Abstract
Doxorubicin (DOX) has a cytotoxic effect on many tumor cells; however, its clinical application is limited owing to its strong side effects. Although Doxil® reduces the cardiotoxicity of free DOX, it has also introduced a new dose-limiting toxicity. In a previous study, a sialic acid-cholesterol conjugate (SA-CH) was synthesized and modified onto the surface of DOX-loaded liposomes to target tumor-associated macrophages (TAMs), further improving the efficacy of DOX-loaded liposomes over that of Doxil®. Meanwhile, the good retention characteristics and promising antitumor ability of sphingomyelin/cholesterol (SM/CH) system for water-soluble drugs have attracted wide attention. Therefore, we aimed to use SA-CH as the target and hydrogenated soybean phosphatidylcholine (HSPC) or egg sphingomyelin (ESM) as the membrane material to develop a more stable DOX-loaded liposome with stronger antitumor activity. The liposomes were evaluated for particle size, polydispersity index, zeta potential, entrapment efficiency, in vitro release, long-term storage, cytotoxicity, cellular uptake, pharmacokinetics, tumor targetability, and in vivo antitumor activity. In the liposomes prepared using HSPC/CH, sialic acid (SA) modification considerably increased the accumulation of DOX-loaded liposomes in the tumor, thus exerting a better antitumor effect. However, SA modification in DOX-ESL (SA-CH-modified DOX-loaded liposomes prepared by ESM/CH) destroyed the strong retention effect of the ESM/CH system on DOX, resulting in a reduced antitumor effect. Notably, DOX-ECL (DOX-loaded liposome prepared by ESM/CH) had the optimal storage stability, lowest toxicity, and optimal antitumor effect due to better drug retention properties. Thus, the ESM/CH liposome of DOX is a potential drug delivery system.
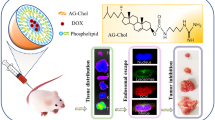
Graphical Abstract
Sketch of the effect of two DOX-loaded liposomes with hydrogenated soybean phospholipid (HSPC) and egg sphingomyelin (ESM) as lipid membrane material and surface-modified SA derivative on tumor growth inhibition.








Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- DOX:
-
Doxorubicin
- TAMs:
-
Tumor-associated macrophages
- TME:
-
Tumor microenvironment
- SA-CH:
-
Sialic acid-cholesterol conjugate
- DOX-S:
-
DOX solution
- DOX-HCL:
-
DOX-loaded liposome prepared by HSPC/CH
- DOX-HSL:
-
SA-CH-modified DOX-loaded liposome prepared by HSPC/CH
- DOX-ECL:
-
DOX-loaded liposome prepared by ESM/CH
- DOX-ESL:
-
SA-CH-modified DOX-loaded liposome prepared by ESM/CH
- DiR-HCL:
-
DiR-loaded liposome prepared by HSPC/CH
- DiR-HSL:
-
SA-CH-modified DiR-loaded liposome prepared by HSPC/CH
- DiR-ECL:
-
DiR-loaded liposome prepared by ESM/CH
- DiR-ESL:
-
SA-CH-modified DiR-loaded liposome prepared by ESM/CH
References
Chen Y, Du Q, Zou Y, et al. Co-delivery of doxorubicin and epacadostat via heparin coated pH-sensitive liposomes to suppress the lung metastasis of melanoma. Int J Pharm. 2020;584: 119446.
Apetoh L, Ghiringhelli F, Tesniere A, et al. Toll-like receptor 4–dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med. 2007;13:1050–9.
Von Hoff DD, Layard MW, Basa P, et al. Risk factors for doxorubicin-induced congestive heart failure. Ann Intern Med. 1979;91:710–7.
Lefrak EA, Piťha J, Rosenheim S, Gottlieb JA. A clinicopathologic analysis of adriamycin cardiotoxicity. Cancer. 1973;32:302–14.
Singal PK, Deally CM, Weinberg LE. Subcellular effects of adriamycin in the heart: a concise review. J Mol Cell Cardiol. 1987;19:817–28.
Bangham AD, Horne RW. Negative staining of phospholipids and their structural modification by surface-active agents as observed in the electron microscope. J Mol Biol. 1964;8:660–8.
