Abstract
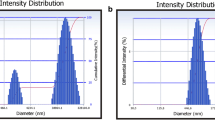
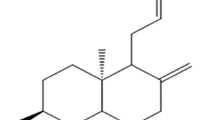
The present research focuses on the development of a nanoparticulate (nanocrystals-loaded) rapidly dissolving (orodispersible) tablet with improved solubility and bioavailability. The nanosuspension (NS) was prepared by antisolvent sonoprecipitation technique and the optimized NS was lyophilized to obtain nanocrystals (NCs), which were evaluated for various parameters. The nitrendipine (NIT) nanoparticulate orodispersible tablet (N-ODT) was prepared by direct compression method. The optimized N-ODT was evaluated for pre and post compression characteristics, in vivo pharmacokinetic and stability profile. The optimized NS showed a particle size of 505.74 ± 15.48 nm with a polydispersity index (PDI) of 0.083 ± 0.006. The % NIT content in the NCs was found to be 78.4 ± 2.3%. The saturation solubility of NIT was increased remarkably (26.14 times) in comparison to plain NIT, post NCs development. The DSC and p-XRD analysis of NCs revealed the perseverance of the integrity and crystallinity of NIT on lyophilization. The results of micromeritic studies revealed the good flow-ability and compressibility of NCs blend. All the post-compression properties of N-ODT were observed within the standard intended limit. The dispersion, wetting, and disintegration time of the optimized batch of N-ODT was found to be 39 ± 1.13 s, 44.66 ± 1.52 s, and 33.91 ± 0.94 s respectively. The in vitro dissolution study displayed 100.28 ± 2.64% and 100.61 ± 3.3% of NIT released from NCs (in 8 min) and N-ODT (in 6 min) respectively, while conventional NIT tablet took 30 min to release 99.94 ± 1.57% of NIT. The in vivo pharmacokinetic study in rabbits demonstrated significantly (p < 0.05) higher bioavailability of NIT on release from N-ODT than the conventional NIT tablet. Thus, N-ODT could be a promising tool for improving the solubility and bioavailability of NIT and to treat cardiovascular diseases effectively.







Similar content being viewed by others
Data Availability
Applicable
References
Meera C, Anthony C, Clare H. The use of nanotechnology in cardiovascular disease. Applied Nanoscience. 2018;8:1607-1619. https://doi.org/10.1007/s13204-018-0856-z.
Keck CM, Müller RH. Drug nanocrystals of poorly soluble drugs produced by high pressure homogenization. Eur J Pharm Biopharm. 2006;62:3–16. https://doi.org/10.1016/j.ejpb.2005.05.009.
Tripathi KD. Essentials of Medical. Pharmacology. 2003:494–6.
Xia D, Quan P, Piao H, Sun S, Yin Y, Cui F. Preparation of stable nitrendipine nanosuspensions using the precipitation–ultrasonication method for enhancement of dissolution and oral bioavailability. Eur J Pharm Sci. 2010;40(4):325–34. https://doi.org/10.1016/j.ejps.2010.04.006.
Quan P, Xia D, Piao H, Shi K, Jia Y, Cui F. Nitrendipine nanocrystals: its preparation, characterization, and in vitro-in vivo evaluation. AAPS PharmSciTech. 2011;12(4):1136–43. https://doi.org/10.1208/s12249-011-9682-2.
Junyaprasert VB, Morakul B. Nanocrystals for enhancement of oral bioavailability of poorly water-soluble drugs. Asian J Pharm Sci. 2015;10(1):13–23. https://doi.org/10.1016/j.ajps.2014.08.005.
Chen H, Khemtong C, Yang X, Chang X, Gao J. Nanonization strategies for poorly water-soluble drugs. Drug Discov Today. 2010;16(7-8):354–60. https://doi.org/10.1016/j.drudis.2010.02.009.
Bajaj A, Rao M, Pardeshi A, Sali D. Nanocrystallization by evaporative antisolvent technique for solubility and bioavailability enhancement of telmisartan. AAPS PharmSciTech. 2012;13(4):1331–40. https://doi.org/10.1208/s12249-012-9860-x.
Sinha B, Muller RH, Muschwitzer JP. Bottom-up approaches for preparing drug nanocrystals: formulations and factors affecting particle size. Int J Pharm. 2013;453(1):126–41. https://doi.org/10.1016/j.ijpharm.2013.01.019.
Pardeike J, Strohmeier DM, Schrödl N, Voura C, Gruber M. Nanosuspensions as advanced printing ink for accurate dosing of poorly soluble drugs in personalized medicines. Int J Pharm. 2011;420(1):93–100. https://doi.org/10.1016/j.ijpharm.2011.08.033.
Rabinow BE. Nanosuspensions in drug delivery. Nat Rev Drug Discov. 2004;3(9):785–96. https://doi.org/10.1038/nrd1494.
