Abstract
Background
Serum amyloid A (SAA) is a useful biomarker for gastric cancer in an animal model. We investigated the potential of SAA as a biomarker for gastric cancer in humans.
Methods
Serum levels of SAA from 96 gastric cancer patients were measured before and after curative gastrectomy; 32 patients with gastric ulcers and 52 healthy subjects were the control groups. The immunohistochemical study was performed to evaluate the protein expression over gastric cancer tissue slides.
Results
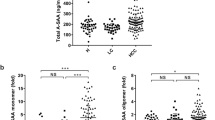
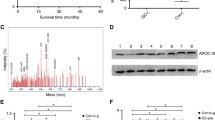
The mean SAA concentration was higher in gastric cancer patients (88.54 ± 50.44 mg/l) than in healthy subjects (3.36 ± 2.29 mg/l) and gastric ulcer patients (10.48 ± 8.97 mg/l) (P < .05). The SAA concentration was associated with tumor stage (P = .0244) and location (P = .0016) but not with Lauren’s histological type (P = .839). In the multivariate analysis, SAA level was correlated with tumor location (P < .0001) and lymph node status (P < .05). During follow-up, the mean SAA concentration increased significantly in 24 patients with tumor recurrence (P < .05) but did not change in 77 patients without recurrence. In the survival analysis, patients with SAA levels > 97 mg/l had a nearly fourfold increase in risk of death. Immunoreactivity was most prominent in blood vessel regions but not within cancer cells.
Conclusions
These data not only demonstrated SAA was useful in predicting survival of patients with gastric cancer, but they also showed that SAA was a valuable tool for postoperative follow-up.






Similar content being viewed by others
References
Stadtlander CT, Waterbor JW. Molecular epidemiology, pathogenesis and prevention of gastric cancer. Carcinogenesis 1999; 20:2195–208
Health and Vital Statistics. Taipei: Department of Health, Executive Yuan, Republic of China, 1997
Collard JM, Malaise J, Mabrut JY, et al. Skeletonizing. En-bloc gastrectomy for adenocarcinoma in Caucasian patients. Gastric Cancer 2003; 6:210–16
Juan HF, Chen JH, Hsu WT, et al. Identification of tumor-associated plasma biomarkers using proteomic techniques: From mouse to human. Proteomics 2004; 4:2766–775
Sheu LF, Chen A, Wei YH, et al. Epstein-Barr virus LMP1 modulates the malignant potential of gastric carcinoma cells involving apoptosis. Am J Pathol 1998; 152:63–74
Yamada T. Serum amyloid A (SAA): a concise review of biology, assay methods and clinical usefulness. Clin Chem Lab Med 1999; 37:381–8
Biran H, Friedman N, Neumann PM, et al. Serum amyloid A (SAA) variations in patients with cancer: correlation with disease activity, stage, primary site, and prognosis. J Clin Pathol 1986; 39:794–7
Kimura M, Tomita Y, Imai T, et al. Significance of serum amyloid A on the prognosis in patients with renal cell carcinoma. Cancer 2001; 92(8):2072–5
Glojnaric I, Casl MT, Simic D, et al. Serum amyloid A protein (SAA) in colorectal carcinoma. Clin Chem Lab Med 2001; 39(2):129–33
Kaneti J, Winikoff Y, Zimlichman S, et al. Importance of serum amyloid A (SAA) level in monitoring disease activity and response to therapy in patients with prostate cancer. Urol Res 1984; 12(5):239–41
Cho WC, Yip TT, Yip C, et al. Identification of serum amyloid a protein as a potentially useful biomarker to monitor relapse of nasopharyngeal cancer by serum proteomic profiling. Clin Cancer Res 2004; 10(1 Pt 1):43–52
O’Hanlon DM, Lynch J, Cormican M, Given HF. The acute phase response in breast carcinoma. Anticancer Res 2002; 22:1289–93
Rosenthal CJ, Sullivan LM. Serum amyloid A to monitor cancer dissemination. Ann Intern Med 1979; 91:383–90
Japanese Research Society for Gastric Cancer. (1993). The General Rules for the Gastric Cancer Study. 12th edn. Tokyo: Kanehara, pp 30–3
Sobin LH, Wittekind CH. (1997). International Union Against Cancer (UICC): TNM classification of malignant tumors, 5th edn. New York: John Wiley.
