Abstract
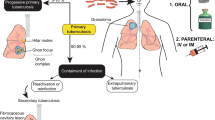
Tuberculosis (TB) is among the top 10 infectious diseases worldwide. It is categorized among the leading killer diseases that are the reason for the death of millions of people globally. Although a standardized treatment regimen is available, non-adherence to treatment has increased multi-drug resistance (MDR) and extensive drug-resistant (XDR) TB development. Another challenge is targeting the death of TB reservoirs in the alveoli via conventional treatment. TB Drug resistance may emerge as a futuristic restraint of TB with the scarcity of effective Anti-tubercular drugs. The paradigm change towards nano-targeted drug delivery systems is mostly due to the absence of effective therapy and increased TB infection recurrent episodes with MDR. The emerging field of nanotechnology gave an admirable opportunity to combat MDR and XDR via accurate diagnosis with effective treatment. The new strategies targeting the lung via the pulmonary route may overcome the new incidence of MDR and enhance patient compliance. Therefore, this review highlights the importance and recent research on pulmonary drug delivery with nanotechnology along with prevalence, the need for the development of nanotechnology, beneficial aspects of nanomedicine, safety concerns of nanocarriers, and clinical studies.
Graphical Abstract



Similar content being viewed by others
Data Availability
The data and materials used during the study are available from the corresponding author upon reasonable request.
Abbreviations
- TB:
-
Tuberculosis
- MDR:
-
Multi-drug resistance
- XDR:
-
Extensive drug-resistant
- NTEP:
-
National Tuberculosis Eradication Programme
- DOT:
-
Directly observed therapy
- HPβCD:
-
Hydroxypropyl-β-cyclodextrin
- RAMED:
-
Methylated beta-cyclodextrin
- Zn-RIF:
-
Zinc and rifampicin
- MIC:
-
Minimum inhibitory concentration
- BDQ:
-
Bedaquiline
- BTZLF: Labrafac:
-
1,3-Benzothiazin-4-one 043
References
Comas I, Coscolla M, Luo T, Borrell S, Holt KE, Kato-Maeda M, et al. Out-of-Africa migration and neolithic coexpansion of mycobacterium tuberculosis with modern humans. Nat Genet. 2013;45:1176–82.
Budha NR, Mehrotra N, Tangallapally R, Rakesh T, Qi J, Daniels AJ, et al. Pharmacokinetically-guided lead optimization of nitrofuranylamide anti-tuberculosis agents. AAPS J. 2008;10:157–65.
Zimmerman MR, Ph D. Pulmonary and osseous tuberculosis in an Egyptian mummy. J Urban Heal. 1977;55:604–8.
Wang C, Muttil P, Lu D, Beltran-Torres AA, Garcia-Contreras L, Hickey AJ. Screening for potential adjuvants administered by the pulmonary route for tuberculosis vaccines. AAPS J. 2009;11:139–47.
Los UMDECDE. WHO global tuberculosis report. 2022. Available at: https://www.who.int/publications/i/item/9789240061729. Accessed 25 Aug 2023.
World Health Organization. WHO | Global tuberculosis report 2019. WHO. 2020.
Tuberculosis [Internet]. 2021 [cited 2023 Aug 5]. Available from: https://www.who.int/news-room/fact-sheets/detail/tuberculosis.
Schildknecht KR, Pratt RH, Feng P-JI, Price SF, Self JL. Tuberculosis — United States, 2022. MMWR Morb Mortal Wkly Rep. 2023;72:297–303.
Thakur G, Thakur S, Thakur H. Status and challenges for tuberculosis control in India - Stakeholders’ perspective. Indian J Tuberc. 2021;68(3):334–9. https://doi.org/10.1016/j.ijtb.2020.10.001.
Central TB Division. India TB report 2022 coming together to end TB altogether. Minist Heal Fam Welf. 2022;1–263
Ramanujam H, Palaniyandi K. Bovine tuberculosis in India: the need for One Health approach and the way forward. One Heal. 2023;16: 100495.