Fu Q, Guo M, He Z. Comparison of solid dispersion and nanosuspension for improvement of drug absorption. Asian J Pharm Sci. 2016;11:10–1. https://doi.org/10.1016/j.ajps.2015.10.009.
Rao JP, Kurt EG. Polymer nanoparticles: preparation techniques and size-control parameters. Prog.Polym.Sci. 2011;36(7):887–913. https://doi.org/10.1016/j.progpolymsci.2011.01.001.
Jermain SV, Brough C, Williams RO. Amorphous solid dispersions and nanocrystal technologies for poorly water-soluble drug delivery-An update. Int J Pharm. 2018;535:379–92. https://doi.org/10.1016/j.ijpharm.2017.10.051.
Junghanns JUA, Müller RH. Nanocrystal technology, drug delivery and clinical applications. Int J Nanomedicine. 2008;3(3):295–309. https://doi.org/10.2147/ijn.s595.
Reddy LH, Ghosh B. Fast-dissolving-drug-delivery-systems: a review of the literature. Indian J Pharm Sci. 2002:31–336.
Chaudhary H, Gauri S, Rathee P, Kumar V. Development and optimization of fast dissolving oro-dispersible films of granisetron HCl using Box–Behnken statistical design. Bull Fac Pharm Cairo Univ. 2013;51:193–201. https://doi.org/10.1016/j.bfopcu.2013.05.002.
Karki S, Kim H, Jeong Na S, Shin D, Jo K, Lee J, et al. Thin films as an emerging platform for drug delivery. Asian J Pharm Sci. 2016;11:559–74.
Siddiqui N, Garg G, Sharma PK. Fast dissolving tablets: preparation, characterization and evaluation: An overview. Int J Pharm Sci Rev Res. 2010;4:287–96.
Nautiyal U, Singh S, Singh R. Gopal, Kakar S. Fast dissolving tablets as a novel boon: A review. J Pharm Chem Biol Sci. 2014;2:25–6.
Kassem MAA, Elmeshad AN, Fares AR. Enhanced solubility and dissolution rate of lacidipine nanosuspension: formulation via antisolvent sonoprecipitation technique and optimization using box–behnken design. AAPS PharmSciTech. 2016;18(4):983–96. https://doi.org/10.1208/s12249-0160604-1.
Müller RH, Jacobs C. Buparvaquone mucoadhesive nanosuspension: preparation, optimization and long-term stability. Int J Pharm. 2002;237:151–61. https://doi.org/10.1016/s0378-5173(02)00040-6.
Tran TT, Tran KA, Tran PH. Modulation of particle size and molecular interactions by sonoprecipitation method for enhancing dissolution rate of poorly water-soluble drug. Ultrason-Sonochem. 2015;24:256–63. https://doi.org/10.1016/j.ultsonch.2014.11.020.
Mishra B, Sahoo J, Dixit PK. Fabrication of cinnarizine nanosuspensions by ultrasonication technique: a systematic study of formulation parameters on particle size and in-vitro dissolution. Biopharm J. 2015;1(1):12–21. https://doi.org/10.22159/ijap.2018v10i2.23075.
Skoaufa MAA. Preparation and characterization of ketoprofen nanosuspension for solubility and dissolution velocity enhancement. Int J Pharm Bio Sci. 2013;4(1):768–80.
Afifi SA, Hassan MA, Abdelhameed AS, Elkhodairy KA. Nanosuspension: an emerging trend for bioavailability enhancement of etodolac. Int J Polymer Sci. 2015;2015:1–16. https://doi.org/10.1155/2015/938594.
Patel J, Dhingani A, Garala K, Raval M, Sheth N. Design and development of solid nanoparticulate dosage forms of telmisartan for bioavailability enhancement by integration of experimental design and principal component analysis. Powder Technol. 2014;258:331–43. https://doi.org/10.1016/j.powtec.2014.03.001.
Kulkarni AS, Ghadge DM, Kokate PB. Formulation and in vitro evaluation of orally disintegrating tablets of olanzapine-2-hydroxypropyl-β-cyclodextrin inclusion complex. Iran J Pharm Res. 2010;9(4):335–47.
Chowdary YA, Soumya M, Madhu BM, Aparna K, Himabindu P. A review on fast dissolving drug delivery systems-a pioneering drug delivery technology. Bull Env Pharmacol Life Sci. 2012;1(12):8–20.
Puttewar TY. Formulation and evaluation of orodispersible tablet of taste masked doxylamine succinate using ion exchange resin. J King Saud Uni. 2010;22:229–40. https://doi.org/10.1016/j.jksus.2010.05.003.
Sandeep N, Gupta MM. Immediate drug release dosage form: a review. J Drug Deliv and Thera. 2013;3(2):155-161. https://doi.org/10.22270/jddt.v3i2.457.
