma. Ann Surg Oncol 16, 948–952 (2009). https://doi.org/10.1245/s10434-009-0331-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-009-0331-x