Coowanitwong I, Arya V, Kulvanich P, Hochhaus G. Slow release formulations of inhaled rifampin. AAPS J. 2008;10:342–8.
What’s new in the TB section of the 2021 WHO model lists of essential medicines [Internet]. WHO. 2021 [cited 2023 Aug 14]. Available from: https://www.who.int/news/item/24-11-2021-what-s-new-in-the-tb-section-of-the-2021-who-model-lists-of-essential-medicines
Pagán AJ, Ramakrishnan L. Immunity and immunopathology in the tuberculous granuloma. Cold Spring Harb Perspect Med. 2015;5:1–19.
Tella JO, Adekoya JA, Ajanaku KO. Mesoporous silica nanocarriers as drug delivery systems for anti-tubercular agents: a review. R Soc Open Sci. 2022;9.
Chan HW, Chow S, Zhang X, Zhao Y, Tong HHY, Chow SF. Inhalable nanoparticle-based dry powder formulations for respiratory diseases: challenges and strategies for translational research. AAPS PharmSciTech 2023 244 [Internet]. 2023 [cited 2023 Aug 1];24:1–28. Available from: https://doi.org/10.1208/s12249-023-02559-y.
Padda IS, Muralidhara Reddy K. Antitubercular medications. StatPearls; 2020.
Ramappa V, Aithal GP. Hepatotoxicity related to anti-tuberculosis drugs: mechanisms and management. J Clin Exp Hepatol. 2013.
Song W, Si S. The rare ethambutol-induced optic neuropathy. Medicine (Baltimore). 2017;96.
De Jager R, Van Altena R. Hearing loss and nephrotoxicity in long-term aminoglycoside treatment in patients with tuberculosis. Int J Tuberc Lung Dis. 2002;6.
Cicero-Sabido R, Meléndez-Mier G. The beginnings of tuberculosis pharmacological treatment. Gac Med Mex. 2023;154.
Bidell MR, Lodise TP. Fluoroquinolone-associated tendinopathy: does levofloxacin pose the greatest risk? Pharmacotherapy. 2016.
Gaida R, Truter I, Peters CA. Adverse effects of bedaquiline in patients with extensively drug-resistant tuberculosis. South African J Infect Dis. 2020;35.
Vinarov Z, Abrahamsson B, Artursson P, Batchelor H, Berben P, Bernkop-Schnürch A, et al. Current challenges and future perspectives in oral absorption research: an opinion of the UNGAP network. Adv Drug Deliv Rev [Internet]. 2021 [cited 2023 Aug 19];171:289–331. Available from: https://pubmed.ncbi.nlm.nih.gov/33610694/
Wen H, Jung H, Li X. Drug delivery approaches in addressing clinical pharmacology-related issues: opportunities and challenges. AAPS J. 2015;17:1327–40.
Mishra B, Singh J. Novel drug delivery systems and significance in respiratory diseases. Target Chronic Inflamm Lung Dis Using Adv Drug Deliv Syst. 2020:57–95. https://doi.org/10.1016/b978-0-12-820658-4.00004-2
Huang Z, Kłodzińska SN, Wan F, Nielsen HM. Nanoparticle-mediated pulmonary drug delivery: state of the art towards efficient treatment of recalcitrant respiratory tract bacterial infections. Drug Deliv Transl Res. 2021. p. 1634–54.
Ghadiri M, Young PM, Traini D. Strategies to enhance drug absorption via nasal and pulmonary routes. Pharmaceutics. 2019;11:113.
Murgia X, De Souza CC, Lehr CM. Overcoming the pulmonary barrier: new insights to improve the efficiency of inhaled therapeutics. Eur J Nanomed. 2014;6:157–69.
Plaunt AJ, Nguyen TL, Corboz MR, Malinin VS, Cipolla DC. Strategies to overcome biological barriers associated with pulmonary drug delivery. Pharmaceutics. 2022.
