Abstract
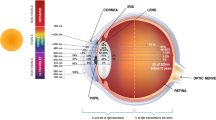
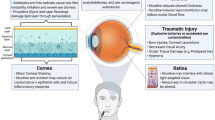
Papaverine, a poorly soluble opium alkaloid, has recently been shown to reduce retinal inflammation due to which it may have therapeutic application in the management of Leber’s hereditary optic neuropathy. In this study, papaverine eyedrops based on medium chain triglycerides were prepared and the effect of diethyl glycol monoethyl ether (DGME) on their ocular distribution was evaluated using an ex vivo porcine eye model. The route of drug penetration was also studied by orienting the eye to expose either only the cornea or the sclera to the formulation. Furthermore, in vivo studies were performed to confirm ocular tolerability and evaluate ocular drug distribution. Our results showed increased papaverine concentrations in the cornea and sclera in the presence of DGME but with a slight reduction in the retina-choroid (RC) drug concentration when administered via the corneal route, suggesting that DGME enhances drug accumulation in the anterior ocular tissues but with little effect on posterior drug delivery. In vivo, the papaverine eyedrop with DGME showed good ocular tolerability with the highest drug concentration being observed in the cornea (1.53 ± 0.28 μg/g of tissue), followed by the conjunctiva (0.74 ± 0.18 μg/g) and sclera (0.25 ± 0.06 μg/g), respectively. However, no drug was detected in the RC, vitreous humor or plasma. Overall, this study highlighted that DGME influences ocular distribution and accumulation of papaverine. Moreover, results suggest that for hydrophobic drugs dissolved in hydrophobic non-aqueous vehicles, transcorneal penetration via the transuveal pathway may be the predominant route for drug penetration to posterior ocular tissues.

Graphical abstract



Similar content being viewed by others
References
Triner L, Vulliemoz Y, Schwartz I, Nahas GG. Cyclic phosphodiesterase activity and the action of papaverine. Biochem Biophys Res Commun. 1970;40(1):64–9.
Resch H, Weigert G, Karl K, Pemp B, Garhofer G, Schmetterer L. Effect of systemic moxaverine on ocular blood flow in humans. Acta Ophthalmol. 2009;87(7):731–5.
Pemp B, Garhofer G, Lasta M, Schmidl D, Wolzt M, Schmetterer L. The effects of moxaverine on ocular blood flow in patients with age-related macular degeneration or primary open angle glaucoma and in healthy control subjects. Acta Ophthalmol. 2012;90(2):139–45.
Lee Y-Y, Park J-S, Leem Y-H, Park J-E, Kim D-Y, Choi Y-H, et al. The phosphodiesterase 10 inhibitor papaverine exerts anti-inflammatory and neuroprotective effects via the PKA signaling pathway in neuroinflammation and Parkinson’s disease mouse models. J Neuroinflamm. 2019;16(1):246.
Yu AK, Datta S, McMackin MZ, Cortopassi GA. Rescue of cell death and inflammation of a mouse model of complex 1-mediated vision loss by repurposed drug molecules. Hum Mol Genet. 2017;26(24):4929–36.
Yu AK, Song L, Murray KD, van der List D, Sun C, Shen Y, et al. Mitochondrial complex I deficiency leads to inflammation and retinal ganglion cell death in the Ndufs4 mouse. Hum Mol Genet. 2015;24(10):2848–60.
Datta S, Tomilov A, Cortopassi G. Identification of small molecules that improve ATP synthesis defects conferred by Leber’s hereditary optic neuropathy mutations. Mitochondrion. 2016;30:177–86.
Ahmed I. The noncorneal route in ocular drug delivery. In: Mitra AK, editor. Ophthalmic drug delivery systems. New York: Marcel Dekker; 2003. p. 335–63.
Bodor N, Buchwald P. Ophthalmic drug design based on the metabolic activity of the eye: soft drugs and chemical delivery systems. AAPS J. 2005;7(4):E820–E33.