Elbary AA, Ali AA, Aboud HM. Enhanced dissolution of meloxicam from orodispersible tablets prepared by different methods. Bull Fac Pharmac. 2012;50(2):89–97. https://doi.org/10.1016/j.bfopcu.2012.07.001.
Prusty A. Formulation and in-vitro evaluation of amlodipine besylate-hp-b-cd inclusion complex incorporated mouth dissolving tablets. Bull Pharm Res2014. 2014;4(3):124–8.
Winarti L, Ameliana L, Nurahmanto D. Formula optimization of orally disintegrating tablet containing meloxicam nanoparticles. Indonesian J. Pharm. 2017; 28(1): 53 – 64. https://doi.org/10.14499/indonesianjpharm28iss1pp53.
Patel NK, Jethara SI, Patel MS. A review on orodispersible tablets-As a novel formulation for oral drug delivery systems. J Pharm Sci Biosci Res. 2015;5(3):286–94.
Rewar S, Singh CJ, Bansal BK, Pareek R, Sharma AK. Oral dispersible tablets: an overview; development, technologies and evaluation. Int J Res Dev in Pharmacy and Life Sci. 2014;3(6):1223–35.
Singh S, Mandal S, Verma N. Formulation and evaluation of orodispersible tablets of ofloxacin by using different natural super disintegrating agents. Int. J. Pharma. Sci. Res. 2020; 11(2): 884-895. https://doi.org/10.13040/IJPSR.0975-8232.11(2).884-95.
Jassim ZE, Hussein AA. Formulation and evaluation of clopidogrel tablet incorporating drug nanoparticles. Int J Pharm Pharm Sci. 2014;6(1):838–51.
Hao J, Gao Y, Zhao J, Zhang J, Li Q, Liu J. Preparation and optimization of resveratrol nanosuspensions by antisolvent precipitation using Box-Behnken design. AAPS PharmSciTech. 2015;16(1):118–28. https://doi.org/10.1208/s12249-014-0211-y.
Vijayanand P, Patil JS, Reddy MV. Formulation and comparative pharmacokinetic evaluation of orodispersible tablets and films of nebivolol hydrochloride. J. Pharmceu. Inves. 2015;45:237-247. https://doi.org/10.1007/s40005-014-0169-5.
El-setouhy DA, El-malak NSA. Formulation of a novel tianeptine sodium orodispersible film. AAPS PharmSciTech. 2010;11(3):1018–25. https://doi.org/10.1208/s12249-010-9464-2.
Chatwal GR, Anand SK. Instrumental methods of chemical analysis. Himalaya Publishing House, New Delhi. 2.60-2.71.
Chauhan A. Chauhan P. Powder XRD technique and its applications in science and technology. J Anal Bioanal Tech 2014;5(6).
Desai PM, Liew CV, Heng PWS. Review of disintegrants and the disintegration phenomena. J Pharm Sci. 2016;105(9):2545–55. https://doi.org/10.1016/j.xphs.2015.12.019.
Papadimitriou S, Bikiaris D. Novel self-assembled core–shell nanoparticles based on crystalline amorphous moieties of aliphatic copolyesters for efficient controlled drug release. J Control Release. 2009;138:177–84. https://doi.org/10.1016/j.jconrel.2009.05.013.
Bhowmik D, Chiranjib B, Chandira RM. Fast dissolving tablet: an overview. J Chem Pharm Res. 2009;1(1):163–77.
Singh J, Garg R, Gupta G. Enhancement of solubility of lamotrigine by solid dispersion and development of orally disintegrating tablets using 32 full factorial design. Aust J Pharm. 2015;2015:1–8.
Tanuwijaya J. Karsono, Harahap U. Characterization of piroxicam nanoparticles in orally disintegrating tablet (ODT). Int. J. Chem Tech. Res. 2014;6(2):955–61.
Giri TK, Sa B. Preparation and evaluation of rapidly disintegrating fast release tablet of inclusion complex. Sci Res Pharmacol Pharm. 2010;01:18–26. https://doi.org/10.4236/pp.2010.11003.
Acknowledgements
The authors would like to acknowledge Government College of Pharmacy, Aurangabad (M.S.), India, Wockhardt Ltd., Aurangabad (M.S.), India and M.E.S.’s College of Pharmacy, Ahmednagar (M.S.), India, for the resource support provided.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Applicable (IAEC Approval No. - MES/COP/IAEC/04/2017-18)
Consent for Publication
Not applicable
Competing Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gandhi, N.V., Deokate, U.A. & Angadi, S.S. Development of Nanonized Nitrendipine and Its Transformation into Nanoparticulate Oral Fast Dissolving Drug Delivery System. AAPS PharmSciTech 22, 113 (2021). https://doi.org/10.1208/s12249-021-01963-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-021-01963-6