Sahakijpijarn S, Smyth HDC, Miller DP, Weers JG. Post-inhalation cough with therapeutic aerosols: formulation considerations. Adv Drug Deliv Rev. 2020.
The mechanics of inhaled pharmaceutical aerosols. Mech. Inhaled Pharm. Aerosols. 2019.
Moon C, Smyth HDC, Watts AB, Williams RO. Delivery technologies for orally inhaled products: an update. AAPS PharmSciTech [Internet]. 2019 [cited 2023 Aug 19];20:117. Available from: https://pubmed.ncbi.nlm.nih.gov/30783904/
Liang W, Pan HW, Vllasaliu D, Lam JKW. Pulmonary delivery of biological drugs. Pharmaceutics [Internet]. 2020 [cited 2023 Aug 19];12:1–28. Available from: https://pubmed.ncbi.nlm.nih.gov/33114726/
Chan JGY, Wong J, Zhou QT, Leung SSY, Chan HK. Advances in device and formulation technologies for pulmonary drug delivery. AAPS PharmSciTech [Internet]. 2014 [cited 2023 Aug 19];15:882–97. Available from: https://pubmed.ncbi.nlm.nih.gov/24728868/
Morales JO, Fathe KR, Brunaugh A, Ferrati S, Li S, Montenegro-Nicolini M, et al. Challenges and future prospects for the delivery of biologics: oral mucosal, pulmonary, and transdermal routes. AAPS J [Internet]. 2017 [cited 2023 Jul 26];19:652–68. Available from: https://doi.org/10.1208/s12248-017-0054-z.
Wang W, Huang Z, Huang Y, Zhang X, Huang J, Cui Y, et al. Pulmonary delivery nanomedicines towards circumventing physiological barriers: strategies and characterization approaches. Adv Drug Deliv Rev. 2022;185: 114309.
Hassan MS, Lau RWM. Effect of particle shape on dry particle inhalation: study of flowability, aerosolization, and deposition properties. AAPS PharmSciTech [Internet]. 2009 [cited 2023 Aug 19];10:1252–62. Available from: https://pubmed.ncbi.nlm.nih.gov/19866362/
Hastedt JE, Bäckman P, Clark AR, Doub W, Hickey A, Hochhaus G, et al. Scope and relevance of a pulmonary biopharmaceutical classification system AAPS/FDA/USP Workshop March 16–17th, 2015 in Baltimore, MD. AAPS Open 2016 21 [Internet]. 2016 [cited 2023 Aug 19];2:1–20. Available from: https://doi.org/10.1186/s41120-015-0002-x.
Knudsen L, Ochs M. The micromechanics of lung alveoli: structure and function of surfactant and tissue components. Histochem Cell Biol. 2018;150:661–76.
Tan ZM, Lai GP, Pandey M, Srichana T, Pichika MR, Gorain B, Bhattamishra SK, Choudhury H. Novel approaches for the treatment of pulmonary tuberculosis. Pharmaceutics. 2020;12(12):1196. https://doi.org/10.3390/pharmaceutics12121196.
Marzuki OA, Fauzi ARM, Ayoub SKIM. Prevalence and risk factors of anti- tuberculosis drug -induced hepatitis in. Singapore Med J. 2008;49:688–93.
Yee D, Valiquette C, Pelletier M, Parisien I, Rocher I, Menzies D. Incidence of serious side effects from first-line antituberculosis drugs among patients treated for active tuberculosis. Am J Respir Crit Care Med. 2003;167:1472–7.
Mase SR, Chorba T. Treatment of drug-resistant tuberculosis. Clin Chest Med. 2019;40:775.
Gandhi NR, Moll A, Sturm AW, Pawinski R, Govender T, Lalloo U, et al. Extensively drug-resistant tuberculosis as a cause of death in patients co-infected with tuberculosis and HIV in a rural area of South Africa. Lancet. 2006;368:1575–80.