Akbarzadeh A, Rezaei-Sadabady R, Davaran S, et al. Liposome: classification, preparation, and applications. Nanoscale Res Lett. 2013;8:1–9.
Wagner A, Vorauer-Uhl K. Liposome technology for industrial purposes. J Drug Deliv. 2011;2011:591325.
Working P, Dayan A. Pharmacological-toxicological expert report CAELYXTM:(stealth® liposomal doxorubicin HCl). Hum Exp Toxicol. 1996;15:751–85.
Ranson MR, Cheeseman S, White S, Margison J. Caelyx (stealth liposomal doxorubicin) in the treatment of advanced breast cancer. Crit Rev Oncol Hematol. 2001;37:115–20.
Rastogi M, Saha RN, Alexander A, et al. Role of stealth lipids in nanomedicine-based drug carriers. Chem Phys Lipids. 2021;235: 105036.
Waterhouse DN, Tardi PG, Mayer LD, Bally MB. A comparison of liposomal formulations of doxorubicin with drug administered in free form: changing toxicity profiles. Drug Saf. 2001;24:903–20.
Gabizon A, Martin F. Polyethylene glycol-coated (pegylated) liposomal doxorubicin. Rationale for use in solid tumours. Drugs. 1997;54:15–21.
Skubitz KM. Phase II trial of pegylated-liposomal doxorubicin (Doxil™) in renal cell cancer. Invest New Drugs. 2002;20:101–4.
Maung K, O’Shaughnessy J, Overmoyer B. Pegylated liposomal doxorubicin (Doxil®) versus doxorubicin for first-line treatment of metastatic breast cancer. Clin Breast Cancer. 2002;3:183–4.
Ding J, Zhao D, Hu Y, et al. Terminating the renewal of tumor-associated macrophages: a sialic acid-based targeted delivery strategy for cancer immunotherapy. Int J Pharm. 2019;571: 118706.
Zhou S, Zhang T, Peng B, et al. Targeted delivery of epirubicin to tumor-associated macrophages by sialic acid-cholesterol conjugate modified liposomes with improved antitumor activity. Int J Pharm. 2017;523:203–16.
Schauer R, Kamerling JP. Exploration of the sialic acid world. Adv Carbohydr Chem Biochem. 2018;75:1–213.
Brown GD, Willment JA, Whitehead L. C-type lectins in immunity and homeostasis. Nat Rev Immunol. 2018;18:374–89.
Ding D, Yao Y, Zhang S, et al. C-type lectins facilitate tumor metastasis. Oncol Lett. 2017;13:13–21.
Biswas SK, Allavena P, Mantovani A. Tumor-associated macrophages: functional diversity, clinical significance, and open questions. In: Seminars in immunopathology. Springer; 2013. p. 585–600.
Grabowska J, Lopez-Venegas MA, Affandi AJ, Den Haan JM. CD169+ macrophages capture and dendritic cells instruct: the interplay of the gatekeeper and the general of the immune system. Front Immunol. 2018;9:2472.
Li C, Qiu Q, Gao X, et al. Sialic acid conjugate-modified liposomal platform modulates immunosuppressive tumor microenvironment in multiple ways for improved immune checkpoint blockade therapy. J Control Release. 2021;337:393–406.
Chen M, Wu W, Wang S, et al. Neutrophils as emerging immunotherapeutic targets: indirect treatment of tumors by regulating the tumor immune environment based on a sialic acid derivative-modified nanocomplex platform. Int J Pharm. 2022;620: 121684.
McIntosh TJ, Simon SA, Needham D, Huang CH. Structure and cohesive properties of sphingomyelin/cholesterol bilayers. Biochemistry. 1992;31:2012–20.
Barenholz Y, Thompson TE. Sphingomyelin: biophysical aspects. Chem Phys Lipids. 1999;102:29–34.
Li J, Wang X, Zhang T, et al. A review on phospholipids and their main applications in drug delivery systems. Asian J Pharm Sci. 2015;10:81–98.
