Abstract
Background
S-1 is the most effective oral fluoropyrimidine derivative widely used for patients with gastric carcinoma in Japan. Although S-1 plus taxane has been a promising candidate as an effective chemotherapeutic regimen, the mechanisms of its additive or synergistic anticancer effects and changes in gene expression after the administration of these agents have not yet been fully elucidated.
Methods
Experimental chemotherapy was performed using human gastric carcinoma xenografts, MKN-45 and TMK-1, to examine anticancer effects and gene expressions of fluoropyrimidine metabolism-related enzymes including thymidine phosphorylase (TP), thymidylate synthase (TS), dihydropyrimidine dehydrogenase (DPD), orotate phosphoribosyltransferase (OPRT), and uridine phosphorylase (UP). Nude mice were treated with S-1, paclitaxel, and their combination. After treatment, in vivo antitumor effects of S-1, paclitaxel alone, and their combination and the effects on gene expressions of enzymes involved in 5-fluorouracil metabolism were examined using the RT-PCR method.
Results
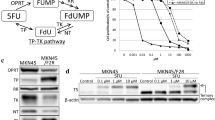
The combined use of S-1 and paclitaxel showed additive to synergistic antitumor effects on both gastric cancer xenografts. While consistent upregulation of dThPase and DPD gene expression was exhibited after administration of S-1, no further increase of dThPase gene expression after combined use of S-1 with paclitaxel was observed. There was no increase in TS gene expression after the administration of either S-1 alone or paclitaxel alone.
Conclusion
These results provide some insight into the mechanism and/or rationale underlying the additive to synergistic effect of combined administration of S-1 and paclitaxel in gastric carcinoma.




Similar content being viewed by others
References
Macdonald JS, Gohmann JJ. Chemotherapy of advanced gastric cancer: present status, future prospects. Semin Oncol 1988; 15:42–9.
Maehara Y. S-1 in gastric cancer: a comprehensive review. Gastric Cancer 2004; 6 Suppl 1:2–8.
Shirasaka T, Shimamato Y, Ohshimo H, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 1996; 7:548–57.
Shirasaka T, Nakano K, Takechi T, et al. Antitumor activity of 1 M tegafur-0.4 M 5-chloro-2,4-dihydroxypyridine-1 M potassium oxonate (S-1) against human colon carcinoma orthotopically implanted into nude rats. Cancer Res 1996; 56:2602–6.
Schoffski P. The modulated oral fluoropyrimidine prodrug S-1, and its use in gastrointestinal cancer and other solid tumors. Anticancer Drugs 2004; 15:85–106.
Sugimachi K, Maehara Y, Horikoshi N, et al. An early phase II study of oral S-1, a newly developed 5-fluorouracil derivative for advanced and recurrent gastrointestinal cancers. The S-1 Gastrointestinal Cancer Study Group. Oncology 1999; 57:202–10.
Sakata Y, Ohtsu A, Horikoshi N, et al. Late phase II study of novel oral fluoropyrimidine anticancer drug S-1 (1 M tegafur-0.4 M gimestat-1 M otastat potassium) in advanced gastric cancer patients. Eur J Cancer 1998; 34:1715–20.
Koizumi W, Kurihara M, Nakano S, et al. Phase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer. For the S-1 Cooperative Gastric Cancer Study Group. Oncology 2000; 58:191–7.
Wils JA, Klein HO, Wagener DJ, et al. Sequential high-dose methotrexate and fluorouracil combined with doxorubicin—a step ahead in the treatment of advanced gastric cancer: a trial of the European Organization for Research and Treatment of Cancer Gastrointestinal Tract Cooperative Group. J Clin Oncol 1991; 9:827–31.
Kelsen D, Atiq OT, Saltz L, et al. FAMTX versus etoposide, doxorubicin, and cisplatin: a random assignment trial in gastric cancer. J Clin Oncol 1992; 10:541–8.
Kim NK, Park YS, Heo DS, et al. A phase III randomized study of 5-fluorouracil and cisplatin versus 5-fluorouracil, doxorubicin, and mitomycin C versus 5-fluorouracil alone in the treatment of advanced gastric cancer. Cancer 1993; 71:3813–8.
Webb A, Cunningham D, Scarffe JH, et al. Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J Clin Oncol 1997; 15:261–7.