Lin H, Shin S, Blaya JA, Zhang Z, Cegielski P, Contreras C, et al. Assessing spatiotemporal patterns of multidrug-resistant and drug-sensitive tuberculosis in a South American setting. Epidemiol Infect. 2011;139:1784–93.
Chakraborty S, Rhee KY. Tuberculosis drug development: history and evolution of the mechanism-based paradigm. Cold Spring Harb Perspect Med. 2015;5:1–11.
Souza LLL, Santos FL dos, Crispim J de A, Fiorati RC, Dias S, Bruce ATI, et al. Causes of multidrug-resistant tuberculosis from the perspectives of health providers: challenges and strategies for adherence to treatment during the COVID-19 pandemic in Brazil. BMC Health Serv Res [Internet]. 2021 [cited 2023 Jul 27];21:1–10. Available from: https://doi.org/10.1186/s12913-021-07057-0.
Xi Y, Zhang W, Qiao RJ, Tang J. Risk factors for multidrug-resistant tuberculosis: a worldwide systematic review and meta-analysis. PLoS One [Internet]. 2022 [cited 2023 Aug 1];17:e0270003. Available from: https://doi.org/10.1371/journal.pone.0270003.
Seung KJ, Keshavjee S, Rich ML. Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis. Cold Spring Harb Perspect Med. 2015;5.
Demile B, Zenebu A, Shewaye H, Xia S, Guadie A. Risk factors associated with multidrug-resistant tuberculosis (MDR-TB) in a tertiary armed force referral and teaching hospital Ethiopia. BMC Infect Dis. 2018;18:249.
Ambaye GY, Tsegaye GW. Factors associated with multi-drug resistant tuberculosis among TB patients in selected treatment centers of Amhara region: a case-control study. Ethiop J Health Sci [Internet]. 2021 [cited 2023 Jul 27];31:34. Available from: https://www.pmc/articles/PMC8188099/
Jang JG, Chung JH. Diagnosis and treatment of multidrug-resistant tuberculosis. Yeungnam Univ J Med [Internet]. 2020 [cited 2023 Aug 1];37:277. Available from: https://www.pmc/articles/PMC7606956/
Nimmo C, Millard J, Faulkner V, Monteserin J, Pugh H, Johnson EO. Evolution of mycobacterium tuberculosis drug resistance in the genomic era. Front Cell Infect Microbiol. 2022.
Reva O, Korotetskiy I, Ilin A. Role of the horizontal gene exchange in evolution of pathogenic Mycobacteria. BMC Evol Biol. 2015;15.
Malhotra A, Kumar V, Juyal D, Gautam D, Malhotra R. Knowledge, attitude, and practices of health-care providers toward antibiotic prescribing, antibiotic resistance, and multidrug-resistant tuberculosis. Perspect Clin Res. 2021;12.
Braunstein M, Hickey AJ, Ekins S. Why wait? The case for treating tuberculosis with inhaled drugs. Pharm Res. 2019;36.
Kumar R, Mehta P, Shankar KR, Rajora MAK, Mishra YK, Mostafavi E, et al. Nanotechnology-assisted metered-dose inhalers (MDIs) for high-performance pulmonary drug delivery applications. Pharm Res 2022.
Kia P, Ruman U, Pratiwi AR, Hussein MZ. Innovative therapeutic approaches based on nanotechnology for the treatment and management of tuberculosis. Int J Nanomed. 2023.
Debnath SK, Debnath M, Srivastava R. Opportunistic etiological agents causing lung infections: emerging need to transform lung-targeted delivery. Heliyon. 2022.
Acosta MF, Abrahamson MD, Encinas-Basurto D, Fineman JR, Black SM, Mansour HM. Inhalable nanoparticles/microparticles of an AMPK and Nrf2 activator for targeted pulmonary drug delivery as dry powder inhalers. AAPS J [Internet]. 2021 [cited 2023 Jul 26];23:1–14. Available from: https://doi.org/10.1208/s12248-020-00531-3.