Lauren P. The two histological main types of gastric carcinoma: Diffuse and so-called intestinal-type carcinoma. Acta Pathol Microbiol Scand 1965; 64:31–49
Juan HF, Lin JY, Chang WH, et al. Biomic study of human myeloid leukemia cells differentiation to macrophages using DNA array, proteomic, and bioinformatics analytical methods. Electrophoresis 2002; 23:2490–504
d’Eril GM, Anesi A, Maggiore M, et al. Biological variation of serum amyloid A in healthy subjects. Clin Chem 2001; 47:1498–9
Shimizu N, Wakatsuki T, Murakami A. Carcinoembryonic antigen in gastric cancer patients. Oncology 1987; 44:240–4
Nakane Y, Okamura S, Akehira K, et al. Correlation of preoperative carcinoembryonic antigen levels and prognosis of gastric cancer patients. Cancer 1994; 73:2703–8
Koga T, Kano T, Souda K. The clinical usefulness of preoperative CEA determination in gastric cancer. Jpn J Surg 1987; 17:342–7
Sakamoto K, Haga Y, Yoshimura R, et al. Comparative effectiveness of the tumour diagnostics, CA 19-9, CA125 and carcinoembryonic antigen in patients with diseases of the digestive system. Gut 1987; 28:323–9
Marrelli D, Roviello F, De Stefano A, et al. Prognostic significance of CEA, CA 19-9 and CA 72-4 preoperative serum levels in gastric carcinoma. Oncology 1999; 57:55–62
Safi F, Kuhns V, Beger HG. Comparison of CA72-4, CA19-9 and CEA in the diagnosis and monitoring of gastric cancer. Int J Biol Markers 1995; 10:100–6
Parsonnet J, Friedman GD, Vandersteen DP, et al. Helicobacter pylori infection and the risk of gastric carcinoma. N Engl J Med 1991; 325:1127–31
Forman D, Newell DG, Fullerton F, et al. Association between infection with Helicobacter pylori and risk of gastric cancer: evidence from a prospective investigation. BMJ 1991; 302:1302–5
Hansen S, Melby KK, Aase S, Jellium E, Vollset SE. Helicobacter pylori infection and risk of cardia cancer and non-cardia cancer. Scand J Gastroenterol 1999; 4:353–60
Delanghe JR, Langlois MR, De Bacquer D, Mak R, Capel P, Van Renterghem L, De Backer G. Discriminative value of serum amyloid A and other acute-phase proteins for coronary heart disease. Atherosclerosis 2002; 160(2): 471–6
Hagihara K, Nishikawa T, Isobe T, et al. IL-6 plays a critical role in the synergistic induction of human serum amyloid A (SAA) gene when stimulated with proinflammatory cytokines as analyzed with an SAA isoform real-time quantitative RT-PCR assay system. Biochem Biophys Res Commun 2004; 314(2):363–9
Galizia G, Lieto E, De Vita F, et al. Circulating levels of interleukin-10 and interleukin-6 in gastric and colon cancer patients before and after surgery: Relationship with radicality and outcome. J Interferon Cytokine Res 2002; 22:473–82
Yamaoka Y, Kodama T, Kita M, et al. Relation between cytokines and Helicobacter pylori in gastric cancer. Helicobacter 2001; 6:116–24
De Vita F, Romano C, Orditura M, et al. Interleukin-6 serum level correlates with survival in advanced gastrointestinal cancer patients but is not an independent prognostic indicator. J Interferon Cytokine Res 2001; 21:45–52
Wu CW, Wang SR, Chao MF, et al. Serum interleukin-6 levels reflect disease status of gastric cancer. Am J Gastroenterol 1996; 91:1417–22
Kabir S, Daar GA. Serum levels of interleukin-1, interleukin-6 and tumor necrosis factor-alpha in patients with gastric cancer. Cancer Lett 1995; 95:207–12
Tatsuno I, Nishikawa T, Sasano H, et al. Interleukin 6-producing gastric carcinoma with fever, hypergammaglobulinemia, and plasmacytosis in bone marrow. Gastroenterology 1994; 107:543–7
Lin MT, Juan CY, Chang KJ, et al. IL-6 inhibits apoptosis and retains oxidative DNA lesions in human gastric cancer AGS cells through up-regulation of anti-apoptotic gene mcl-1. Carcinogenesis 2001; 22:1947–53
Ding SZ, Cho CH, Lam SK. Regulation of IL-6 production in human gastric epithelial cell line MKN-28. Cytokine 2000; 12:1129–35
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chan, DC., Chen, CJ., Chu, HC. et al. Evaluation of Serum Amyloid A as a Biomarker for Gastric Cancer. Ann Surg Oncol 14, 84–93 (2007). https://doi.org/10.1245/s10434-006-9091-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-006-9091-z