Iwase H, Shimada M, Tsuzuki T, et al. A phase I study of S-1 administration and a 24-h infusion of cisplatin plus paclitaxel in patients with advanced gastric cancer. Anticancer Res 2006; 26:1605–9.
Kondo K, Kobayashi M, Kojima H, et al. Phase I evaluation of continuous 5-fluorouracil infusion followed by weekly paclitaxel in patients with advanced or recurrent gastric cancer. Jpn J Clin Oncol 2005; 35:332–7.
Kano Y, Akutsu M, Tsunoda S, et al. Schedule-dependent interaction between paclitaxel and 5-fluorouracil in human carcinoma cell lines in vitro. Br J Cancer 1996; 74:704–10.
Fujitani K, Narahara H, Takiuchi H, et al. Phase I and pharmacokinetic study of S-1 combined with weekly paclitaxel in patients with advanced gastric cancer. Oncology 2005; 69:414–20.
Ueda Y, Yamagishi H, Ichikawa D, et al. Phase I study of a combination of S-1 and weekly paclitaxel in patients with advanced or recurrent gastric cancer. Oncology 2005; 69:261–8.
Sawada N, Nose T, Ishikawa T, et al. Enhancement of antitumor activity of 5′-deoxy-5-fluorouridine (Furtulon) by taxane in human gastric cancer xenografts. Oncol Rep 2005; 14:53–7.
Yoshida K, Ninomiya M, Takakura N, et al. Phase II study of docetaxel and S-1 combination therapy for advanced or recurrent gastric cancer. Clin Cancer Res 2006; 12:3402–7.
Sakurai Y, Yoshida I, Tonomura S, et al. Weekly administration of paclitaxel attenuated rectal stenosis caused by multiple peritoneal recurrence 8 years after the resection of gastric carcinoma. Gastric Cancer 2003; 6:243–9.
Hironaka S, Zenda S, Boku N, et al. Weekly paclitaxel as second-line chemotherapy for advanced or recurrent gastric cancer. Gastric Cancer 2006; 9:14–8.
Ohtsu A, Boku N, Tamura F, et al. An early phase II study of a 3-hour infusion of paclitaxel for advanced gastric cancer. Am J Clin Oncol 1998; 21:416–9.
Yamada Y, Shirao K, Ohtsu A, et al. Phase II trial of paclitaxel by three-hour infusion for advanced gastric cancer with short premedication for prophylaxis against paclitaxel-associated hypersensitivity reactions. Ann Oncol 2003; 12:1133–7.
Yamaguchi K, Tada M, Horikoshi N, et al. Phase II study of paclitaxel with 3-h infusion in patients with advanced gastric cancer. Gastric Cancer 2002; 5:90–5.
Mochiki E, Ohno T, Kamiyama Y, et al. Phase I/II study of S-1 combined with paclitaxel in patients with unresectable and/or recurrent advanced gastric cancer. Br J Cancer 2006; 95:1642–7.
Hokita S, Aikou T, Miyazono F, et al. A phase I combination chemotherapy study of biweekly paclitaxel and S-1 administration in patients with advanced gastric cancer. Cancer Chemother Pharmacol 2006; 57:736–40.
Yamaguchi K, Shimamura T, Hyodo I, et al. Phase I/II study of docetaxel and S-1 in patients with advanced gastric cancer. Br J Cancer 2006; 94:1803–8.
Motoyama T, Hojo H, Watanabe H. Comparison of seven cell lines derived from human gastric carcinomas. Acta Pathol Jpn 1986; 36:65–83.
Ochiai A, Yasui W, Tahara E. Growth-promoting effect of gastrin on human gastric carcinoma cell line TMK-1. Jpn J Cancer Res 1985; 76:1064–71.
Yamada Y, Yoshida T, Hayashi K, et al. p53 gene mutations in gastric cancer metastases and in gastric cancer cell lines derived from metastases. Cancer Res 1991; 51:5800–5.
Geran RI, Greenberg NH, Schumacher AM, et al. Protocols for screening chemical agents and natural products against animal tumors and other biological systems. 3rd ed. Cancer Chemother Rep 1972; 3:51–61.
Ovejera AA, Houchens DP, Barker AD. Chemotherapy of human tumor xenografts in genetically athymic mice. Ann Clin Lab Sci 1978; 8:50–6.
