Abstract
Background
Distal intramural spread is present within 1 cm from visible tumor in a substantial proportion of patients. Therefore, ≥1 cm of distal bowel clearance is recommended as minimally acceptable. However, clinical results are contradictory in answering the question of whether this rule is valid. The aim of this review was to evaluate whether in patients undergoing anterior resection, a distal bowel gross margin of <1 cm jeopardizes oncologic safety.
Methods
A systematic review of the literature identified 17 studies showing results in relation to margins of approximately <1 cm (948 patients) versus >1 cm (4626 patients); five studies in relation to a margin of ≤5 mm (173 patients) versus >5 mm (1277 patients), and five studies showing results in a margin of ≤2 mm (73 patients). In most studies, pre- or postoperative radiation was provided.
Results
A multifactorial process was identified resulting in selection of favorable tumors for anterior resection with the short bowel margin and unfavorable tumors for abdominoperineal resection or for anterior resection with the long margin. In total, the local recurrence rate was 1.0% higher in the <1-cm margin group compared to the >1-cm margin group (95% confidence interval [CI] −0.6 to 2.7; P = 0.175). The corresponding figures for ≤5 mm cutoff point were 1.7% (95% CI −1.9 to 5.3; P = 0.375). The pooled local recurrence rate in patients having ≤2 mm margin was 2.7% (95% CI 0 to 6.4).
Conclusions
In the selected group of patients, <1 cm margin did not jeopardize oncologic safety.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Subclinical distal bowel intramural spread is present within 1 cm distally from visible tumor in a substantial proportion of patients.1–5 For these reasons, for patients with low-lying cancer who are undergoing anterior resection (AR), ≥1 cm of distal bowel clearance is recommended as minimally acceptable.6 The 1-cm rule is occasionally violated, particularly after preoperative radiation.7–23 This is because preoperative chemoradiotherapy may lead to tumor regression and treatment of microscopic disease, facilitating complete tumor resection with a less than 1-cm bowel margin in patients who are otherwise candidates for abdominoperineal resection (APR).
For patients with rectal cancer, the adequacy of the distal margin is dependent on both the risk for intramural tumor spread and on the distal mesorectal lymphatic spread. Tumor cell deposits within mesorectal lymph nodes have been identified up to 5 cm distal to the inferior aspect of the tumor, emphasizing the need to adhere to the principles of total mesorectal excision and giving rise to the concept of tumor-specific mesorectal excision (mesorectal transection 5 cm distal to the inferior border of the tumor) for more proximal rectal cancers.24–26 In such circumstances, ensuring an adequate distal margin does not jeopardize the potential for sphincter preservation. However, for patients with low-lying tumors treated with total mesorectal excision, the primary concern in the absence of lateral or inguinal lymphatic metastases is distal intramural spread. Here the clinical evidence is less clear regarding what constitutes an adequate distal margin, particularly in the setting of neoadjuvant chemoradiotherapy.7–9
Therefore, the purpose of this study was to perform a systematic review of the impact of distal rectal margins of less than approximately 1 cm on oncologic outcomes after sphincter-preserving surgery for rectal cancer.
Materials and Methods
Studies qualified for this review if they separately described, for patients undergoing AR, the local control rates for patients whose distal gross bowel resection margins were shorter than or greater than approximately 1 cm. Only English-language studies were included. Studies were identified by an electronic search of the PubMed database with the keywords “rectal cancer” and “distal margin” or “distal clearance” with inclusion years from 1982 (the beginning of the total mesorectal excision era) through January 2011.27 The computerized search was supplemented with manual searches of reference lists of relevant articles. Full text of all pertinent studies were obtained. Data were independently extracted by two investigators using a data collection form. Patients with microscopically positive bowel margin in case of hand-sewn anastomosis and patients with microscopically positive stapler doughnuts were excluded. If the 5-year survival had not been provided in the text, the relevant figures were read from the survival curves. Disagreements were resolved by consensus by the reviewing authors. In case of a lack of relevant information in the reports published by our group, information was extracted from the original databases.5,9,20,23 In cases requiring additional clarification, the authors of the reports were contacted.
The literature search revealed that decisions of whether to precede to AR or to APR had been based not only on the distance between tumor and anal sphincter, but also on a variety of other clinical factors. This resulted in a selection of favorable tumors for AR with short margins and unfavorable tumors for APR or for AR with long margins.19,23,28,29 We therefore assessed the potential for treatment bias in a subcohort of patients to determine the frequency of intramural spread among patients undergoing AR versus APR. As a result of the inherent bias produced by a selection process that favored patients with short margins, the question of whether a short distal bowel margin per se jeopardizes oncologic safety cannot be answered. We hypothesized that a short distal bowel margin is not safe in spite of this selection.
