Abstract
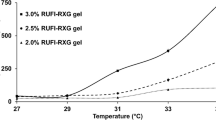
Over the past decade, intranasal (IN) delivery has been gaining attention as an alternative approach to conventional drug delivery routes targeting the brain. Carbamazepine (CBZ) is available as an orally ingestible formulation. The present study aims to develop a thermoreversible in situ gelling system for delivering CBZ via IN route. A cold method of synthesis has been used to tailor and optimize the thermoreversible gel composition, using poloxamer 407 (P407) (15–20% w/v) and iota carrageenan (ɩ-Cg) (0.15–0.25% w/v). The developed in situ gel showed gelation temperatures (28–33°C), pH (4.5–6.5), rheological properties (pseudoplastic, shear thinning), and mucoadhesive strength (1755.78–2495.05 dyne/cm2). The in vitro release study has shown sustained release behavior (24 h) for gel, containing significant retardation of CBZ release. The release kinetics fit to the Korsmeyer–Peppas model, suggesting the non-Fickian diffusion type controlled release behavior. Ex vivo permeation through goat nasal mucosa showed sustained release from the gel containing 18% P407 with the highest cumulative drug permeated (243.94 µg/cm2) and a permeation flux of 10.16 µg/cm2/h. After treatment with CBZ in situ gel, the barrier function of nasal mucosa remained unaffected. Permeation through goat nasal mucosa using in situ gel has demonstrated a harmless nasal delivery, which can provide a new dimension to deliver CBZ directly to the brain bypassing the blood–brain barrier.







Similar content being viewed by others
References
Löscher W, Potschka H, Sisodiya SM, Vezzani A. Drug resistance in epilepsy: clinical impact, potential mechanisms, and new innovative treatment options. Pharmacol Rev. 2020;72:606–38.
Elmowafy M, Shalaby K, Badran MM, Ali HM, Abdel-Bakky MS, Ibrahim HM. Multifunctional carbamazepine loaded nanostructured lipid carrier (NLC) formulation. Int J Pharm. 2018;550:359–71.
Khan N, Shah FA, Rana I, Ansari MM, Din F ud, Rizvi SZH, et al. Nanostructured lipid carriers-mediated brain delivery of carbamazepine for improved in vivo anticonvulsant and anxiolytic activity. Int J Pharm. Elsevier; 2020;577:119033.
Choudhury H, Zakaria NFB, Tilang PAB, Tzeyung AS, Pandey M, Chatterjee B, et al. Formulation development and evaluation of rotigotine mucoadhesive nanoemulsion for intranasal delivery. J Drug Deliv Sci Technol. 2019.
Patsalos PN, Spencer EP, Berry DJ. Therapeutic drug monitoring of antiepileptic drugs in epilepsy: a 2018 update. Ther. Drug Monit. 2018.
Singh RMP, Kumar A, Pathak K. Thermally triggered mucoadhesive in situ gel of loratadine: β-cyclodextrin complex for nasal delivery. AAPS PharmSciTech. 2013;14:412–24.
Mutalabisin MF, Chatterjee B, Jaffri JM. PH responsive polymers in drug delivery. Res J Pharm Technol. 2018;11:5115.
Shelke S, Shahi S, Jalalpure S, Dhamecha D, Shengule S. Formulation and evaluation of thermoreversible mucoadhesive in-situ gel for intranasal delivery of naratriptan hydrochloride. J Drug Deliv Sci Technol. 2015.
Ahmad N, Ahmad R, Ahmad FJ, Ahmad W, Alam MA, Amir M, et al. Poloxamer-chitosan-based Naringenin nanoformulation used in brain targeting for the treatment of cerebral ischemia. Saudi J Biol Sci. 2020.
Zhang L, Pang L, Zhu S, Ma J, Li R, Liu Y, et al. Intranasal tetrandrine temperature-sensitive in situ hydrogels for the treatment of microwave-induced brain injury. Int J Pharm. 2020.
Ur-Rehman T, Tavelin S, Gröbner G. Chitosan in situ gelation for improved drug loading and retention in poloxamer 407 gels. Int J Pharm. 2011.
