Abstract
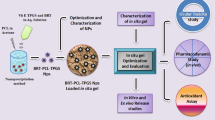
Brinzolamide is an effective carbonic anhydrase inhibitor widely used in glaucoma therapy but limits its application due to inadequate aqueous solubility and permeability. The aim of the present research work is the development and characterization of brinzolamide-loaded ultradeformable bilosomes to enhance the corneal permeation of the drug. These ultradeformable bilosomes were prepared by ethanol injection method and evaluated for physicochemical properties, particle size, morphology, drug release, ultra-deformability, corneal permeation, and irritation potential. The optimized formulation exhibited an average particle size of 205.4 ± 2.04 nm with mono-dispersity (0.109 ± 0.002) and showed entrapment efficiency of 75.02 ± 0.017%, deformability index of 3.91, and release the drug in a sustained manner. The brinzolamide-loaded ultradeformable bilosomes released 76.29 ± 3.77% of the drug in 10 h that is 2.25 times higher than the free drug solution. The bilosomes were found non-irritant to eyes with a potential irritancy score of 0 in Hen’s egg-chorioallantoic membrane assay. Brinzolamide-loaded ultradeformable bilosomes showed 83.09 ± 5.1% of permeation in 6 h and trans-corneal permeability of 8.78 ± 0.14 cm/h during the ex vivo permeation study. The acquired findings clearly revealed that the brinzolamide-loaded ultradeformable bilosomes show promising output and are useful in glaucoma therapy.
Graphical Abstract









Similar content being viewed by others
Data Availability
Data will be available based upon request.
References
Parsadaniantz SM, Goazigo AR, Sapienza A, Habas C, Baudouin C. Glaucoma : a degenerative optic neuropathy related to neuroinflammation? Cells. 2020;9:1–14. https://doi.org/10.3390/cells9030535.
Raghu PK, Bansal KK, Thakor P, Bhavana V, Madan J, Rosenholm JM, et al. Evolution of nanotechnology in delivering drugs to eyes, skin and wounds via topical route. Pharmaceuticals. 2020;13:1–30. https://doi.org/10.3390/ph13080167.
Allison K, Patel D, Alabi O. Epidemiology of glaucoma : the past, present, and predictions for the future. Cureus. 2020;12:1–9. https://doi.org/10.7759/cureus.11686.
Pascolini D, Mariotti SP. Global estimates of visual impairment: 2010. Br J Ophthalmol. 2012;96:614–8. https://doi.org/10.1136/bjophthalmol-2011-300539.
Mcmonnies CW. Glaucoma history and risk factors. J Optom. 2017;10:71–8. https://doi.org/10.1016/j.optom.2016.02.003.
Cheng KJ, Hsieh CM, Nepali K, Liou JP. Ocular disease therapeutics: design and delivery of drugs for diseases of the eye. J Med Chem. 2020;63:10533–93. https://doi.org/10.1021/acs.jmedchem.9b01033.
Noecker RJ. The management of glaucoma and intraocular hypertension : current approaches and recent advances. Ther Clin Risk Manag. 2006;2:193–206. https://doi.org/10.2147/tcrm.2006.2.2.193.
Tiwari R, Sethiya NK, Gulbake AS, Mehra NK, Murty USN, Gulbake A. A review on albumin as a biomaterial for ocular drug delivery. Int J Biol Macromol. 2021;191:591–9. https://doi.org/10.1016/j.ijbiomac.2021.09.112.
DeSantis L. Preclinical overview of brinzolamide. Surv Ophthalmol. 2000;44:119–29. https://doi.org/10.1016/S0039-6257(99)00108-3.
Naageshwaran V, Ranta V, Gum G. Comprehensive ocular and systemic pharmacokinetics of brinzolamide in rabbits after intracameral, topical, and intravenous administration. J Pharm Sci 2020:1–7. https://doi.org/10.1016/j.xphs.2020.09.051.
Shah P, Thakkar V, Anjana V, Christian J, Trivedi R, Patel K. Exploring of Taguchi design in the optimization of brinzolamide and timolol maleate ophthalmic in-situ gel used in treatment of glaucoma. Curr Drug Ther. 2020;15:1–19. https://doi.org/10.2174/1574885514666190916151506.
Srivastava V, Chary PS, Rajana N, Pardhi ER, Singh V, Khatri D, et al. Complex ophthalmic formulation technologies: advancement and future perspectives. J Drug Deliv Sci Technol. 2023;82:1–22. https://doi.org/10.1016/j.jddst.2023.104317.