Nabi B, Rehman S, Aggarwal S, Baboota S, Ali J. Nano-based anti-tubercular drug delivery: an emerging paradigm for improved therapeutic intervention. Drug Deliv Transl Res [Internet]. 2020 [cited 2023 Aug 19];10:1111–21. Available from: https://pubmed.ncbi.nlm.nih.gov/32418158/
Khan FA, Salim MAH, du Cros P, Casas EC, Khamraev A, Sikhondze W, et al. Effectiveness and safety of standardised shorter regimens for multidrug-resistant tuberculosis: individual patient data and aggregate data meta-analyses. Eur Respir J. 2017;50:1–13.
Gelperina S, Kisich K, Iseman MD, Heifets L. The potential advantages of nanoparticle drug delivery systems in chemotherapy of tuberculosis. Am J Respir Crit Care Med. 2005;172:1487–90.
He S, Gui J, Xiong K, Chen M, Gao H, Fu Y. A roadmap to pulmonary delivery strategies for the treatment of infectious lung diseases. J Nanobiotechnol. 2022;20:1–23.
Ghodake V, Vishwakarma J, Vavilala SL, Patravale V. Cefoperazone sodium liposomal formulation to mitigate P. aeruginosa biofilm in cystic fibrosis infection: a QbD approach. Int J Pharm. 2020;587:119696.
Dhand C, Dwivedi N, Loh XJ, Jie Ying AN, Verma NK, Beuerman RW, et al. Methods and strategies for the synthesis of diverse nanoparticles and their applications: a comprehensive overview. RSC Adv. 2015.
Ang CW, Tan L, Qu Z, West NP, Cooper MA, Popat A, et al. Mesoporous silica nanoparticles improve oral delivery of antitubercular bicyclic nitroimidazoles. ACS Biomater Sci Eng. 2022;8.
Pinheiro M, Lcio M, José JLFC, Reis S. Liposomes as drug delivery systems for the treatment of TB. Nanomedicine. 2011;6:1413–28.
Hwang AA, Lee BY, Clemens DL, Dillon BJ, Zink JI, Horwitz MA. pH-responsive isoniazid-loaded nanoparticles markedly improvetuberculosis treatment in mice. HHS Public Access. 2015;11:5066.
Wang L, Hu C, Shao L. The antimicrobial activity of nanoparticles: present situation and prospects for the future. Int J Nanomed. 2017;12:1227–49.
Nasiruddin M, Neyaz MK, Das S. Nanotechnology-based approach in tuberculosis treatment. Tuberc Res Treat. 2017;2017:1–12.
Lecai J, Mijiti P, Chuangyue H, Qian G, Weiguo T, Jihong C. Treatment outcomes of multidrug-resistant tuberculosis patients receiving ambulatory treatment in Shenzhen, China: a retrospective cohort study. Front Public Heal. 2023;11:1134938.
Alsayed SSR, Gunosewoyo H. Tuberculosis: pathogenesis, current treatment regimens and new drug targets. Int J Mol Sci [Internet]. 2023 [cited 2023 Aug 1];24:5202. Available from: https://www.pmc/articles/PMC10049048/
Naderinezhad S, Amoabediny G, Haghiralsadat F. Co-delivery of hydrophilic and hydrophobic anticancer drugs using biocompatible pH-sensitive lipid-based nano-carriers for multidrug-resistant cancers. RSC Adv. 2017;7:30008–19.
Kole E, Jadhav K, Shirsath N, Dudhe P, Verma RK, Chatterjee A, et al. Nanotherapeutics for pulmonary drug delivery: an emerging approach to overcome respiratory diseases. J Drug Deliv Sci Technol. 2023;81: 104261.
Chaudhary KR, Puri V, Singh A, Singh C. A review on recent advances in nanomedicines for the treatment of pulmonary tuberculosis. J Drug Deliv Sci Technol. 2022;69: 103069.
Dhiman N, Awasthi R, Sharma B, Kharkwal H, Kulkarni GT. Lipid Nanoparticles as carriers for bioactive delivery. Front Chem. 2021;9.