Maurice DM, Mishima S. Ocular pharmacokinetics. In: Sears ML, editor. Pharmacology of the eye. Berlin: Springer Berlin Heidelberg; 1984. p. 19–116.
Maurice DM. Drug delivery to the posterior segment from drops. Surv Ophthalmol. 2002;47(Suppl 1):S41–52.
Gomes JAP, Azar DT, Baudouin C, Efron N, Hirayama M, Horwath-Winter J, et al. TFOS DEWS II iatrogenic report. Ocul Surf. 2017;15(3):511–38.
Ghate D, Edelhauser HF. Barriers to glaucoma drug delivery. J Glaucoma. 2008;17(2):147–56.
Ishibashi T, Yokoi N, Kinoshita S. Comparison of the short-term effects on the human corneal surface of topical timolol maleate with and without benzalkonium chloride. J Glaucoma. 2003;12(6):486–90.
Datta S, Baudouin C, Brignole-Baudouin F, Denoyer A, Cortopassi GA. The eye drop preservative benzalkonium chloride potently induces mitochondrial dysfunction and preferentially affects LHON mutant cells. Invest Ophthalmol Vis Sci. 2017;58(4):2406–12.
Agarwal P, Khun D, Krösser S, Eickhoff K, Wells FS, Willmott GR, et al. Preclinical studies evaluating the effect of semifluorinated alkanes on ocular surface and tear fluid dynamics. Ocul Surf. 2019.
Agarwal P, Scherer D, Günther B, Rupenthal ID. Semifluorinated alkane based systems for enhanced corneal penetration of poorly soluble drugs. Int J Pharm. 2018;538(1):119–29.
Osborne DW, Musakhanian J. Skin penetration and permeation properties of Transcutol®—neat or diluted mixtures. AAPS PharmSciTech. 2018;19(8):3512–33.
Box K, Comer J, Huque F. (2006), Correlations between PAMPA permeability and log P. In: Pharmacokinetic profiling in drug research. p. 243-57.
Agarwal P, Craig JP, Krösser S, Eickhoff K, Swift S, Rupenthal ID. Topical semifluorinated alkane-based azithromycin suspension for the management of ocular infections. Eur J Pharm Biopharm. 2019;142:83–91.
Griffith JF, Nixon GA, Bruce RD, Reer PJ, Bannan EA. Dose-response studies with chemical irritants in the albino rabbit eye as a basis for selecting optimum testing conditions for predicting hazard to the human eye. Top Catal. 1980;55(3):501–13.
Draize JH, Woodard G, Calvery HO. Methods for the study of irritation and toxicity of substances applied topically to the skin and mucous membranes. J Pharmacol Exp Ther. 1944;82(3):377–90.
Liu Z, Nie S, Guo H, Pan W, Li J. Effects of Transcutol P on the corneal permeability of drugs and evaluation of its ocular irritation of rabbit eyes. J Pharm Pharmacol. 2006;58(1):45–50.
Li X, Nie S-F, Kong J, Li N, Ju C-Y, Pan W-S. A controlled-release ocular delivery system for ibuprofen based on nanostructured lipid carriers. Int J Pharm. 2008;363(1):177–82.
Varela-Fernández R, Díaz-Tomé V, Luaces-Rodríguez A, Conde-Penedo A, García-Otero X, Luzardo-Álvarez A, et al. Drug delivery to the posterior segment of the eye: biopharmaceutic and pharmacokinetic considerations. Pharmaceutics. 2020;12(3):269.
Ghorbanzade M, Fatemi MH, Karimpour M, Andersson PL. Quantitative and qualitative prediction of corneal permeability for drug-like compounds. Talanta. 2011;85(5):2686–94.