Gorantla S, Rapalli VK, Waghule T, Singh PP, Dubey SK, Saha RN, et al. Nanocarriers for ocular drug delivery: current status and translational opportunity. R Soc Chem Adv. 2020;46:27835–55. https://doi.org/10.1039/d0ra04971a.
Bachu RD, Chowdhury P, Al-Saedi ZHF, Karla PK, Boddu SHS. Ocular drug delivery barriers—role of nanocarriers in the treatment of anterior segment ocular diseases. Pharmaceutics. 2018;10:1–31. https://doi.org/10.3390/pharmaceutics10010028.
Ten ZJ. years of knowledge of nano-carrier based drug delivery systems in ophthalmology: current evidence, challenges, and future prospective. Int J Nanomedicine. 2021;16:6497–530. https://doi.org/10.2147/IJN.S329831.
Mehra NK, Cai D, Kuo L, Hein T, Palakurthi S. Safety and toxicity of nanomaterials for ocular drug delivery applications. Nanotoxicology. 2016;10:836–60. https://doi.org/10.3109/17435390.2016.1153165.
Rajput T, Chauhan MK. Bilosome: a bile salt based novel carrier system gaining interest in pharmaceutical research. J Drug Deliv Ther. 2022;7:4–16. https://doi.org/10.22270/jddt.v7i5.1479.
Conacher M, Alexander J, Brewer JM. Oral immunisation with peptide and protein antigens by formulation in lipid vesicles incorporating bile salts (bilosomes). Vaccine. 2001;19:2965–74. https://doi.org/10.1016/S0264-410X(00)00537-5.
Palekar-shanbhag P, Lande S, Chandra R, Rane D. Bilosomes: superior vesicular carriers. Curr Drug Ther. 2020;15:312–20. https://doi.org/10.2174/1574885514666190917145510.
Abdelbary AA, Abd-elsalam WH, Al-mahallawi AM. Fabrication of novel ultradeformable bilosomes for enhanced ocular delivery of terconazole: in vitro characterization, ex vivo permeation and in vivo safety assessment. Int J Pharm. 2016;513:688–96. https://doi.org/10.1016/j.ijpharm.2016.10.006.
Saifi Z, Rizwanullah M, Mir SR, Amin S. Bilosomes nanocarriers for improved oral bioavailability of acyclovir: a complete characterization through in vitro, ex-vivo and in vivo assessment. J Drug Deliv Sci Technol. 2020;57:2–13. https://doi.org/10.1016/j.jddst.2020.101634.
Bhosale VA, Srivastava V, Valamla B, Yadav R, Singh SB, Mehra NK. Preparation and evaluation of modified chitosan nanoparticles using anionic sodium alginate polymer for treatment of ocular disease. Pharmaceutics. 2022;14:1–20. https://doi.org/10.3390/pharmaceutics14122802.
Balakrishna T, Shirsat MK, Amol K. Analytical method development and validation for the determination of Brinzolamide by RP-HPLC. J Drug Deliv Ther. 2020;10:92–6. https://doi.org/10.22270/jddt.v10i1.3845.
Dubey V, Mohan P, Dangi JS, Kesavan K. Brinzolamide loaded chitosan-pectin mucoadhesive nanocapsules for management of glaucoma: formulation, characterization and pharmacodynamic study. Int J Biol Macromol. 2019;19:1–40. https://doi.org/10.1016/j.ijbiomac.2019.10.219.
Abdelrahman EF, Elsayed I, Gad MK, Elshafeey AH, Mohamed IM. Response surface optimization, ex vivo and in vivo investigation of nasal spanlastics for bioavailability enhancement and brain targeting of risperidone. Int J Pharm. 2017;17:2–42. https://doi.org/10.1016/j.ijpharm.2017.07.050.
Zhu X, Zhang N, Lin W, Tang C. Food Hydrocolloids freeze-thaw stability of pickering emulsions stabilized by soy and whey protein particles. Food Hydrocoll. 2017;69:173–84. https://doi.org/10.1016/j.foodhyd.2017.02.001.
Cheng Y, Qin H, Acevedo NC, Jiang X, Shi X. 3D printing of extended-release tablets of theophylline using hydroxypropyl methylcellulose (HPMC) hydrogels. Int J Pharm. 2020;591:1–55. https://doi.org/10.1016/j.ijpharm.2020.119983.
Jain K, Verma AK, Mishra PR, Jain NK. Characterization and evaluation of amphotericin B loaded MDP conjugated poly(propylene imine) dendrimers. Nanomed Nanotechnol Biol Med. 2015;11:705–13. https://doi.org/10.1016/j.nano.2014.11.008.