Lu D, Garcia-Contreras L, Muttil P, Padilla D, Xu D, Liu J, et al. Pulmonary immunization using antigen 85-b polymeric microparticles to boost tuberculosis immunity. AAPS J. 2010;12:338–47.
Choudhury H, Gorain B, Pandey M, Khurana RK, Kesharwani P. Strategizing biodegradable polymeric nanoparticles to cross the biological barriers for cancer targeting. Int J Pharm. 2019;565.
ElKasabgy NA, Adel IM, Elmeligy MF. Respiratory tract: structure and attractions for drug delivery using dry powder inhalers. AAPS PharmSciTech [Internet]. 2020 [cited 2023 Aug 1];21:1–14. Available from: https://doi.org/10.1208/s12249-020-01757-2.
Newman SP. Drug delivery to the lungs: challenges and opportunities. Ther Deliv [Internet]. 2017 [cited 2023 Aug 1];8:647–61. Available from: https://doi.org/10.4155/tde-2017-0037.
Xu A, Zhu H, Gao B, Weng H, Ding Z, Li M, et al. Diagnosis of severe community-acquired pneumonia caused by Acinetobacter baumannii through next-generation sequencing: a case report. BMC Infect Dis [Internet]. 2020 [cited 2023 Aug 1];20:1–7. Available from: https://doi.org/10.1186/s12879-019-4733-5.
Zhuang X, Li L, Liu T, Zhang R, Yang P, Wang X, et al. Mechanisms of isoniazid and rifampicin-induced liver injury and the effects of natural medicinal ingredients: a review. Front Pharmacol. 2022;13:1037814.
Tewes F, Brillault J, Couet W, Olivier JC. Formulation of rifampicin-cyclodextrin complexes for lung nebulization. J Control Release. 2008;129:93–9.
Shao Z, Tai W, Qiu Y, Man RCH, Liao Q, Chow MYT, et al. Spray-dried powder formulation of capreomycin designed for inhaled tuberculosis therapy. Pharmaceutics. 2021;13:1–19.
Shao Z, Chow MYT, Chow SF, Lam JKW. Co-delivery of D-LAK antimicrobial peptide and capreomycin as inhaled powder formulation to combat drug-resistant tuberculosis. Pharm Res. 2023;1:14.
Verma S, Dal NJK, Srivastava A, Bharti R, Siva Reddy D V., Sofi HS, et al. Inhaled Adjunct therapy with second-line drug candidates for dose reduction in chemotherapeutic regimens for multi-drug-resistant tuberculosis. AAPS PharmSciTech [Internet]. 2023 [cited 2023 Aug 1];24:1–12. Available from: https://doi.org/10.1208/s12249-023-02585-w.
Ali M, Ahmed M, Hafiz S, Kamal M, Mumtaz M, Ayatollahi SA. Design, synthesis and antitubercular evaluation of novel series of pyrazinecarboxamide metal complexes. Iran J Pharm Res. 2018;17:93–9.
Chen CC, Chen YY, Yeh CC, Hsu CW, Yu SJ, Hsu CH, et al. Alginate-capped silver nanoparticles as a potent anti-mycobacterial agent against mycobacterium tuberculosis. Front Pharmacol. 2021;12.
Rudolph D, Redinger N, Schwarz K, Li F, Hädrich G, Cohrs M, et al. Amorphous drug nanoparticles for inhalation therapy of multidrug-resistant tuberculosis. ACS Nano [Internet]. 2023 [cited 2023 Aug 5];17:9478–86. Available from: https://doi.org/10.1021/acsnano.3c01664.
Sheikhpour M, Delorme V, Kasaeian A, Amiri V, Masoumi M, Sadeghinia M, et al. An effective nano drug delivery and combination therapy for the treatment of Tuberculosis. Sci Rep. 2022;12.