Meta-analysis methodology was used to evaluate the issue in question by Meta-Analyst software.30 The significance of heterogeneity was tested by chi-square Q Cochran test using the fixed-effects model.
Results
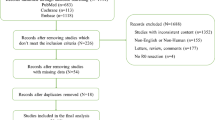
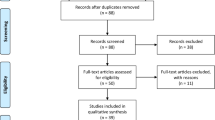
A total of 478 references were identified by the initial literature search within PubMed. Further manual search of the relevant references identified an additional 50 references. Of these, 439 abstracts were excluded because of lack of relevant data. Eighty-nine full-text articles were obtained and assessed for eligibility. Of these, 67 articles were excluded because of lack of relevant data and 5 because of duplication of data. The remaining 17 studies constituted the material for the current review.7–23 Only one report prospectively explored the issue in question as the secondary end point.20 The remaining reports were retrospective. Two reports gathered the data from multicentre randomized studies.17,20 The rest presented single-center data.
Outcomes in Relation to Distal Bowel Margin Shorter or Longer than 1 cm
Local recurrence rates in relation to the distal bowel margin shorter (number of patients, n = 948) versus longer than 1 cm (n = 4626) are presented in Table 1. In 11 reports pre- or postoperative radiotherapy or radiochemotherapy was used in all patients or in the substantial proportion of patients. In the remaining six studies all or the vast majority of patients underwent surgery alone. Two studies showed statistically significantly higher actuarial local recurrence rate in the short margin group compared to the long margin group.14,18 Cox multivariable analysis confirmed this finding in one study.14 In another study the difference was insignificant after correction for other variables.18 In one of these studies pre- or postoperative radiation was not given and in another one it was given in 4.4% of patients. In the remaining 15 studies the difference in local recurrence rate between the short margin group and to the long margin group was not significant in univariate analysis.7–13,15–17,19–23
Of the eight reports showing long-term survival, survival did not differ statistically between the two groups in seven; in one remaining study survival was lower in the short margin group compared to the long margin group (Table 1).
The results of the meta-analysis methodology used to evaluate all of the above reports showed that the observed local recurrence rate overall was 1.0% higher in the < 1-cm margin group compared to the > 1-cm margin group (95% confidence interval [CI] −0.6 to 2.7), P = 175 (Fig. 1). We further separately evaluated the local recurrence rates for the patients stratified by use of radiotherapy (i.e. subcohort of patients treated without or rarely treated with pre- or postoperative radiation or subcohort of patients treated with or majority treated with pre- or postoperative radiation) and observed a difference of 1.2% (95% CI −2.6 to 5.0), P = 0.323 and 0.9% (95% CI −0.8 to 2.7), P = 0.277, respectively (Fig. 1).
Anastomotic Recurrences in Relation to Distal Bowel Margin Shorter or Longer than 1 cm
We had hypothesized that anastomotic recurrences were likely due to inadequate distal bowel margins. Therefore, in six studies with relevant data, the rates of anastomotic failure were compared between the < 1-cm margin groups (n = 404) and the > 1-cm margin groups (n = 2079) (Table 2). In a series of patients not treated with pre- or postoperative radiation, more the anastomotic recurrences were recorded in the ≤ 0.8 cm margin group than in the > 0.8 cm margin group (P = 0.01) (Table 2).13 Similarly, another study, observed that distal bowel margin shorter that 0.8 cm was associated with higher 5-year actuarial anastomotic recurrence rate, 5% vs. 2% at 5 years, P < 0.001.7 However, in this series, in the subgroup of 19 patients with distal bowel margin of 1 mm, only one (5.3%) anastomotic recurrence occurred. Four remaining series showed no association between the length of bowel clearance and anastomotic recurrence.9,11,15,21 This was confirmed by multivariable analysis carried out in the largest series reported by Kim et al.21
The results of the meta-analysis methodology used to evaluate all of the above reports showed that the estimated overall anastomotic recurrence rate was 1.6% higher in the <1-cm margin group compared to the >1-cm margin group (95% CI −0.8 to 4.1) but this was not statistically significant (P = 0.221); test for heterogeneity, P = 0.97.
Outcomes in Relation to Gross Distal Bowel Margin Shorter or Equal to 5 mm Compared to Longer Margins
Local recurrence rates in relation to the gross distal bowel margin ≤ 5 mm (n = 173) compared to longer margin (n = 1277) were reported in five studies (Table 3).8,9,15,16,20 In none of these studies was the difference in local recurrence rate between the two groups statistically significant. In one study reported by Kiran et al., the multivariable analysis could not demonstrate an association.8 Survival did not differ between the two groups in any of the four studies reporting relevant data.