Webb M, Harasym T, Masin D, et al. Sphingomyelin-cholesterol liposomes significantly enhance the pharmacokinetic and therapeutic properties of vincristine in murine and human tumour models. Br J Cancer. 1995;72:896–904.
Semple SC, Leone R, Wang J, et al. Optimization and characterization of a sphingomyelin/cholesterol liposome formulation of vinorelbine with promising antitumor activity. J Pharm Sci. 2005;94:1024–38.
Liu M, Luo X, Qiu Q, et al. Redox-and pH-sensitive glycan (polysialic acid) derivatives and F127 mixed micelles for tumor-targeted drug delivery. Mol Pharm. 2018;15:5534–45.
Ding J, Sui D, Liu M, et al. Sialic acid conjugate-modified liposomes enable tumor homing of epirubicin via neutrophil/monocyte infiltration for tumor therapy. Acta Biomater. 2021;134:702–15.
Webb MS, Saxon D, Wong FM, et al. Comparison of different hydrophobic anchors conjugated to poly (ethylene glycol): effects on the pharmacokinetics of liposomal vincristine. Biochim Biophys Acta. 1998;1372:272–82.
Zheng H, Li J, Wang M, et al. Exhausting tumor associated macrophages with sialic acid-polyethyleneimine-cholesterol modified liposomal doxorubicin for enhancing sarcoma chemotherapy. Int J Pharm. 2019;558:187–200.
Karmakar S, Bhaumik SK, Paul J, De T. Leishmania donovani cell surface sialoglycans regulate susceptibility for siglec mediated macrophage invasion and parasite survival. J Mol Biochem. 2012;1:6–20.
Slotte JP. Sphingomyelin–cholesterol interactions in biological and model membranes. Chem Pys Lipids. 1999;102:13–27.
Boman NL, Cullis PR, Mayer LD, et al. Liposomal vincristine: the central role of drug retention in defining therapeutically optimized anticancer formulations. In: Long circulating liposomes: old drugs, new therapeutics. Springer; 1998. p. 29–49.
Bhuvaneswaran C, Mitropoulos KA. Effect of liposomal phospholipid composition on cholesterol transfer between microsomal and liposomal vesicles. Biochem J. 1986;238:647–52.
Allen T, Everest J. Effect of liposome size and drug release properties on pharmacokinetics of encapsulated drug in rats. J Pharmacol Exp Ther. 1983;226:539–44.
Senior J, Gregoriadis G. Stability of small unilamellar liposomes in serum and clearance from the circulation: the effect of the phospholipid and cholesterol components. Life Sci. 1982;30:2123–36.
Luo X, Hu L, Zheng H, et al. Neutrophil-mediated delivery of pixantrone-loaded liposomes decorated with poly (sialic acid)–octadecylamine conjugate for lung cancer treatment. Drug Deliv. 2018;25:1200–12.
Byrne JD, Betancourt T, Brannon-Peppas L. Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv Drug Deliv Rev. 2008;60:1615–26.
Solinas G, Germano G, Mantovani A, Allavena P. Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. J Leukoc Biol. 2009;86:1065–73.
Qian B-Z, Pollard JWJC. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141:39–51.
Acknowledgements
The authors are grateful for the laboratory facilities support provided by Shenyang Pharmaceutical University.
Funding
This study was supported by the Career Development Support Plan for Young and Middle-aged Teachers at Shenyang Pharmaceutical University (ZDN2021009) and the Natural National Science Foundation of China (Grant Numbers 81973271).
Author information
Authors and Affiliations
Contributions
Conceptualization: Hongxia Zhang, Yihui Deng, and Yanzhi Song; data curation: Xianmin Meng, Hongxia Zhang, Yihui Deng, and Yanzhi Song; acquisition: Hongxia Zhang, Lingyan Chen, Mingqi Wang, and Kaituo Zhang; writing and analysis: Xianmin Meng; supervision: Xinrong Liu. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, X., Zhang, H., Chen, L. et al. Liposomal Doxorubicin: the Sphingomyelin/Cholesterol System Significantly Enhances the Antitumor Efficacy of Doxorubicin. AAPS PharmSciTech 24, 64 (2023). https://doi.org/10.1208/s12249-022-02489-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02489-1