Petkar KC, Chavhan S, Kunda N, Saleem I, Somavarapu S, Taylor KMG, et al. Development of novel octanoyl chitosan nanoparticles for improved rifampicin pulmonary delivery: optimization by factorial design. AAPS PharmSciTech [Internet]. 2018 [cited 2023 Aug 19];19:1758–72. Available from: https://pubmed.ncbi.nlm.nih.gov/29589222/
Nemati E, Mokhtarzadeh A, Panahi-Azar V, Mohammadi A, Hamishehkar H, Mesgari-Abbasi M, et al. Ethambutol-loaded solid lipid nanoparticles as dry powder inhalable formulation for tuberculosis therapy. AAPS PharmSciTech [Internet]. 2019 [cited 2023 Aug 19];20:120. Available from: https://pubmed.ncbi.nlm.nih.gov/30796625/
Gairola A, Benjamin A, Weatherston JD, Cirillo JD, Wu HJ. Recent developments in drug delivery for treatment of tuberculosis by targeting macrophages. Adv Ther. 2022.
Pati R, Sahu R, Panda J, Sonawane A. Encapsulation of zinc-rifampicin complex into transferrin-conjugated silver quantum-dots improves its antimycobacterial activity and stability and facilitates drug delivery into macrophages. Sci Rep. 2016;6:1–14.
Marcianes P, Negro S, Barcia E, Montejo C, Fernández-Carballido A. Potential active targeting of gatifloxacin to macrophages by means of surface-modified PLGA microparticles destined to treat tuberculosis. AAPS PharmSciTech [Internet]. 2020 [cited 2023 Aug 1];21:1–14. Available from: https://doi.org/10.1208/s12249-019-1552-3.
Scolari IR, Páez PL, Sánchez-Borzone ME, Granero GE. Promising chitosan-coated alginate-tween 80 nanoparticles as rifampicin coadministered ascorbic acid delivery carrier against mycobacterium tuberculosis. AAPS PharmSciTech [Internet]. 2019 [cited 2023 Aug 19];20:67. Available from: https://pubmed.ncbi.nlm.nih.gov/30627867/
Ha-Young L, Myeong-Sik Y, Yu-Mi K, Sun-Kyung L, Eun-Mi H, Ji-Eun P, et al. Nanoparticles formed by encapsulating an anticancer drug into glycolchitosan-cholanic acid complex and a process for the preparation thereof [Internet]. Korea; 2011. Available from: https://patents.google.com/patent/KR101323102B1/en?oq=KR101323102B1
Guiney AE, Xia N, LI R, Hersam MC, D. N, Mansukhan iLinda. Identification and optimization of carbon radicals on hydrated graphene oxide for ubiquitous antibacterial coatings [Internet]. USA; 2021. Available from: https://patents.google.com/patent/US20220153589A1/en?q=(multidrug+resistant+%22tuberculosis%22%2C+Nano%2C+pulmonary)&oq=multidrug+resistant+%22tuberculosis%22%2C+Nano%2C+pulmonary
Andre Yi Nelfan, Liu MS. Mesoporous silica nanoparticles with lipid bilayer coating for cargo delivery [Internet]. Japan; 2017. Available from: https://patents.google.com/patent/JP7068173B2/en
Ahmad J, Akhter S, Rizwanullah M, Amin S, Rahman M, Ahmad MZ, et al. Nanotechnology-based inhalation treatments for lung cancer: state of the art. Nanotechnol Sci Appl. 2015;8:55–66.
Gaspar R, Duncan R. Polymeric carriers: preclinical safety and the regulatory implications for design and development of polymer therapeutics. Adv Drug Deliv Rev. 2009;61:1220–31.
Xi WS, Song ZM, Chen Z, Chen N, Yan GH, Gao Y, et al. Short-term and long-term toxicological effects of vanadium dioxide nanoparticles on A549 cells. Environ Sci Nano. 2019;6.
Cheng YS. Mechanisms of pharmaceutical aerosol deposition in the respiratory tract. AAPS PharmSciTech. 2014;15:630–40.
