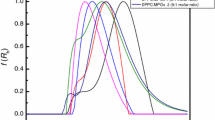
Abstract
Liposomes composed of soy lecithin (SL) have been studied widely for drug delivery applications. The stability and elasticity of liposomal vesicles are improved by incorporating additives, including edge activators. In this study, we report the effect of sodium taurodeoxycholate (STDC, a bile salt) upon the microstructural characteristics of SL vesicles. Liposomes, prepared by the thin film hydration method, were characterized by dynamic light scattering (DLS), small-angle neutron scattering (SANS), electron microscopy, and rheological techniques. We noticed a reduction in the size of vesicles with the incremental addition of STDC. Initial changes in the size of spherical vesicles were ascribed to the edge-activating action of STDC (0.05 to 0.17 µM). At higher concentrations (0.23 to 0.27 µM), these vesicles transformed into cylindrical structures. Morphological transitions at higher STDC concentrations would have occurred due to its hydrophobic interaction with SL molecules in the bilayer. This was ascertained from nuclear magnetic resonance observations. Whereas shape transitions underscored the deformability of vesicles in the presence of STDC, the consistency of bilayer thickness ruled out any dissociative effect. It was interesting to notice that SL-STDC mixed structures could survive high thermal stress, electrolyte addition, and dilution.
Graphical Abstract










Similar content being viewed by others
Data Availability
Data will be made available on request.
Abbreviations
- SL:
-
Soy lecithin
- STDC:
-
Sodium taurodeoxycholate
- MLV:
-
Multilamellar vesicles
- UL:
-
Unilamellar vesicles
- NMR:
-
Nuclear magnetic resonance
- HR-TEM:
-
High-resolution transmission electron microscopy
- PDI:
-
Polydispersity index
- Dh :
-
Hydrodynamic diameter
- PEG:
-
Polyethylene glycol
- DLS:
-
Dynamic light scattering
- SANS:
-
Small angle neutron scattering
References
Rehman AU, Omran Z, Anton H, Mely Y, Akram S, Vandamme TF, et al. Development of doxorubicin hydrochloride loaded pH-sensitive liposomes: investigation on the impact of chemical nature of lipids and liposome composition on pH-sensitivity. Eur J Pharm Biopharm. 2018;133:331–8.
Lu B, Ma Q, Zhang J, Liu R, Yue Z, Xu C, et al. Preparation and characterization of bupivacaine multivesicular liposome: a QbD study about the effects of formulation and process on critical quality attributes. Int J Pharm. 2021;598:120335.
Guimaraes D, Cavaco-Paulo A, Nogueira E. Design of liposomes as drug delivery system for therapeutic applications. Int J Pharm. 2021;601:120571.
Wang C, Xiao J, Hu X, Liu Q, Zheng Y, Kang Z, et al. Liquid core nanoparticle with high deformability enables efficient penetration across biological barriers. Adv Healthc Mater. 2023;12:2201889.
Li S, Goins B, Zhang L, Bao A. Novel multifunctional theranostic liposome drug delivery system: construction, characterization, and multimodality MR, near-infrared fluorescent, and nuclear imaging. Bioconjug Chem. 2012;23:1322–32.
Celia C, Cristiano MC, Froiio F, Di Francesco M, d’Avanzo N, Di Marzio L, et al. Nanoliposomes as multidrug carrier of gemcitabine/paclitaxel for the effective treatment of metastatic breast cancer disease: a comparison with gemzar and taxol. Adv Therap. 2021;4:2000121.
Xu S, Zhang P, Heing-Becker I, Zhang J, Tang P, Bej R, et al. Dual tumor-and subcellular-targeted photodynamic therapy using glucose-functionalized MoS2 nanoflakes for multidrug-resistant tumor ablation. Biomaterials. 2022;290:121844.
Crommelin DJA, Van Hoogevest P, Storm G. The role of liposomes in clinical nanomedicine development. What now? Now what? J Control Release. 2020;318:256–63.
Szabova J, Misik O, Havlikova M, Lizal F, Mravec F. Influence of liposomes composition on their stability during the nebulization process by vibrating mesh nebulizer. Colloids Surf B Biointerfaces. 2021;204: 111793.
Awad NS, Paul V, Mahmoud MS, Al Sawaftah NM, Kawak PS, Al Sayah MH, et al. Effect of pegylation and targeting moieties on the ultrasound-mediated drug release from liposomes. ACS Biomater Sci Eng. 2020;6:48–57.
Sudhakar K, Mishra V, Jain S, Rompicherla NC, Malviya N, Tambuwala MM. Development and evaluation of the effect of ethanol and surfactant in vesicular carriers on lamivudine permeation through the skin. Int J Pharm. 2021;610:121226.
