Abstract
Background
Endometrial carcinoma is the most common gynecological malignancy. Several molecular biological characteristics have been studied for their potential value in patient management.
Objectives
Our objectives were to compare p53 immunohistochemical expression with P53 gene status determined by fluorescence in situ hybridization (FISH) and to compare these characteristics with ploidy and with classical clinical and histological prognostic factors.
Materials and Methods
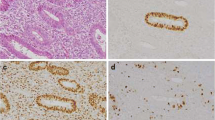
We reviewed stored specimens from 43 patients with endometrial cancer diagnosed in 1999–2004. P53 FISH and immunohistochemistry were performed, together with imaging cytometry to calculate DNA ploidy.
Results
Thirteen of the 43 endometrial carcinomas (30.2%) showed P53 loss of heterozygosity (LOH). P53 LOH correlated with the histological type (P = .03) and the histological grade (P = .004). Quantitative immunohistochemical expression of p53 protein correlated with the histological type (P = .0001). With a cutoff of 10% of p53-positive cells, p53 overexpression correlated with the histological type (P = .003) and grade (P = .0008). No relation was found between P53 LOH or immunohistochemical expression and the disease stage, the depth of myometrial invasion, lymph node status, lymphovascular space involvement, recurrence, or death from cancer. Nondiploid carcinomas showed deeper myometrial invasion than diploid carcinomas (P = .01). No relation was observed between ploidy and qualitative or semiquantitative p53 expression or P53 LOH.
Conclusion
In endometrial cancer, FISH analysis of P53 status adds no significant prognostic information compared with immunohistochemical p53 analysis.


Similar content being viewed by others
References
Cook LS, Kmet LM, Magliocco AM, et al. Endometrial cancer survival among U.S. black and white women by birth cohort. Epidemiology 2006; 17:469–72
Rose PG. Endometrial carcinoma. N Engl J Med 1996; 335:640–9
Fisher B, Costantino JP, Redmond CK, et al. Endometrial cancer in tamoxifen-treated breast cancer patients: findings from the National Surgical Adjuvant Breast and Bowel Project (NSABP) B-14. J Natl Cancer Inst 1994; 86:527–37
Mignotte H, Lasset C, Bonadona V, et al. Iatrogenic risks of endometrial carcinoma after treatment for breast cancer in a large French case-control study. FNCLCC. Int J Cancer 1998; 76:325–30
Watson P, Vasen HF, Mecklin JP, et al. The risk of endometrial cancer in hereditary nonpolyposis colorectal cancer. Am J Med 1994; 96:516–20
May P, May E. Twenty years of p53 research: structural and functional aspects of the p53 protein. Oncogene 1999; 18:7621–36
Drane P, Bravard A, Bouvard V, et al. Reciprocal downregulation of p53 and SOD2 gene expression: implication in p53 mediated apoptosis. Oncogene 2001; 20:430–9
Mariani A, Sebo TJ, Webb MJ, et al. Molecular and histopathologic predictors of distant failure in endometrial cancer. Cancer Detect Prevent 2003; 27:434–41
Alkushi A, Lim P, Coldman A, et al. Interpretation of p53 immunoreactivity in endometrial carcinoma: establishing a clinically relevant cut-off level. Int J Gynaecol Pathol 2004; 23:129–37
Coronado PJ, Vidart JA, Lopez-Asenjo JA. P53 overexpression predicts endometrial carcinoma better than HER-2/neu overexpression. Eur J Obstet Gynecol Reprod Biol 2001; 98:103–8
Ragni N, Ferrero S, Prefumo F, et al. The association between p53 expression, stage and histological features in endometrial cancer. Eur J Obstet Gynecol Reprod Biol 2005; 123:111–6
Kounelis S, Kapranos N, Kouri E, et al. Immunohistochemical profile of endometrial adenocarcinoma: a study of 61 cases and review of the literature. Mod Pathol 2000; 13:379–88
Davidoff AM, Humphrey PA, Iglehart JD, et al. Genetic basis for p53 overexpression in human breast cancer. Proc Natl Acad Sci USA 1991; 88:5006–10
Iggo R, Gatter K, Bartek J, et al. Increased expression of mutant forms of p53 oncogene in primary lung cancer. Lancet 1990; 335:675–9
Hall PA, Lane DP. P53 in tumour pathology: can we trust immunohistochemistry? J Pathol 1994; 172:1–4
Soong R, Knowles S, Williams KE, et al. Overp53 expression protein is an independent prognostic indicator in human endometrial carcinoma. Br J Cancer 1996; 74:562–7
Taskin M, Lallas TA, Barber HR, et al. bcl-2 and p53 in endometrial adenocarcinoma. Mod Pathol 1997; 10:728–34
Erdem O, Erdem M, Dursun A, et al. Angiogenesis, p53, and bcl-2 expression as prognostic factors in endometrial cancer: comparison with traditional clinicopathological variables. Int J Gynecol Pathol 2003; 22:254–60
Zheng W, Cao P, Zheng M, et al. p53 overexpression and bcl-2 persistence in endometrial carcinoma: comparison of papillary serous and endometrioid subtypes. Gynecol Oncol 1996; 61:167–74
Ito K, Watanabe K, Nasim S, et al. Prognostic significance of p53 overexpression in endometrial cancer. Cancer Res 1994; 54:4667–70
Williams JA Jr, Wang ZR, Parrish RS, et al. Fluorescence in situ hybridization analysis of Her-2/neu, c-myc and p53 in endometrial cancer. Exp Mol Pathol 1999; 67:135–43