Ourani-Pourdashti S, Mirzaei E, Heidari R, Ashrafi H, Azadi A. Preparation and evaluation of niosomal chitosan-based in situ gel formulation for direct nose-to-brain methotrexate delivery. Int J Biol Macromol [Internet]. 2022;213:1115–26. Available from: https://www.sciencedirect.com/science/article/pii/S0141813022012314. Accessed 28 June 2022
Kojarunchitt T, Hook S, Rizwan S, Rades T, Baldursdottir S. Development and characterisation of modified poloxamer 407 thermoresponsive depot systems containing cubosomes. Int J Pharm. 2011.
Li C, Li C, Liu Z, Li Q, Yan X, Liu Y, et al. Enhancement in bioavailability of ketorolac tromethamine via intranasal in situ hydrogel based on poloxamer 407 and carrageenan. Int J Pharm. 2014.
Okur NÜ, Yozgatlı V, Okur ME, Yoltaş A, Siafaka PI. Improving therapeutic efficacy of voriconazole against fungal keratitis: thermo-sensitive in situ gels as ophthalmic drug carriers. J Drug Deliv Sci Technol Elsevier. 2019;49:323–33.
Gugleva V, Titeva S, Ermenlieva N, Tsibranska S, Tcholakova S, Rangelov S, et al. Development and evaluation of doxycycline niosomal thermoresponsive in situ gel for ophthalmic delivery. Int J Pharm. Elsevier; 2020;591:120010.
Verekar RR, Gurav SS, Bolmal U. Thermosensitive mucoadhesive in situ gel for intranasal delivery of Almotriptan malate: formulation, characterization, and evaluation. J Drug Deliv Sci Technol. 2020.
Qian S, Wong YC, Zuo Z. Development, characterization and application of in situ gel systems for intranasal delivery of tacrine. Int J Pharm Elsevier. 2014;468:272–82.
Majithiya RJ, Ghosh PK, Umrethia ML, Murthy RSR. Thermoreversible-mucoadhesive gel for nasal delivery of sumatriptan. AAPS PharmSciTech. 2006;7:67.
Nižić Nodilo L, Perkušić M, Ugrina I, Špoljarić D, Jakobušić Brala C, Amidžić Klarić D, et al. In situ gelling nanosuspension as an advanced platform for fluticasone propionate nasal delivery. Eur J Pharm Biopharm Off J Arbeitsgemeinschaft fur Pharm Verfahrenstechnik eV. Netherlands; 2022;175:27–42.
Chen Y, Liu Y, Xie J, Zheng Q, Yue P, Chen L, et al. Nose-to-brain delivery by nanosuspensions-based in situ gel for breviscapine. Int J Nanomedicine. 2020.
Bai L, Lei F, Luo R, Fei Q, Zheng Z, He N, et al. Development of a thermosensitive in-situ gel formulations of vancomycin hydrochloride: design, preparation, in vitro and in vivo evaluation. J Pharm Sci: Elsevier; 2022.
Xu X, Shen Y, Wang W, Sun C, Li C, Xiong Y, et al. Preparation and in vitro characterization of thermosensitive and mucoadhesive hydrogels for nasal delivery of phenylephrine hydrochloride. Eur J Pharm Biopharm. Elsevier B.V.; 2014;88:998–1004.
Zadbuke N, Shahi S, Jadhav A, Borde S. Development and validation of UV-visible spectroscopic method for estimation of carbamazepine in bulk and tablet dosage form. Int J Pharm Pharm Sci. 2016;8:234–8.
Galgatte UC, Kumbhar AB, Chaudhari PD. Development of in situ gel for nasal delivery: design, optimization, in vitro and in vivo evaluation. Drug Deliv. 2014;21:62–73.
Chen Y, Cheng G, Hu R, Chen S, Lu W, Gao S, et al. A nasal temperature and pH dual-responsive in situ gel delivery system based on microemulsion of huperzine A: formulation, evaluation, and in vivo pharmacokinetic study. AAPS PharmSciTech. Springer; 2019;20:301.
Omar MM, Eleraky NE, El Sisi AM, Hasan OA. Development and evaluation of in-situ nasal gel formulations of nanosized transferosomal sumatriptan: design, optimization, in vitro and in vivo evaluation. Drug Des Devel Ther. Dove Press; 2019;13:4413.
Ban E, Park M, Jeong S, Kwon T, Kim EH, Jung K, et al. Poloxamer-based thermoreversible gel for topical delivery of emodin: influence of P407 and P188 on solubility of emodin and its application in cellular activity screening. Molecules. 2017;22.
Kempwade A, Taranalli A. Formulation and evaluation of thermoreversible, mucoadhesive in situ intranasal gel of rizatriptan benzoate. J Sol-Gel Sci Technol. 2014;72:43–8.