Barone A, Cristiano MC, Cilurzo F, Locatelli M, Iannotta D, Di Marzio L, et al. Ammoniumglycyrrhizate skin delivery from ultradeformable liposomes: a novel use as anti-inflammatory agent in topical drug delivery. Colloids Surf B Biointerfaces. 2020;193: 111152.
Liang X, Mao G, Ng KYS. Mechanical properties and stability measurement of cholesterol-containing liposome on mica by atomic force microscopy. J Colloid Interface Sci. 2004;278:53–62.
Trilli J, Caramazza L, Paolicelli P, Casadei MA, Liberti M, Apollonio F, et al. The impact of bilayer rigidity on the release from magnetoliposomes vesicles controlled by PEMFs. Pharmaceutics.2021;13:1712.
Benne N, Leboux RJT, Glandrup M, van Duijn J, Lozano Vigario F, Neustrup MA, et al. Atomic force microscopy measurements of anionic liposomes reveal the effect of liposomal rigidity on antigen-specific regulatory T cell responses. J Control Release. 2020;318:246–55.
Mouritsen OG, Jorgensen K. Dynamical order and disorder in lipid bilayers. Chem Phys Lipids. 1994;73:3–25.
Lim E-B, Haam S, Lee S-W. Sphingomyelin-based liposomes with different cholesterol contents andpolydopamine coating as a controlled delivery system. Colloids Surf A Physicochem Eng Asp. 2021;618:126447.
Cui M, Wu W, Hovgaard L, Lu Y, Chen D, Qi J. Liposomes containing cholesterol analogues of botanical origin as drug delivery systems to enhance the oral absorption of insulin. Int J Pharm. 2015;489:277–84.
Ding WX, Qi XR, Li P, Maitani Y, Nagai T. Cholesteryl hemisuccinate as a membrane stabilizer in dipalmitoylphosphatidylcholine liposomes containing saikosaponin-d. Int J Pharm. 2005;300:38–47.
Van den Bergh BA, Wertz PW, Junginger HE, Bouwstra JA. Elasticity of vesicles assessed by electronspin resonance, electron microscopy and extrusion measurements. Int J Pharm. 2001;217:13–24.
Zhang S, Wang X. Effect of vesicle-to-micelle transition on the interactions of phospholipid/sodium cholate mixed systems with curcumin in aqueous solution. J Phy Chem B. 2016;120:7392–400.
Rathod S, Joshi A, Ray D, Aswal VK, Verma G, Bahadur P, et al. Changes in aggregation properties of TPGS micelles in the presence of sodium cholate. Colloids Surf A Physicochem Eng Asp. 2021;610:125938.
Niu M, Lu Y, Hovgaard L, Guan P, Tan Y, Lian R, et al. Hypoglycemic activity and oral bioavailability of insulin-loaded liposomes containing bile salts in rats: the effect of cholate type, particle size and administered dose. Eur J Pharm Biopharm. 2012;81:265–72.
Kafle A, Akamatsu M, Bhadani A, Sakai K, Kaise C, Kaneko T, et al. Phase behavior of the bilayers containing hydrogenated soy lecithin and β-sitosteryl sulfate. Langmuir. 2020;36:6025–32.
Zhang X, Lei B, Wang Y, Xu S, Liu H. Dual-sensitive on–off switch in liposome bilayer for controllable drug release. Langmuir. 2019;35:5213–20.
Touti R, Noun M, Guimberteau F, Lecomte S, Faure C. What is the fate of multi-lamellar liposomes of controlled size, charge and elasticity in artificial and animal skin? Eur J Pharm Biopharm. 2020;151:18–31.
Lichtenberg D, Ahyayauch H, Goni FM. The mechanism of detergent solubilization of lipid bilayers. Biophys J. 2013;105:289–99.
Lee EH, Kim A, Oh Y-K, Kim C-K. Effect of edge activators on the formation and transfection efficiency of ultradeformable liposomes. Biomaterials. 2005;26:205–10.
Zhang Y, Shen L, Zhang K, Guo T, Zhao J, Li N, et al. Enhanced antioxidation via encapsulation of isooctyl p-methoxycinnamate with sodium deoxycholate-mediated liposome endocytosis. Int J Pharm. 2015;496:392–400.
Mohapatra M, Mishra AK. Effect of submicellar concentrations of conjugated and unconjugated bile salts on the lipid bilayer membrane. Langmuir. 2011;27:13461–7.
Rathod S, Arya S, Shukla R, Ray D, Aswal VK, Bahadur P, et al. Investigations on the role of edge activator upon structural transitions in span vesicles. Colloids Surfaces A Physicochem Eng Asp. 2021;627:127246.
