Abstract
Background
Total mesorectal excision (TME) is the standard surgical choice for rectal adenocarcinoma. Better prognostic results, achieved with a retroperitoneal and lateral pelvic lymphadenectomy (RLPL), have questioned that TME might not be satisfactory for adequate patient staging, affecting therapeutic definitions. The aims of the ongoing study are to define the accuracy of dye and probe search in the detection of metastatic retroperitoneal and lateral pelvic nodes (RLPN) resected with RLPL, and to evaluate the metastasis frequency in these nodes and its eventual upstaging impact.
Methods
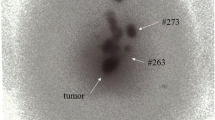
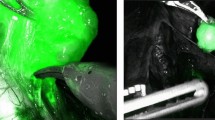
Thirty rectal adenocarcinoma patients were submitted to RLPL, with RLPN mapping using technetium (Tc 99 m) and patent blue, having nodes examined histopathologically and immunohistochemically.
Results
Eight hundred and two nodes were analyzed, mean of 26.7 per patient; RLPL was responsible for 41% (330) of the examined nodes, mean of 11 per patient. Metastatic RLPN have occurred in 20% of the patients; the RLPN were metastatic in only 6.7% of the patients; RLPL upstaged 13.3%. For identification of metastatic RLPN with technetium, sensitivity was 33%, specificity 79%, positive predictor value (PPV) 29%, negative predictor value (NPV) 83% and false negative (FN) rate 67%. For patent blue and technetium metastatic RLPN identification, sensitivity was 17%, specificity 92%, PPV 33%, NPV 82% and FN 83%.
Conclusions
Preliminary results have pointed out technetium and blue dye low accuracy to identify metastatic RLPN; no metastatic RLPN were reported in the patients submitted to preoperative chemoradiation and important upstaging with RLPL. Considering no increase in morbi-mortality rates with RLPL, definitive conclusions will be obtained as the study carries on.
Similar content being viewed by others
References
Dukes CE. The classification of cancer of the rectum. J Pathol Bacteriol 1932; 35:323–32
Greene FL, Page DL, Fleming ID, et al. (2002). AJCC cancer staging manual, 6th edn. Berlin Heidelberg, New York: Springer.
Japanese Society for Cancer of the Colon and Rectum, eds. (1998). General rules for clinical and pathological studies on cancer of the colon, rectum and anus, 6th edn. Tokyo: Kanehara & Co.
Cserni G. Nodal staging of colorectal carcinomas and sentinel nodes. J Clin Pathol 2003; 56:327–35
Nelson H, Petrelli N, Carlin A, et al. Guidelines 2000 for colon and rectal cancer surgery. J Natl Cancer Inst 2001; 93:583–96
Cserni G, Vinh-Hung V, Burzykowsi T. Are there a minimum number of lymph nodes that should be histologically assessed for a reliable nodal staging of T3N0M0 colorectal carcinomas? J Surg Oncol 2002; 81:63–9
Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery—the clue to pelvic recurrence? Br J Surg 1982; 69:613–6
Jessuo JM, Stewart AK, Menck HR. The national cancer data base report on patterns of care for adenocarcinoma of the rectum, 1985–1995. Cancer 1998; 83(11):2408–18
Japanese Society for Cancer of the Colon and Rectum. Multi-institutional registry of large bowel cancer in Japan, cases treated in 1994, vol 23. Tokyo, 2002
Moriya Y, Sugihara K, Akasu T, Fujita S. Nerve-sparing surgery with lateral node dissection for advanced lower rectal cancer. Eur J Cancer 1995; 31A(7):1229–32
Francini G, Petrioli R, Lorenzini L, et al. Folinic acid and 5-fluorouracil as adjuvant chemotherapy in colon cancer. Gastroenterology 1994; 106(4):899–906
IMPACT. Efficacy of adjuvant flourouracil and folinic acid in colon cancer. International multicentre pooled analysis of colon cancer trials (IMPACT) investigators. Lancet 1995; 345(8955):939–44
Moertel CG, Fleming TR, Macdonald JS, et al. Fluoracil plus levamisole as effective adjuvant therapy after resection of stage III colon carcinoma: a final report. Ann Intern Med 1995; 122(5):321–6
Joosten JJ, Strobbe LJ, Wauters CA, Pruszczynski M, Wobbes T, Ruers TJ. Intraoperative lymphatic mapping and the sentinel node concept in colorectal carcinoma. Br J Surg 1999; 86(4):482–6
Saha S, Wiese D, Badin J, et al. Technical details of sentinel lymph node mapping in colorectal cancer and its impact on staging. Ann Surg Oncol 2000; 7(2):120–4
Saha S, Monson KM, Bilchik A, et al. Comparative analysis of nodal upstaging between colon and rectal cancers by sentinel node mapping: a prospective trial. Dis Colon Rectum 2004; 47(11):1767–72
Trocha SD, Nora DT, Saha SS, Morton DL, Wiese D, Bilchik AJ. Combination probe and dye-directed lymphatic mapping detects micrometastases in early colorectal cancer. J Gastrointest Surg 2003; 7(3):340–5
Cserni G, Vajda K, Tarján M, Bori R, Svebis M, Baltas B. Nodal staging of colorectal carcinomas from quantitative and qualitative aspects. Can lymphatic mapping help staging? Pathol Oncol Res 1999; 5(4):291–6
Bilchik AJ, Nora D, Tollenaar, et al. Ultrastaging of early colon cancer using lymphatic mapping and molecular analysis. Eur J Cancer 2002; 38(7):977–85
Keshtgar MR, Amin A, Taylor I. Intraoperative lymphatic mapping and the sentinel node concept in colorectal carcinoma. Br J Surg 1999; 86(9):1225–6
Viehl CT, Hamel CT, Marti WR, et al. Identification of sentinel lymph nodes in colon cancer depends on the amount of dye injection relative to tumor size. World J Surg 2003; 27(12):1285–90
Acknowledgments
The authors thank Dr. Aristides Maltez Filho, President of the Liga Bahiana Contra o Cancer – LBCC, who supported this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Almeida Quadros, C., Lopes, A., Araújo, I. et al. Retroperitoneal and Lateral Pelvic Lymphadenectomy Mapped by Lymphoscintigraphy and Blue Dye for Rectal Adenocarcinoma Staging: Preliminary Results. Ann Surg Oncol 13, 1617–1621 (2006). https://doi.org/10.1245/s10434-006-9116-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-006-9116-7