The results of the meta-analysis methodology used to evaluate the above reports showed that the overall local recurrence rate was 1.7% higher in the ≤5 mm margin group compared to the >5 mm margin group (95% CI −1.9 to 5.3), but this was not statistically significant (P = 0.375, Fig. 2).
We additionally examined outcomes in a subgroup of 73 patients from five studies with ultra-close microscopically negative distal margins of ≤2 mm (Table 4).7–9,16,20 Eligible patients included also those with positive gross bowel margin but with microscopically negative circular stapler tissue rings or with microscopically negative distal bowel margin in patients with hand-sewn coloanal anastomosis. The pooled analysis of these reports showed a local recurrence rate of 2.7% (95% CI 0 to 6.4). The rates of long-term survival did not seem much different than that observed in patients with larger margins (Tables 1 and 3).
Discussion
The current review shows that in a highly selected group of patients with favorable tumors, a distal gross rectal bowel resection margin less than 1 cm was not associated with differences in anastomotic recurrence, overall local control, or survival. No impairment in the oncologic safety was observed. We have additionally evaluated shorter distal margin thresholds of 5 mm or ultraclose negative margins and still observed no differences in local control or survival. Thus, the 1-cm rule for distal rectal cancers does not seem to be supported by the clinical evidence.
In the present review, the measurements of distal bowel margin were performed by pathologist on fresh tissue or on formalin-fixed pinned or unpinned specimens (Table 1), whereas the 1-cm rule often refers to margins as measured by surgeons on the fresh anatomically restored ex vivo condition.6 Because of the bowel shrinkage occurring during the first 10 to 20 minutes after removal from the patients and additionally shrinkage after fixation, a correction factor of 12% reduction in anatomically restored (pinned) fixed specimens and 50% reduction in nonrestored fixed specimens has been proposed.6,31 Because the measurements were based on histologic assessment during pathologic evaluation and not routinely by the surgeon’s operative findings, the results of this review should be considered as subject to this limitation. However, even if a correction factor as high as 50% was used to account for shrinkage of the distal margin, the favorable results found in patients with ≤ 5-mm or ≤ 2-mm margins may still call into question the 1-cm rule. Another potential source of bias is the heterogeneity of studies included in the review. This is reflected in proportions of patients in the < 1-cm margin groups in relation to the > 1-cm margin groups (Table 1). Similarly, there was a large variation between studies in the median distance between tumor and the anal verge (Table 1). Because of this heterogeneity, a result of single summary measure is highly problematic, although the meta-analysis methodology was used.32 Therefore, it cannot be viewed as a result of the proper meta-analysis.
During the analysis, we also observed that patients undergoing AR in the short margin groups were frequently described to have more favorable tumors than patients undergoing AR in the long margin groups or than patients undergoing APR. Patients undergoing AR with a short bowel clearance more likely had well or moderately differentiated tumor histology, early lesions, low carcinoembryonic antigen serum level or tumors downstaged or completely responding to preoperative radiation compared to those undergoing AR in the long margin groups or patients undergoing APR.7–9,12 Of note, the rules for the above patients’ selection were not predefined in any of the relevant articles. In this context, it is notable that long distal intramural spread is more often seen in unfavorable tumors compared to early or well-differentiated tumors.1,2,33 Furthermore, in some instances, the tumor regressive response to neoadjuvant chemoradiotherapy by intraoperative histopathologic evaluation may have influenced the decision of the surgeon to accept the shorter margin or to convert to APR. Thus, patients who underwent APR may have had longer distal intramural spread compared to those who underwent AR. We acknowledged this potentially important selection bias and separately analyzed clinical material of 192 patients previously published by our group.5 Distal intramural spread of ≥5 mm was more frequently observed in patients undergoing APR (23.4%, 18 of 77) than in those undergoing AR (12.2%, 14 of 115), P = 0.049 (two-sided chi-square test, Chmielik, unpublished data). These findings are consistent with existing pathologic data demonstrating the risk for distal intramural spread within 1 cm from visible tumor also in patients after chemoradiation and show the selection explaining why patients with < 5 mm bowel clearance have had low risk of local recurrence.1–3,5,16 Thus, the results of the present study should be considered with caution in light of this potential bias, and the short margins may be considered to be applicable to patients with low-risk tumors or good response to neoadjuvant therapy. Of note, it is not possible to adjust results for this bias.
