Abstract
Background
We have identified a novel function of MAL (T-cell differentiation-related gene) as a candidate suppressor gene in esophageal cancer. As the role of MAL expression in esophageal carcinogenesis is as yet undetermined, MAL expression in a rat multi-step carcinogenic model and in precancerous lesions of the human esophagus was investigated. Microarray analysis between MAL-transfectant and control cells was also carried out to clarify how MAL confers its anti-tumor effects.
Materials and Methods
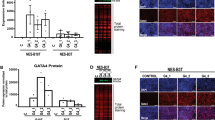
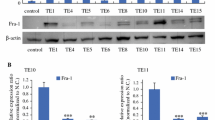
(1) In the rat model, MAL expression levels in laser microdissected normal esophageal epithelium, dysplastic tissues and carcinoma tissues were examined by reverse transcription (RT)-PCR. (2) Immunostaining with MAL antibody was performed in 10 dysplastic lesions adjacent to cancer in six cases of esophageal cancer. (3) We established a MAL transfectant using a Tet-off vector in esophageal cancer cells and performed microarray analysis under MAL-positive and MAL-negative conditions.
Results
(1) In the rat model, MAL mRNA expression was observed only in the normal samples. (2) MAL expression was observed distinctively in differentiated or keratinized normal tissues and was not observed in either dysplastic lesions or carcinoma tissue. (3) Up-regulated genes in MAL-positive cells included keratin 18 (transfectant/control = 2.94) and keratin 10 (t/c = 2.82).
Conclusion
MAL expression was lost in dysplastic lesions of the rat carcinoma model as well as the human esophagus. The up-regulated keratins revealed by microarray analysis and the strong staining of the differentiated normal tissues in immunohistochemical study support the role of MAL as a regulator of differentiation in esophageal epithelium.



Similar content being viewed by others
References
Alonso MA, Barton DE, Francke U. Assignment of the T-cell differentiation gene MAL to human chromosome 2, region cenq13. Immunogenetics 1988; 27:91–95
Alonso MA, Weissman SM. cDNA cloning and sequence of MAL, a hydrophobic protein associated with human T-cell differentiation. Proc Natl Acad Sci USA 1987; 84:1997–2001
Mimori K, Shiraishi T, Mashino K, et al. MAL gene expression in esophageal cancer suppresses motility, invasion and tumorigenicity and enhances apoptosis through the Fas pathway. Oncogene 2003; 22:3463–3471
Matsufuji H, Ueo H, Mori M, Kuwano H, Sugimachi K. Enhancement of esophageal carcinogenesis induced in rats by N-amyl-N-methylnitrosamine in the presence of 12-O-tetradecanoylphorbol-13-acetate. J Natl Cancer Inst 1987; 79:1123–1129
Nishida K, Mine S, Utsunomiya T, et al. Global analysis of altered gene expressions during the process of esophageal squamous cell carcinogenesis in the rat: a study combined with a laser microdissection and a cDNA microarray. Cancer Res 2005;65:401–409
Jiang W, Kahn SM, Tomita N, Zhang YJ, Lu SH, Weinstein IB. Amplification and expression of the human cyclin D gene in esophageal cancer. Cancer Res 1992;52:2980–2983
Naitoh H, Shibata J, Kawaguchi A, Kodama M, Hattori T. Overexpression and localization of cyclin D1 mRNA and antigen in esophageal cancer. Am J Pathol 1995;146:1161–9
Itoshima T, Fujiwara T, Waku T, et al. Induction of apoptosis in human esophageal cancer cells by sequential transfer of the wild-type p53 and E2F-1 genes: involvement of p53 accumulation via ARF-mediated MDM2 down-regulation. Clin Cancer Res 2000; 6:2851–2859
Boynton RF, Blount PL, Yin J, et al. Loss of heterozygosity involving the APC and MCC genetic loci occurs in the majority of human esophageal cancers. Proc Natl Acad Sci USA 1992; 89:3385–3388
Powell SM, Papadopoulos N, Kinzler KW, Smolinski KN, Meltzer SJ. APC gene mutations in the mutation cluster region are rare in esophageal cancers. Gastroenterology 1994; 107:1759–1763
Goessel G, Quante M, Hahn WC, et al. Creating oral squamous cancer cells: a cellular model of oral-esophageal carcinogenesis. Proc Natl Acad Sci USA 2005; 102:15599–15604
Marazuela M, Alonso MA. Expression of MAL and MAL2, two elements of the protein machinery for raft-mediated transport, in normal and neoplastic human tissue. Histol Histopathol 2004; 19:925–933