Sandberg AA. Cancer cytogenetics for clinicians. Ca Cancer J Clin 1994; 44:136–59
Lim P, Aquino-Parsons CF, Wong F, et al. Low-risk endometrial carcinoma: assessment of a treatment policy based on tumor ploidy and identification of additional prognostic indicators. Gynecol Oncol 1999; 73:191–5
Santala M, Talvensaari-Mattila A. DNA ploidy is an independent prognostic indicator of overall survival in stage I endometrial endometrioid carcinoma. Anticancer Res 2003; 23:5191–96
Benedet JL, Bender H, Jones H 3rd, et al. FIGO staging classifications and clinical practice guidelines in the management of gynaecologic cancers. FIGO committee on Gynecologic Oncology. Int J Gynaecol Obstet 2000; 70:209–62
Kallioniemi A, Kallioniemi OP, Waldman FM, et al. Detection of retinoblastoma gene copy number in metaphase chromosomes and interphase nuclei by fluorescence in situ hybridization. Cytogenet Cell Genet 1992; 60:190–3
De Jong JS, Van Diest PJ, Baak JPA. Methods in laboratory investigation. Heterogeneity and reproducibility of microvessel counts in breast cancer. Lab Invest 1995; 73:922–6
Weidner N. Intratumor microvessel density as a prognostic factor in cancer. Am J Pathol 1995; 147:9–19
Matias-Guiu X, Catasus L, Bussaglia E, et al. Molecular pathology of endometrial hyperplasia and carcinoma. Hum Pathol 2001; 32:569–77
Tashiro H, Isacson C, Levine R, et al. P53 gene mutations are common in uterine serous carcinoma and occur early in their pathogenesis. Am J Pathol 1997; 150:177–85
Jovanovic AS, Boynton KA, Mutter GL. Uteri of women with endometrial carcinoma contain a histopathologic spectrum of monoclonal putative precancers, some with microsatellite instability. Cancer Res 1996; 56:1917–21
Oehler MK, Brand A, Wain GV. Molecular genetics and endometrial cancer. J Br Menopause Soc 2003; 9:27–31
Inoue M, Okayama A, Fujita M, et al. Clinicopathological characteristics of p53 overexpression in endometrial cancers. Int J Cancer 1994; 58:14–9
Lukes AS, Kohler MF, Pieper CF, et al. Multivariable analysis of DNA ploidy, P53, and HER-2/NEU as prognostic factors in endometrial cancer. Cancer 1994; 73:2380–5
Leong AS-Y. Quantitation in immunohistology: fact or fiction? Appl Immunohistochem Mol Morphol 2004; 12:1–7
McCluggage WG, Connolly LE, McGregor G, et al. A strategy for defining biologically relevant levels of p53 protein expression in clinical samples with reference to endometrial neoplasia. Int J Gynecol Pathol 2005; 24:307–12
Nenutil R, Smardova J, Pavlova S, et al. Discriminating functional and non functional p53 in human tumours by p53 and MDM2 immunohistochemistry. J Pathol 2005; 207:251–9
Wynford-Thomas D. P53 in tumour pathology: can we trust immunohistochemistry? J Pathol 1992; 166:329–30
Soussi T, Beroud C. Assessing TP53 status in human tumours to evaluate clinical outcome. Nature Rev Cancer 2001; 1:233–40
Soussi T, Kato S, Levy PP, et al. Reassessment of the TP53 mutation database in human disease by data mining with a library of TP53 missense mutations. Hum Mutat 2005; 25:6–17
Ludwig RL, Bates S, Vousden KH. Differential activation of target cellular promoters by p53 mutants with impaired apoptotic function. Mol Cell Biol 1996; 16:4952–60
Semczuk A, Marzec B, Skomra D, et al. Allelic loss at TP53 is not related to p53 protein overexpression in primary human endometrial carcinomas. Oncology 2005; 69:317–25
Stewart RL, Royds JA, Burton JL, et al. Direct sequencing of the p53 gene shows absence of mutations in endometrioid endometrial adenocarcinomas expressing p53 protein. Histopathology 1998; 33:440–5
Coleman RL, Schinck JC, Miller DS, et al. DNA flow cytometric analysis of clinical stage I endometrial carcinomas with lymph node metastases. Gynecol Oncol 1993; 50:20–4
Terada K, Mattson D, Goo D, et al. DNA aneuploidy is associated with increased mortality for stage I endometrial cancer. Gynecol Oncol 2004; 95:483–7
Lundgren C, Auer G, Frankendal B, et al. Prognostic factors in surgical stage I endometrial carcinoma. Acta Oncol 2000; 43:49–56
Orbo A, Rydningen M, Straume B, et al. Significance of morphometric, DNA cytometric features, and other prognostic markers on survival of endometrial cancer patients in Northern Norway. Int J Gynecol Cancer 2002; 12:49–56
Biesterfeld S, Leitloff M, Rath W, et al. DNA image cytometry in the differential diagnosis of endometrial hyperplasia and adenocarcinoma. Analyt Quant Cytol Histol 2001; 23:123–8
Osmanagaoglu MA, Kadioglu S, Osmanagaoglu S, et al. The relationship between mutant p53 gene, DNA contents and conventional clinicopathological prognostic variables in cases with endometrial carcinoma. Eur J Gynaecol Oncol 2005; 26:64–70
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graesslin, O., Chantot-Bastaraud, S., Lorenzato, M. et al. Fluorescence in situ Hybridization and Immunohistochemical Analysis of p53 Expression in Endometrial Cancer: Prognostic Value and Relation to Ploidy. Ann Surg Oncol 15, 484–492 (2008). https://doi.org/10.1245/s10434-007-9712-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-007-9712-1