Debye P, Anacker EW. Micelle shape from dissymmetry measurements. J Phys Chem. 1951;55:644–55.
Agrawal NR, Omarova M, Burni F, John VT, Raghavan SR. Spontaneous formation of stable vesicles and vesicle gels in polar organic solvents. Langmuir. 2021;37:7955–65.
Yang L, Tucker IG, Ostergaard J. Effects of bile salts on propranolol distribution into liposomes studied by capillary electrophoresis. J Pharm Biomed Anal. 2011;56:553–9.
Prameela G, Kumar BP, Reddy RR, Pan A, Subramanian J, Kumar S, et al. Vesicle to micelle transition in the ternary mixture of L121/SDS/D2O: NMR, EPR and SANS studies. Phys Chem Chem Phys. 2017;19:31747–55.
Celino M. Biomembrane solubilization mechanism by Triton X-100: a computational study of the three stage model. 2017;19:29780–94.
Zhou Q, Wu S, Gong N, Li X, Dou J, Mu M, Yu X, Yu J, Liang P. Liposomes loading sodium chloride as effective thermo-seeds for microwave ablation of hepatocellular carcinoma. Nanoscale. 2017;9:11068–76.
Jin Y, Liang X, An Y, Dai Z. Microwave-triggered smart drug release from liposomes co-encapsulating doxorubicin and salt for local combined hyperthermia and chemotherapy of cancer. Bioconjug Chem. 2016;27:2931–42.
Dobrzynska I. Association equilibria of divalent ions on the surface of liposomes formed from phosphatidylcholine. Eur Phys J E. 2019;42:1–6.
Kotynska J, Figaszewski ZA. Binding of trivalent metal ions (Al3+, In3+, La3+) with phosphatidylcholine liposomal membranes investigated by microelectrophoresis. Eur Phys J E. 2018;41:1–6.
Tiwari S, Singh K, Marangoni DG, Bahadur P. Amphiphilic star block copolymer micelles in saline as effective vehicle for quercetin solubilization. J Mol Liq. 2022;345:118259.
Patidar P, Bahadur A, Prasad K, Tiwari S, Aswal VK, Bahadur P. Synthesis, self-assembly and micellization characteristics of choline alkanoate ionic liquids in association with a star block copolymer. Colloids Surf A Physicochem Eng Asp. 2018;555:691–8.
Moore EW, Celic L, Ostrow JD. Interactions between ionized calcium and sodium taurocholate: bile salts are important buffers for prevention of calcium-containing gallstones. Gastroenterology. 1982;83:1079–89.
Lopez-Pena C, Arroyo-Maya IJ, McClements DJ. Interaction of a bile salt (sodium taurocholate) with cationic (ε-polylysine) and anionic (pectin) biopolymers under simulated gastrointestinal conditions. Food Hydrocoll. 2019;87:352–9.
Gou J, Chao Y, Liang Y, Zhang N, He H, Yin T, et al. Humid heat autoclaving of hybrid nanoparticles achieved by decreased nanoparticle concentration and improved nanoparticle stability using medium chain triglycerides as a modifier. Pharm Res. 2016;33:2140–51.
Yalcinkaya H, Feoktystov A, Gradzielski M. Formation of well-defined vesicles by styrene addition to a nonionic surfactant and their polymerization leading to viscous hybrid systems. Langmuir. 2018;34:9184–94.
Wang X, Liu L, Xia S, Muhoza B, Cai J, Zhang X, et al. Sodium carboxymethyl cellulose modulates the stability of cinnamaldehyde-loaded liposomes at high ionic strength. Food Hydrocoll. 2019;93:10–8.
Cheng C-Y, Wang T-Y, Tung S-H. Biological hydrogels formed by swollen multilamellar liposomes. Langmuir. 2015;31:13312–20.
Acknowledgements
ST acknowledges the Central Instrument Facility of NIPER-Raebareli and CRF IIT-Delhi for NMR and HR-TEM data collection.
Funding
DK and AS thank the Ministry of Chemicals and Fertilizers (Govt. of India) for providing the fellowship to carry out this research, communication no.: NIPER-R/397.
Author information
Authors and Affiliations
Contributions
Deepak Kumar and Abhishek Suna: investigation, writing—original draft. Debes Ray and V.K. Aswal: formal analysis—SANS data. Pratap Bahadur: writing—review and editing. Sanjay Tiwari: conceptualization, writing—review and editing, and project administration.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, D., Suna, A., Ray, D. et al. Structural Changes in Liposomal Vesicles in Association with Sodium Taurodeoxycholate. AAPS PharmSciTech 24, 95 (2023). https://doi.org/10.1208/s12249-023-02550-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-023-02550-7