Morton WB. The settling of a suspension flowing along a tube. Proc R Irish Acad Sect A Math Phys Sci. 1935;43:1–4.
Patton JS, Byron PR. Inhaling medicines: delivering drugs to the body through the lungs. Nat Rev Drug Discov 2007 61. 2007;6:67–74.
Hofmann W. Modelling inhaled particle deposition in the human lung—a review. J Aerosol Sci. 2011;42:693–724.
Chaurasiya B, Zhao YY. Dry powder for pulmonary delivery: a comprehensive review. Pharmaceutics. 2021;13:1–28.
Nguyen TTL, Anton N, Vandamme TF. Oral pellets loaded with nanoemulsions. Nanostructures Oral Med. 2017;203–30.
Choudhury H, Pandey M, Gorain B, Chatterjee B, Madheswaran T, Md S, et al. Nanoemulsions as effective carriers for the treatment of lung cancer. Nanotechnology-Based Target Drug Deliv Syst Lung Cancer. 2019;217–47.
Maurya, Priyanka, Singh S, Saraf SA. Inhalable hybrid nanocarriers for respiratory disorders. Target Chronic Inflamm Lung Dis Using Adv Drug Deliv Syst [Internet]. 2020;281–302. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7499343/
Hadiwinoto GD, Kwok PCL, Lakerveld R. A review on recent technologies for the manufacture of pulmonary drugs. Ther Deliv. 2017;9:47–70.
Ng AW, Bidani A, Heming TA. Innate host defense of the lung: effects of lung-lining fluid pH. Lung. 2004;182:297–317.
Fahy JV, Dickey BF. Airway mucus function and dysfunction. N Engl J Med. 2010;363:2233.
Roy R, Tiwari M, Donelli G, Tiwari V. Strategies for combating bacterial biofilms: a focus on anti-biofilm agents and their mechanisms of action. Virulence. 2018;9:522–54.
El-Sherbiny IM, El-Baz NM, Yacoub MH. Inhaled nano- and microparticles for drug delivery. Glob Cardiol Sci Pract. 2015;2015.
Wiedmann TS, Bhatia R, Wattenberg LW. Drug solubilization in lung surfactant. J Control Release. 2000;65:43–7.
Arora S, Jain J, Rajwade JM, Paknikar KM. Cellular responses induced by silver nanoparticles: in vitro studies. Toxicol Lett. 2008;179:93–100.
Sung JH, Ji JH, Yoon JU, Kim DS, Song MY, Jeong J, et al. Lung function changes in Sprague-Dawley rats after prolonged inhalation exposure to silver nanoparticles. Inhal Toxicol. 2008;20:567–74.
Niwa Y, Hiura Y, Sawamura H, Iwai N. Inhalation exposure to carbon black induces inflammatory response in rats. Circ J. 2008;72:144–9.
Lundborg M, Bouhafs R, Gerde P, Ewing P, Camner P, Dahlén SE, et al. Aggregates of ultrafine particles modulate lipid peroxidation and bacterial killing by alveolar macrophages. Environ Res. 2007;104:250–7.
Beck-Broichsitter M, Ruppert C, Schmehl T, Guenther A, Betz T, Bakowsky U, et al. Biophysical investigation of pulmonary surfactant surface properties upon contact with polymeric nanoparticles in vitro. Nanomed Nanotechnol Biol Med. 2011;7:341–50.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study. The original draft was prepared by DS, P, SN, SO, and MP. Conceptualization, supervision, reviewing, and editing were done by MP and TK. Reviewing and editing were done by NM, NJ, and PK. Adding resources, viz., figures, was done by NJ.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This is only an observational and theoretical study. No ethical approval is required.
Consent for Publication
The authors give their consent for the publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, D., Pooja, Nirban, S. et al. Nano vs Resistant Tuberculosis: Taking the Lung Route. AAPS PharmSciTech 24, 252 (2023). https://doi.org/10.1208/s12249-023-02708-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-023-02708-3