Martin-Belmonte F, Martinez-Menarguez JA, Aranda JF, Ballesta J, de Marco MC, Alonso MA. MAL regulates clathrin-mediated endocytosis at the apical surface of Madin-Darby canine kidney cells. J Cell Biol 2003; 163:155–164
Sasajima K, Taniguchi Y, Okazaki S, et al. Sequential morphological studies of the esophageal carcinoma of rats induced by N-methyl-N-amylnitrosamine. Eur J Cancer Clin Oncol 1982; 18:559–564
Mimori K, Kataoka A, Yoshinaga K, et al. Identification of molecular markers for metastasis-related genes in primary breast cancer cells. Clin Exp Metastasis 2005; 22:59–67
Mori M, Mimori K, Yoshikawa Y, et al. Analysis of the gene-expression profile regarding the progression of human gastric carcinoma. Surgery 2002; 131:S39–47
Mimori K, Ishii H, Nagahara H, et al. FHIT is up-regulated by inflammatory stimuli and inhibits prostaglandin E2-mediated cancer progression. Cancer Res 2006; 66:2683–2690
Buhler H, Schaller G. Transfection of keratin 18 gene in human breast cancer cells causes induction of adhesion proteins and dramatic regression of malignancy in vitro and in vivo. Mol Cancer Res 2005; 3:365–371
Reichelt J, Furstenberger G, Magin TM. Loss of keratin 10 leads to mitogen-activated protein kinase (MAPK) activation, increased keratinocyte turnover, and decreased tumor formation in mice. J Invest Dermatol 2004; 123:973–981
Santos M, Ballestin C., Garcia-Martin R, Jorcano JL. Delays in malignant tumor development in transgenic mice by forced epidermal keratin 10 expression in mouse skin carcinomas. Mol Carcinog 1997; 20:3–9
Schutte B, Henfling M, Kolgen W, et al. Keratin 8/18 breakdown and reorganization during apoptosis. Exp Cell Res 2004; 297:11–26
Caulin C, Salvesen GS, Oshima RG. Caspase cleavage of keratin 18 and reorganization of intermediate filaments during epithelial cell apoptosis. J Cell Biol 1997; 138:1379–94
Tognon C, Garnett M, Kenward E, Kay R, Morrison K, Sorensen PH. The chimeric protein tyrosine kinase ETV6-NTRK3 requires both Ras-Erk1/2 and PI3-kinase-Akt signaling for fibroblast transformation. Cancer Res 2001; 61:8909–8916
Cowling V, Downward J. Caspase-6 is the direct activator of caspase-8 in the cytochrome c-induced apoptosis pathway: absolute requirement for removal of caspase-6 prodomain. Cell Death Differ 2002; 9:1046–56
Nyormoi O, Wang Z, Bar-Eli M. Sequence-based discovery of a synthetic peptide inhibitor of caspase 6. Apoptosis 2003; 8:371–376
Banks-Schlegel SP, Harris CC. Tissue-specific expression of keratin proteins in human esophageal and epidermal epithelium and their cultured keratinocytes. Exp Cell Res 1983; 146:271–280
Viaene AI, Baert JH. Expression of cytokeratin mRNAs in normal human esophageal epithelium. Anat Rec 1995; 241:88–98
Preston SL, Wong WM, Chan AO, et al. Bottom-up histogenesis of colorectal adenomas: origin in the monocryptal adenoma and initial expansion by crypt fission. Cancer Res 2003;63:3819–25
Shih IM, Wang TL, Traverso G, et al. Top-down morphogenesis of colorectal tumors. Proc Natl Acad Sci USA 2001; 98:2640–2645
Acknowledgments
We thank Ms. M. Oda and Ms. T. Shimooka for their technical support and Dr. K. Obara for his advice for the regarding pathological information. We also appreciate the cooperation of Drs. Kosaka and Motoyama. Financial support: This work was supported by the following grant sponsors: CREST; Japan Science and Technology Agency (JST); Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research (Grant nos. 17109013, 17591411, 17591413, and 17015032).
Author information
Authors and Affiliations
Corresponding author
Additional information
This investigation was presented on March 25, 2006 at the 59th Annual Meeting of the Society of Surgical Oncology at the San Diego Marriott Hotel, San Diego, California.
Rights and permissions
About this article
Cite this article
Mimori, K., Nishida, K., Nakamura, Y. et al. Loss of MAL Expression in Precancerous Lesions of the Esophagus. Ann Surg Oncol 14, 1670–1677 (2007). https://doi.org/10.1245/s10434-006-9064-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-006-9064-2