Mohsen AM, Salama A, Kassem AA. Development of acetazolamide loaded bilosomes for improved ocular delivery: preparation, characterization and in vivo evaluation. J Drug Deliv Sci Technol. 2020;59:1–16. https://doi.org/10.1016/j.jddst.2020.101910.
Spielmann H. HET-CAM test. Methods Mol Biol. 1997;43:199–204. https://doi.org/10.1385/0-89603-282-5:199.
ICCVAM-recommended test method protocol: Hen’s egg test – chorioallantoic membrane (HET-CAM) test method 2010:3–30. https://ntp.niehs.nih.gov/iccvam/docs/ocutox_docs/invitro-2010/tmer-vol1.pdf. Accessed 12 Jan 2022.
Gade S, Patel KK, Gupta C, Anjum M, Deepika D, Agrawal AK, et al. An ex vivo evaluation of moxifloxacin nanostructured lipid carrier enriched in situ gel for transcorneal permeation on goat cornea. J Pharm Sci. 2019:1–12. https://doi.org/10.1016/j.xphs.2019.04.005.
Janga KY, Tatke A, Balguri SP, Surya P, Ibrahim MM, Maria DN, et al. Ion-sensitive in situ hydrogels of natamycin bilosomes for enhanced and prolonged ocular pharmacotherapy: in vitro permeability, cytotoxicity and in vivo evaluation. Artif Cells Nanomed Biotechnol. 2018;2:1–12. https://doi.org/10.1080/21691401.2018.1443117.
Farghaly DA, Aboelwafa AA, Hamza MY, Mohamed MI. Topical delivery of fenoprofen calcium via elastic nano-vesicular spanlastics: optimization using Experimental Design and In Vivo Evaluation. AAPS PharmSciTech. 2017;24:1–12. https://doi.org/10.1208/s12249-017-0771-8.
Zafar A, Alruwaili NK, Sarim S, Hadal N, Saad K, Afzal M, et al. Bioactive apigenin loaded oral nano bilosomes: formulation optimization to preclinical assessment. Saudi Pharm J. 2021;29:269–79. https://doi.org/10.1016/j.jsps.2021.02.003.
Vargas A, Zeisser-labouèbe M, Lange N, Gurny R, Delie F. The chick embryo and its chorioallantoic membrane (CAM) for the in vivo evaluation of drug delivery systems ☆. Adv Drug Deliv Rev. 2007;59:1162–76. https://doi.org/10.1016/j.addr.2007.04.019.
Dai Y, Zhaou R, Liu L, Lu Y, Qi J, Wu W. Liposomes containing bile salts as novel ocular delivery systems for tacrolimus (FK506): in vitro characterization and improved corneal permeation. Int J Nanomedicine. 2013;8:1921–33. https://doi.org/10.2147/IJN.S44487.
Gaballa SA, Kompella UB, Elgarhy O, Alqahtani AM, Pierscionek B, Alany RG, et al. Corticosteroids in ophthalmology: drug delivery innovations, pharmacology, clinical applications, and future perspectives. Drug Deliv Transl Res. 2021;11:866–93. https://doi.org/10.1007/s13346-020-00843-z.
Di PG, Bongiovì F, Palumbo FS, Pitarresi G, Licciardi M, Giammona G. Mucoadhesive PEGylated inulin-based self-assembling nanoparticles: in vitro and ex vivo transcorneal permeation enhancement of corticosteroids. J Drug Deliv Sci Technol. 2019;49:195–208. https://doi.org/10.1016/j.jddst.2018.10.028.
Acknowledgements
The authors would like to acknowledge the Department of Pharmaceutics, National Institute of Pharmaceutical Education & Research (NIPER), Hyderabad (Ministry of Chemical and Fertilizers, India) for providing extended facilities during this manuscript (manuscript communication no.: NIPER-H/2022).
Funding
The author (NKM) received a research grant from the Department of Science and Technology (DST), New Delhi (F. No. DST/NM/NS/2021/405) under the nanomission scheme.
Author information
Authors and Affiliations
Contributions
Vishnumaya S. Nair and Vaibhavi Srivastava: investigation, data curation, visualization, and original draft writing.
Vishnumaya S. Nair and Neelesh Kumar Mehra: design and conceptualization.
Valamla Bhawana and Rati Yadav: reviewing, editing, and supervision.
N. K. Mehra and Shashi Bala Singh: supervision.
N. K. Mehra: guidance, acquired funding and approval of the original draft.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nair, V.S., Srivastava, V., Bhavana, V. et al. Exploring Penetration Ability of Carbonic Anhydrase Inhibitor–Loaded Ultradeformable Bilosome for Effective Ocular Application. AAPS PharmSciTech 24, 157 (2023). https://doi.org/10.1208/s12249-023-02617-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-023-02617-5