Houchens DP, Ovejera AA, Baker A. (2007) The therapy of human tumors in athymic (nude) mice. In: Houchens DP, Ovejera AA (eds). Proceedings of the Symposium on the Use of Athymic (nude) Mice in Cancer Research. Gustav Fisher, New York, Stuttgart, pp 267–80
Gavrieli Y, Sherman Y, Ben-Sasson SA. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 1992; 119:493–501.
Leers MP, Kolgen W, Bjorklund V, et al. Immunocytochemical detection and mapping of a cytokeratin 18 neo-epitope exposed during early apoptosis. J Pathol 1999; 187:567–72.
Sawada N, Ishikawa T, Sekiguchi F, et al. X-ray irradiation induces thymidine phosphorylase and enhances the efficacy of capecitabine (Xeloda) in human cancer xenografts. Clin Cancer Res 1999; 5:2948–53.
Eda H, Fujimoto K, Watanabe S, et al. Cytokines induce thymidine phosphorylase expression in tumor cells and make them more susceptible to 5′-deoxy-5-fluorouridine. Cancer Chemother Pharmacol 1993; 32:333–8.
Fukushima M, Okabe H, Takechi T, et al. Induction of thymidine phosphorylase by interferon and taxanes occurs only in human cancer cells with low thymidine phosphorylase activity. Cancer Lett 2002; 187:103–10.
Sawada N, Ishikawa T, Fukase Y, et al. Induction of thymidine phosphorylase activity and enhancement of capecitabine efficacy by taxol/taxotere in human cancer xenografts. Clin Cancer Res 1998; 4:1013–9.
Sakurai Y, Uraguchi T, Imazu H, et al. Changes in thymidylate synthase and its inhibition rate and changes in dihydropyrimidine dehydrogenase after the administration of 5-fluorouracil with cisplatin to nude mice with gastric cancer xenograft SC-1-NU. Gastric Cancer 2004; 7:110–6.
Omura K, Kawakami K, Kanehira E, et al. The number of 5-fluoro-2′-deoxyuridine-5′-monophosphate binding sites and reduced folate pool in human colorectal carcinoma tissues: changes after tegafur and uracil treatment. Cancer Res 1995; 55:3897–901.
Langenbach RJ, Danenberg PV, Heidelberger C. Thymidylate synthetase: mechanism of inhibition by 5-fluoro-2′-deoxyuridylate. Biochem Biophys Res Commun 1972; 48:1565–71.
Santi DV, McHenry CS. 5-Fluoro-2′-deoxyuridylate: covalent complex with thymidylate synthetase. Proc Natl Acad Sci USA 1972; 69:1855–7.
Spears CP, Shahinian AH, Moran RG, et al. In vivo kinetics of thymidylate synthetase inhibition of 5-fluorouracil-sensitive and -resistant murine colon adenocarcinomas. Cancer Res 1982; 42:450–6.
Parker WB, Cheng YC. Metabolism and mechanism of action of 5-fluorouracil. Pharmacol Ther 1990; 48:381–95.
Johnston PG, Lenz HJ, Leichman CG, et al. Thymidylate synthase gene and protein expression correlate and are associated with response to 5-fluorouracil in human colorectal and gastric tumors. Cancer Res 1995; 55:1407–12.
Salonga D, Danenberg KD, Johnson M, et al. Colorectal tumors responding to 5-fluorouracil have low gene expression levels of dihydropyrimidine dehydrogenase, thymidylate synthase, and thymidine phosphorylase. Clin Cancer Res 2000; 6:1322–7.
Sakurai Y, Sakamoto K, Sugimoto Y, et al. Orotate phosphoribosyltransferase levels measured by a newly established enzyme-linked immunosorbent assay in gastric carcinoma. Cancer Sci 2006; 97:492–8.
Acknowledgements
This study was supported in part by grants from Ministry of Health, Labor and Welfare (#14571242) and from Fujita Health University. The authors thank Miss Akemi Someya for an excellent technical assistance for performing the animal experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakurai, Y., Yoshida, I., Kamoshida, S. et al. Effects of Combined Administration of DPD-Inhibitory Oral Fluoropyrimidine, S-1, Plus Paclitaxel on Gene Expressions of Fluoropyrimidine Metabolism-Related Enzymes in Human Gastric Xenografts. Ann Surg Oncol 15, 2301–2309 (2008). https://doi.org/10.1245/s10434-008-9963-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-008-9963-5