In some studies, a clear distal stapler doughnuts were regarded as evidence of margin negative resection, even when distal bowel margin was microscopically positive. However, because the stapler doughnut may not include the entire circumference of distal rectal margin, with this policy, there is a risk of residual cancer cells in the rectal stump.
In patients receiving preoperative radiation, positive gross margin might be microscopically negative because residual fibrous tissue can mimic cancer (Table 4). On the other hand, on rare occasions, distal intramural spread up to 5 mm was reported below a residual ulceration in patients with a clinical complete response after chemoradiation.5 All of the above indicate that at surgery, the clinical evaluation of tumor response to preoperative radiation may not adequately represent what is happening at the microscopic level and the risk of distal intramural spread after radiation is difficult to predict. This may lead to a need for conversion to APR as a result of positive distal bowel margin. However, in clinical practice, it happens rarely, in approximately in 2% of patients in the subgroups with ≤1 cm distal bowel margin.9,20,21,34
In conclusion, our findings support the practice of sphincter preservation in selected settings of close distal margins (<1 cm) after total mesorectal excision for distal rectal cancer. Our review could not find a statistically significant difference in either local control or survival with margins of <1 cm. Further subgroup study suggests that margins as close as ≤5 mm—indeed negative—may be acceptable. However, the importance of patient and tumor selection for this approach must be emphasized. The precise rules for this selection have not been defined. Therefore, further study is needed to identify the criteria for selecting patients to an approach of close distal margins for sphincter preservation.
References
Williams NS, Dixon MF, Johnston D. Reappraisal of the 5 centimetre rule of distal excision for carcinoma of the rectum: a study of distal intramural spread and of patients’ survival. Br J Surg. 1983;70:150–4.
Shirouzu K, Isomoto H, Kakegawa T. Distal spread of rectal cancer and optimal distal margin of resection for sphincter-preserving surgery. Cancer. 1995;76:388–92.
Mezhir JJ, Smith KD, Fichera A, Hart J, Posner MC, Hurst RD. Presence of distal intramural spread after preoperative combined-modality therapy for adenocarcinoma of the rectum: what is now the appropriate distal resection margin? Surgery. 2005;138:658–63.
Kwok SP, Lau WY, Leung KL, Liew CT, Li AK. Prospective analysis of the distal margin of clearance in anterior resection for rectal carcinoma. Br J Surg. 1996;83:969–72.
Chmielik E, Bujko K, Nasierowska-Guttmejer A, et al. Distal intramural spread of rectal cancer after preoperative radiotherapy: the results of a multicenter randomized clinical study. Int J Radiat Oncol Biol Phys. 2006;65:182–8.
Nelson H, Petrelli N, Carlin A, et al. National Cancer Institute Expert Panel. Guidelines 2000 for colon and rectal cancer surgery. J Natl Cancer Inst. 2001;93:583–96.
Nash GM, Weiss A, Dasgupta R, Gonen M, Guillem JG, Wong WD. Close distal margin and rectal cancer recurrence after sphincter-preserving rectal resection. Dis Colon Rectum. 2010;53:1365–73.
Kiran RP, Lian I, Lavery IC. Does a subcentimeter distal resection margin adversely influence oncologic outcomes in patients with rectal cancer undergoing restorative proctectomy? Dis Colon Rectum. 2011;54:157–63.
Rutkowski A, Nowacki MP, Chwalinski M, et al. Acceptance of a 5 mm distal bowel resection margin for rectal cancer: is it safe? Colorectal Dis. 2010. doi:10.1111/j.1463-1318.2010.02542.x.
McDermott FT, Hughes ES, Pihl E, Johnson WR, Price AB. Local recurrence after potentially curative resection for rectal cancer in a series of 1008 patients. Br J Surg. 1985;72:34–7.
Hojo K. Anastomotic recurrence after sphincter-saving resection for rectal cancer. Length of distal clearance of the bowel. Dis Colon Rectum. 1986;29:11–4.
Karanjia ND, Schache DJ, North WR, Heald RJ. “Close shave” in anterior resection. Br J Surg. 1990;77:510–2.
Vernava AM 3rd, Moran M, Rothenberger DA, Wong WD. A prospective evaluation of distal margins in carcinoma of the rectum. Surg Gynecol Obstet. 1992;175:333–6.
Bokey EL, Ojerskog B, Chapuis PH, Dent OF, Newland RC, Sinclair G. Local recurrence after curative excision of the rectum for cancer without adjuvant therapy: role of total anatomical dissection. Br J Surg. 1999;86:1164–70.