Khutoryanskiy VV. Advances in mucoadhesion and mucoadhesive polymers. Macromol Biosci Wiley Online Library. 2011;11:748–64.
Makshakova ON, Faizullin DA, Zuev YF. Interplay between secondary structure and ion binding upon thermoreversible gelation of κ -carrageenan. Carbohydr Polym. Elsevier; 2020;227:115342.
Garg T, K Goyal A. Liposomes: targeted and controlled delivery system. Drug Deliv Lett. Bentham Science Publishers; 2014;4:62–71.
Mahajan HS, Shah SK, Surana SJ. Nasal in situ gel containing hydroxy propyl β-cyclodextrin inclusion complex of artemether: development and in vitro evaluation. J Incl Phenom Macrocycl Chem Springer. 2011;70:49–58.
Abdulla NA, Balata GF, El-ghamry HA, Gomaa E. Intranasal delivery of Clozapine using nanoemulsion-based in-situ gels: an approach for bioavailability enhancement. Saudi Pharm J. 2021.
Sethia S, Squillante E. Solid dispersion of carbamazepine in PVP K30 by conventional solvent evaporation and supercritical methods. Int J Pharm. 2004;272:1–10.
Gómez-Ordóñez E, Rupérez P. FTIR-ATR spectroscopy as a tool for polysaccharide identification in edible brown and red seaweeds. Food Hydrocoll Elsevier Ltd. 2011;25:1514–20.
Ali W, Williams AC, Rawlinson CF. Stochiometrically governed molecular interactions in drug: poloxamer solid dispersions. Int J Pharm Elsevier. 2010;391:162–8.
Marzouk MA, Osman DA, Abd El-Fattah AI. Formulation and in vitro evaluation of a thermoreversible mucoadhesive nasal gel of itopride hydrochloride. Drug Dev Ind Pharm. Taylor & Francis; 2018;44:1857–67.
Gadad AP, Wadklar PD, Dandghi P, Patil A. Thermosensitive in situ gel for ocular delivery of lomefloxacin. Indian J Pharm Educ Res. 2016;50:S96-105.
Mahajan HS, Gattani S. In situ gels of metoclopramide hydrochloride for intranasal delivery: in vitro evaluation and in vivo pharmacokinetic study in rabbits. Drug Deliv. 2010;17:19–27.
Cai Z, Song X, Sun F, Yang Z, Hou S, Liu Z. Formulation and evaluation of in situ gelling systems for intranasal administration of gastrodin. AAPS PharmSciTech. 2011.
Liu Y, Lu W-L, Wang J-C, Zhang X, Zhang H, Wang X-Q, et al. Controlled delivery of recombinant hirudin based on thermo-sensitive Pluronic® F127 hydrogel for subcutaneous administration: in vitro and in vivo characterization. J Control release Elsevier. 2007;117:387–95.
Liu Y, Zhu Y ying, Wei G, Lu W yue. Effect of carrageenan on poloxamer-based in situ gel for vaginal use: improved in vitro and in vivo sustained release properties. Eur J Pharm Sci. 2009.
Coviello T, Matricardi P, Marianecci C, Alhaique F. Polysaccharide hydrogels for modified release formulations. J Control Release. 2007;119:5–24.
Funding
The research was conducted under the Fundamental Research Grant Scheme (FRGS 17-006-0572), sponsored by the Ministry of Higher Education, Malaysia.
Author information
Authors and Affiliations
Contributions
Keithanchali Mohonanaidu (first author): experimental work and data analysis and writing the manuscript.
Bappaditya Chatterjee (corresponding author): conceptualize the idea and framing the research, review and technical editing of the manuscript, helping fabrication of gel, and responding reviewers’ comments.
Farahidah Mohamed (corresponding author): conceptualize the idea and framing the research, helping physicochemical evaluation of gel, mucoadhesion study, and review of the manuscript.
Syed Mahmood (co-author): ex vivo permeation study and its data interpretation.
Samah Hamed (co-author): rheological analysis and its data interpretation.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohananaidu, K., Chatterjee, B., Mohamed, F. et al. Thermoreversible Carbamazepine In Situ Gel for Intranasal Delivery: Development and In Vitro, Ex Vivo Evaluation. AAPS PharmSciTech 23, 288 (2022). https://doi.org/10.1208/s12249-022-02439-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02439-x