Asano N, Wiseman JM, Tsuji F, Kawazu K. Limited azithromycin localization to rabbit meibomian glands revealed by LC-MS-based bioanalysis and DESI imaging. Biol Pharm Bull. 2017;40(9):1586–9.
Jakubiak P, Cantrill C, Urtti A, Alvarez-Sánchez R. Establishment of an in vitro–in vivo correlation for melanin binding and the extension of the ocular half-life of small-molecule drugs. Mol Pharm. 2019;16(12):4890–901.
Shaaya AN, Kraus C, Bauman DH, Ritschel WA. Pharmacokinetics and bioavailability of papaverine HCl after intravenous, intracorporeal and penis topical administration in beagle dogs. J Ocul Pharmacol Ther. 1992;14(5):373–8.
Kraus C, Shaaya A, Ulmer J, Hutchings D, Menon A, Sakr A, et al. Pharmacokinetics and bioavailability of papaverine HCl following intravenous, peroral, rectal, vaginal, topical and buccal administration in beagle dogs. Biopharm Drug Dispos. 1991;12(7):537–46.
Wen MM, El-kamel A, Khalil S. Systemic enhancement of papaverine transdermal gel for erectile dysfunction. Drug Dev Ind Pharm. 2011;38:912–22.
Huang D, Chen YS, Rupenthal ID. Overcoming ocular drug delivery barriers through the use of physical forces. Adv Drug Deliv Rev. 2018;126:96–112.
Hamalainen KM, Kananen K, Auriola S, Kontturi K, Urtti A. Characterization of paracellular and aqueous penetration routes in cornea, conjunctiva, and sclera. Invest Ophthalmol Vis Sci. 1997;38(3):627–34.
Agrahari V, Mandal A, Agrahari V, Trinh HM, Joseph M, Ray A, et al. A comprehensive insight on ocular pharmacokinetics. Drug Deliv Transl Res. 2016;6(6):735–54.
Panchagnula R, Ritschel WA. Development and evaluation of an intracutaneous depot formulation of corticosteroids using Transcutol as a cosolvent: in-vitro, ex-vivo and in-vivo rat studies. J Pharm Pharmacol. 1991;43(9):609–14.
Becker U, Ehrhardt C, Schaefer UF, Gukasyan HJ, Kim K-J, Lee VH, et al. Tissue distribution of moxaverine–hydrochloride in the rabbit eye and plasma. J Ocul Pharmacol Ther. 2005;21(3):210–6.
Shibata Y, Tanaka Y, Tomita T, Taogoshi T, Kimura Y, Chikama T, et al. Evaluation of corneal damage caused by iodine preparations using human corneal epithelial cells. Jpn J Ophthalmol. 2014;58(6):522–7.
Kuwano M, Ibuki H, Morikawa N, Ota A, Kawashima Y. Cyclosporine A formulation affects its ocular distribution in rabbits. Pharm Res. 2002;19(1):108–11.
Earla R, Boddu SH, Cholkar K, Hariharan S, Jwala J, Mitra AK. Development and validation of a fast and sensitive bioanalytical method for the quantitative determination of glucocorticoids--quantitative measurement of dexamethasone in rabbit ocular matrices by liquid chromatography tandem mass spectrometry. J Pharm Biomed Anal. 2010;52(4):525–33.
Cholkar K, Gilger BC, Mitra AK. Topical, aqueous, clear cyclosporine formulation design for anterior and posterior ocular delivery. Transl Vis Sci Techn. 2015;4(3):1.
Acknowledgements
The authors would like to thank Tarnagulla Ventures Pty. Ltd. for funding this study. IDR’s directorship is provided by the Buchanan Charitable Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Qingguo Xu and Iok-Hou Pang
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Agarwal, P., Behera, S. & Rupenthal, I.D. Ocular Distribution of Papaverine Using Non-aqueous Vehicles. AAPS PharmSciTech 22, 160 (2021). https://doi.org/10.1208/s12249-021-02050-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-021-02050-6