Kuvshinoff B, Maghfoor I, Miedema B, et al. Distal margin requirements after preoperative chemoradiotherapy for distal rectal carcinomas: are < or = 1 cm distal margins sufficient? Ann Surg Oncol. 2001;8:163–9.
Andreola S, Leo E, Belli F, et al. Adenocarcinoma of the lower third of the rectum surgically treated with a <10-mm distal clearance: preliminary results in 35 N0 patients. Ann Surg Oncol. 2001;8:611–5.
Stocchi L, Nelson H, Sargent DJ, et al. North Central Cancer Treatment Group. Impact of surgical and pathologic variables in rectal cancer: a United States community and cooperative group report. J Clin Oncol. 2001;19:3895–902.
Law WL, Chu KW. Local recurrence following total mesorectal excision with double-stapling anastomosis for rectal cancers: analysis of risk factors. World J Surg. 2002;26:1272–6.
Huh JW, Jung EJ, Park YA, Lee KY, Sohn SK. Sphincter-preserving operations following preoperative chemoradiation: an alternative to abdominoperineal resection for lower rectal cancer? World J Surg. 2008;32:1116–23.
Rutkowski A, Bujko K, Nowacki MP, Chmielik E, Nasierowska-Guttmejer A, Wojnar A; Polish Colorectal Study Group. Distal bowel surgical margin shorter than 1 cm after preoperative radiation for rectal cancer: is it safe? Ann Surg Oncol. 2008;15:3124–31.
Kim YW, Kim NK, Min BS, et al. Factors associated with anastomotic recurrence after total mesorectal excision in rectal cancer patients. J Surg Oncol. 2009;99:58–64.
Pricolo VE, Abodeely A, Resnick M. Distal margins in radical resections for rectal cancer after chemoradiation therapy: how short is long enough? Dig Surg. 2010;27:185–9.
Silberfein EJ, Kattepogu KM, Hu CY, et al. Long-term survival and recurrence outcomes following surgery for distal rectal cancer. Ann Surg Oncol. 2010;17:2863–9.
Scott N, Jackson P, Al-Jaberi T, Dixon MF, Quirke P, Finan PJ. Total mesorectal excision and local recurrence: a study of tumour spread in the mesorectum distal to rectal cancer. Br J Surg. 1995;82:1031–3.
Hida J, Yasutomi M, Maruyama T, Fujimoto K, Uchida T, Okuno K. Lymph node metastases detected in the mesorectum distal to carcinoma of the rectum by the clearing method: justification of total mesorectal excision. J Am Coll Surg. 1997;184:584–8.
Reynolds JV, Joyce WP, Dolan J, Sheahan K, Hyland JM. Pathological evidence in support of total mesorectal excision in the management of rectal cancer. Br J Surg. 1996;83:1112–5.
Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery—the clue to pelvic recurrence? Br J Surg. 1982;69:613–66.
Weiser MR, Quah HM, Shia J, et al. Sphincter preservation in low rectal cancer is facilitated by preoperative chemoradiation and intersphincteric dissection. Ann Surg. 2009;249:236–42.
Temple LK, Romanus D, Niland J, et al. Factors associated with sphincter-preserving surgery for rectal cancer at national comprehensive cancer network centers. Ann Surg. 2009;250:260–7.
Meta-Analyst. http://tuftscaes.org/meta_analyst/. Accessed April 2011.
Goldstein NS, Soman A, Sacksner J. Disparate surgical margin lengths of colorectal resection specimens between in vivo and in vitro measurements. The effects of surgical resection and formalin fixation on organ shrinkage. Am J Clin Pathol. 1999;111:349–51.
Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–12.
Kinoshita H, Watanabe T, Yanagisawa A, Nagawa H, Kato Y, Muto T. Pathological changes of advanced lower-rectal cancer by preoperative radiotherapy. Hepatogastroenterology. 2004;51:1362–6.
Guillem JG, Chessin DB, Shia J, et al. A prospective pathologic analysis using whole-mount sections of rectal cancer following preoperative combined modality therapy: implications for sphincter preservation. Ann Surg. 2007;245:88–93.
Acknowledgment
We thank Dr. Garrett M. Nash and Dr. Nam-Kyu for the provision of the additional data. G.J.C. was supported in part by a research grant from the National Cancer Institute (K07-CA133187).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Bujko, K., Rutkowski, A., Chang, G.J. et al. Is the 1-cm Rule of Distal Bowel Resection Margin in Rectal Cancer Based on Clinical Evidence? A Systematic Review. Ann Surg Oncol 19, 801–808 (2012). https://doi.org/10.1245/s10434-011-2035-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-2035-2